
基于一种柔性和角型有机芳香多酸配体的两种3D配位聚合物的合成、结构和荧光性
-
关键词:
- 氯化5-(4-羟基吡啶基甲基)间苯二甲酸
- / 1, 2-二(4-吡啶基)乙烷
- / 锌
- / 镉
English
Syntheses, Structures and Photoluminescent Properties of Two 3D Coordination Polymers Based on a Flexible and Angular Organic Aromatic Multi-carboxylate Ligand
-
In recent years, the complexes based on organic aromatic carboxylate ligands have become an interesting research area due to their pluridentated and excellent coordinated ability[1-4] as well as their charming architectures and potential applications in adsorption, catalysis, and luminescence, and so on[5-6]. Among the reported compounds, the coordination polymers from flexible or angular organic aromatic multi-carboxylate ligands are especially interesting because they can adopt a variety of coordination modes resulting in diverse multidimensional archite-ctures[7-10]. Up to now, many aromatic carboxylate ligands with flexible or angular structures have been synthesized and reported. For example, Li et al. have reported a series of metal-organic frameworks based on flexible ligand 4-carboxy-1-(3, 5-dicarboxy-benzyl) pyridinium chloride[11]; Sun et al. have synthesized a flexible and angular ligands 5-(imidazol-1-ylmethyl)isophthalic acid, and report a series of coordination polymers with novel networks[12-13]; Zang et al. have investigated a series of angular carboxylate ligands, 3, 5-bis(imidazol-1-ylmethyl) benzoic acid hydrochlor-ide[14], 2, 3, 2′, 3′-thiaphthalic acid[15], 1-(pyridin-4-ylthio)benzene-2, 4-dioic acid[16]. Recently, we have also synthesized a versatile ligand, H3LCl (5-(4-hydroxy-pyridinium-1-ylmethyl) isophthalic acid chloride), and its corresponding coordination polymers have been reported[17]. To continue our effort in this area, two coordination polymers, {[Zn(L)(bpa)0.5(H2O)2]·2.25H2O}n (1) and {[Cd(L)(H2O)]·2H2O}n (2), with novel networks from H2L ligand have been hydrothermally synthesized and structurally characterized by X-ray diffraction analyses and further characterized by infrared spectra (IR), elemental analyses and thermogravimetric (TG) analyses.
1 Experimental
1.1 Materials and physical measurement
H3LCl was synthesized according to the literature[17], and other starting materials were of reagent quality and obtained from commercial sources without further purification. Elemental analysis for C, H and N was performed on a Perkin-Elmer 240 elemental analyzer. The FT-IR spectra were recorded from KBr pellets in the range from 4 000 to 400 cm-1 on a Nicolet NEXUS 470-FTIR spectrometer. Thermal analysis was performed on a SDT 2960 thermal analyzer from room temperature to 800 ℃ with a heating rate of 10 ℃·min-1 under nitrogen flow. Powder X-ray diffraction (PXRD) for compounds 1 and 2 were measured at 293 K on a Rigaku D/max-3B diffractometer equipped with Cu Kα (λ=0.154 06 nm) radiation (45 kV, 200 mA). The crushed single crystalline powder samples were prepared by crushing the crystals and the 2θ scanning angle range was from 5° to 50°. Luminescence spectra for the solid samples were recorded on a Hitachi 850 fluorescence spectrophotometer.
1.2 Synthesis
1.2.1 Synthesis of {[Zn(L)(bpa)0.5(H2O)2]·2.25H2O}n (1)
Compound 1 was synthesized hydrothermally in a 25 mL Teflon-lined autoclave by heating a mixture of bpa (0.009 1 g, 0.05 mmol), Zn(OAc)·2H2O (0.022 g, 0.1 mmol), H3LCl (0.015 4 g, 0.05 mmol), and LiOH (0.004 2 g, 0.1 mmol) at 160 ℃ in 6 mL H2O for three days. Colorless block-wise crystals of 1 were obtained in 80% yield based on H2L. Anal. Calcd. for C20H23.5N2O9.25Zn(%): C 47.54, H 4.69, N 5.54; Found(%): C 47.51, H 4.73, N 5.50. IR(KBr, cm-1): 3 388(m, br), 3 120(m), 1 640(s), 1 620(s), 1 582(s), 1 556(s), 1 361(s), 1 239(w), 1 226(w), 1 193(m), 1 029(w), 847(w), 774(w), 729(w).
1.2.2 Synthesis of {[Cd(L)(H2O)]·2H2O}n (2)
A mixture of Cd(OAc)2·2H2O (0.026 g, 0.1 mmol), H2L (0.016 8 g, 0.05 mmol), LiOH (0.004 2 g, 0.1 mmol), and H2O (6 mL) was placed in a 25 mL Teflon -lined stainless steel vessel, and heated at 160 ℃ for three days. After the mixture was slowly cooled to room temperature, colorless crystals of 2 were obtained in 76% yield based on H2L. Anal. Calcd. for C14H15NO8Cd(%): C 38.42, H 3.45, N 3.30; Found(%): C 38.39, H 3.51, N 3.20. IR(KBr, cm-1): 3 410(s, br), 3 045(m), 1 647(s), 1 619(s), 1 563(m), 1 524(s), 1 447(w), 1 340(s), 1 293(w), 1 187(m), 1 049(w), 1 028(w), 856(s), 774(s), 726 (w).
1.3 Crystallographic data collection and structure determination
Single-crystal X-ray diffraction data of compo-unds 1 and 2 were collected on a Bruker SMART APEX CCD diffractometer[18] equipped with graphite monochromatized Mo Kα radiation (λ=0.071 073 nm) at room temperature using the φ-ω scan technique. Empirical absorption corrections were applied to the intensities using the SADABS program[19]. The struct-ures were solved with direct methods using the program SHELXS-97[20] and refined anisotropically with the program SHELXL-97[21] using full matrix least-squares procedures. All non-hydrogen atoms were refined anisotropically. The hydrogen atoms of the coordination water molecules, and ligands were included in the structure factor calculation at idealized positions by using a riding model and refined isotropically. The hydrogen atoms of the solvent water molecules were located from the difference Fourier maps, then restrained at fixed positions and refined isotropically. Analytical expressions of neutral atom scattering factors were employed, and anomalous dispersion corrections were incorporated. The crystall-ographic data for 1 and 2 are summarized in Table 1. Selected bond lengths and angles are given in Table 2.
Compound 1 2 Formula C20H23.5N2O9.25Zn C14HI5NO8Cd Formula weight 505.28 437.67 Crystal system Triclinic Triclinic Space group P1 P1 a/nm 0.969 30(8) 0.738 70(4) b/nm 1.066 54(9) 1.015 35(4) c/nm 1.227 35(11) 1.120 03(7) α/(°) 67.315(8) 70.191(4) β/(°) 78.218(7) 81.994(5) γ/(°) 67.830(8) 76.292(4) V/nm3 1.081 57(18) 0.766 25(7) Z 1 2 Dc/ (g·cm-3) 1.552 1.897 F(000) 523 436 Reflection collected 6 391 5 773 Independent reflection 3 811 2 687 Rint 0.058 9 0.024 9 GOF on F2 1.057 1.028 R1a[I>2σ(I)] 0.044 5 0.028 4 wR2b[I>2σ(I)] 0.110 7 0.068 9 $^{\text{a}}{R_1} = {\sum {\left| {\left| {{F_{\text{o}}}} \right|- \left| {{F_{\text{c}}}} \right|/\sum {\left| {{F_{\text{o}}}} \right|;} } \right.} ^{\text{b}}}w{R_2} = {\left[{\sum {w{{\left( {F_{\text{o}}^2-F_{\text{c}}^2} \right)}^2}} /\sum {w{{\left( {F_{\text{o}}^2} \right)}^2}} } \right]^{1/2}} $ Compound 1 Zn-O(1) 0.195 2(2) Zn-O(3)ⅰ 0.196 9(2) Zn-N(2) 0.205 5(2) Zn(1)-O(1W) 0.214 2(2) Zn(1)-O(2W) 0.229 1(2) O(1)-Zn(1)-N(2) 133.65(11) O(1)-Zn-O(3)ⅰ 125.29(10) O(1)-Zn(1)-O(1W) 88.55(10) O(3)ⅰ-Zn-N(2) 100.60(10) N(2)-Zn(1)-O(1W) 93.77(10) O(3)ⅰ-Zn(1)-О(1W) 95.37(11) O(3)ⅰ-Zn(1)-O(2W) 89.56(10) O(1)-Zn(1)-O(2W) 85.77(10) O(1W)-Zn(1)-O(2W) 173.98(9) N(2)-Zn(1)-0(2W) 88.72(10) Compound 2 Cd(1)-O(1) 0.232 3(2) Cd(1)-O(2) 0.239 8(2) Cd(1)-O(3)ⅰ 0.274 7(1) Cd(1)-O(4)ⅰ 0.228 2(3) Cd(1)-O(5)ⅱ 0.227 6(3) Cd(1)-O(1W) 0.234 2(3) Cd(1)-O(3)ⅲ 0.227 4(3) O(3)ⅰ-Cd(1)-O(4)ⅰ 50.396(2) O(5)ⅱ-Сd(1)-O(3)ⅰ 81.625(2) O(1W)-Cd(1)-O(3)ⅰ 79.926(2) O(1)-Cd(1)-O(3)ⅰ 173.275(3) O(5)ⅱ-Сd(1)-O(3)ⅲ 83.16(10) O(2)-Сd(1)-O(3)ⅰ 131.347(3) O(5)ⅱ-Cd(1)-O(4)ⅰ 93.32(10) 0(3)ⅲ-Сd(1)-O(4)ⅰ 131.81(9) O(1)-Cd(1)-O(3)ⅲ 91.73(10) Symmetry codes: ⅰ x+1, y, z for 1; ⅰ x, 1+y, z; ⅱ 2-x, 1-y, 1-z; ⅲ 3-x, 1-y, -z for 2. CCDC: 908522, 1; 908526, 2.
2 Results and discussion
2.1 Crystal structure of {[Zn(L)(bpa)0.5(H2O)2]·2.25H2O}n (1)
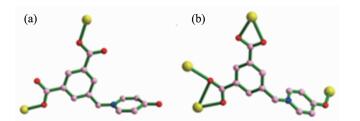
The compound 1 crystallizes in the monoclinic system with space group P1. Its asymmetric unit consists of one Zn(Ⅱ) center, half of a bpa ligand, one L2- anion, two coordinated water molecules, and three free water molecules with the total site occupancies of 2.25 (Fig. 1a). The Zn(Ⅱ) center is five-coordinated by two carboxylic oxygen atoms (O1, O4ⅰ) from two different L2- ligands, two oxygen atoms (O1W, O2W) from two water molecules and one nitrogen atom (N2) from one bpa ligand. The coordination geometry of Zn1 atom is a distorted trigonal bipyramidal with two water oxygen atoms (O1W and O2W) at the axial position (Fig. 1a). The Zn1-O bond lengths range from 0.195 2(2) to 0.229 1(2) nm, and the Zn1-N bond distance is 0.205 5(2) nm. The L2- anion bridges two Zn(Ⅱ) atoms with both two carboxylic groups in μ2-η1:η1 modes (Scheme 1a). In this way, L2- anions link neighboring Zn1 atoms to yield an infinite 1D (Zn-L)n chain. Such (Zn-L)n chains are further bridged by bpa molecules into an infinite 1D ladder-like chain (Fig. 1b). Such chains are further united together by numerous hydrogen bonds involving the coordinated waters, lattice waters and carboxyl oxygen atoms to generate a 3D supramolecular architecture (Fig. 1c). According to graph set analysis nomenclature[22], the hydrogen motifs within the water cluster can be assigned to be R42(8) and R66(16), respectively (Fig. 1d, Table 3).
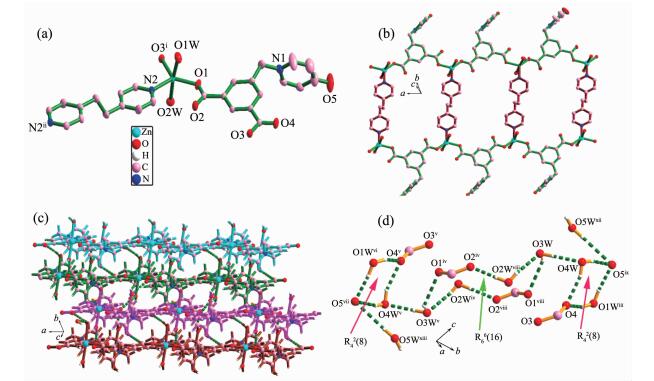 图1
(a) Metal coordination and atom labeling in compound 1 with thermal ellipsoids at 50% probability level; (b) 1D ladder-like chain; (c) View of 3D supramolecular framework of 1 via hydrogen bonds (the dashed lines represent the hydrogen bonds); (d) Hydrogen bonds in compound 1
Figure1.
(a) Metal coordination and atom labeling in compound 1 with thermal ellipsoids at 50% probability level; (b) 1D ladder-like chain; (c) View of 3D supramolecular framework of 1 via hydrogen bonds (the dashed lines represent the hydrogen bonds); (d) Hydrogen bonds in compound 1
图1
(a) Metal coordination and atom labeling in compound 1 with thermal ellipsoids at 50% probability level; (b) 1D ladder-like chain; (c) View of 3D supramolecular framework of 1 via hydrogen bonds (the dashed lines represent the hydrogen bonds); (d) Hydrogen bonds in compound 1
Figure1.
(a) Metal coordination and atom labeling in compound 1 with thermal ellipsoids at 50% probability level; (b) 1D ladder-like chain; (c) View of 3D supramolecular framework of 1 via hydrogen bonds (the dashed lines represent the hydrogen bonds); (d) Hydrogen bonds in compound 1
D-H...A d(D-H)/nm d(H...A)/nm d(D...A)/nm ∠DHA/(°) Compound 1 O(1W)-Н(1WА)...O(5)ⅹⅰ 0.085 6 0.183 1 0.267 7 169.72 O(1W)-H(1WB)...O(4)ⅲ 0.086 6 0.213 7 0.289 0 144.98 O(2W)-H(2WA)...O(2)ⅹ 0.085 1 0.188 0 0.272 9 175.72 O(2W) -H(2WB)...O(3W)ⅰ 0.072 1 0.215 6 0.282 5 154.77 O(3W)-H(3WA)...O(4w) 0.085 8 0.194 8 0.280 6 178.94 O(3W)-H(3WB)...O (1)ⅷ 0.085 0 0.233 4 0.292 3 126.69 O(4W)-H(4WA)...O(4) 0.085 0 0.199 1 0.280 5 160.05 O(4W)-H(4WB)...O (5)ⅸ 0.072 0 0.207 5 0.275 8 158.52 Compound 2 O(1W)-Н(1WА)...O(1)ⅶ 0.083 2 0.227 6 0.298 9 143.85 O(1W)-Н(1WВ)...O(5)ⅵ 0.089 7 0.203 8 0.284 8 149.66 O(2W)-H(2WA)...N(1) 0.082 2 0.242 3 0.319 0 155.83 O(2W)-H(2WC)...O (1W)ⅸ 0.085 0 0.212 9 0.298 0 179.94 O(3W)-H(2WA)...O (2W) 0.085 0 0.234 0 0.318 8 175.81 O(3 W)-H(2WC)...O (2W)ⅷ 0.080 1 0.213 9 0.281 3 141.94 Symmetry codes: ⅰ 1+x, y, z; ⅲ 1-x, 1-y, 1-z; ⅷ -1+x, y, z; ⅸ -x, 1-y, 2-z; ⅹ 2-x, -y, 1-z; ⅹⅰ 1+x, y, -1+z for 1; ⅵ 1+x, 1+y, -1+z; ⅶ 2-x, 2-y, -z; ⅷ 1-x, -y, 1-z; ⅸ 2-x, 1-y, -z for 2. 2.2 Crystal structure of {[Cd(L)(H2O)]·2H2O}n (2)
Compound 2 crystallizes in the triclinic space group P1. The asymmetric unit contains one Cd(Ⅱ) center, one L2- anion, one coordinated water molecule, and two lattice water molecules. The Cd1 is seven-coordinated in a pentagonal bipyramidal geometry. As shown in Fig. 2a, five carboxylate oxygen atoms (O1, O2, O3ⅰ, O4ⅰ, and O3ⅲ) from three distinct L2- anions form the equatorial plane; O5ⅱ (HO-L2) and O1W (H2O) occupy the axial positions. The bond length of Cd1-O is in the range of 0.227 4(3)~0.239 8(2) nm. Each L2- ligand bridges four crystallographically identical Cd1 ions with one of the carboxylate groups in a μ2-η1:η1 mode, while the other in a μ3-η1:η2 coordination mode and the hydroxyl group in monodentate coordination mode (Scheme 1b). Thus, two cadmium ions are first linked by two L2- anions via the carboxylate groups and monodentate hydroxyl group to generate a [Cd2(L)2] metallamacrocycle with the Cd-Cd distance being 1.129 1 nm (Fig. 2b). Each metallamacrocycle links two distinct metallamacrocycle together to form the tube (Fig. 2c). The carboxylate groups in μ3-η1:η2 coordination mode further participate in the coordination with Cd1 ions of the tube. Thus, the tubes are connected together to give rise to a 2D layer with tubular channels (Fig. 2d). Furthermore, the 2D layers are further extended into 3D supramolecular architecture via hydrogen-bonding interactions (Fig. 2e, Table 3).
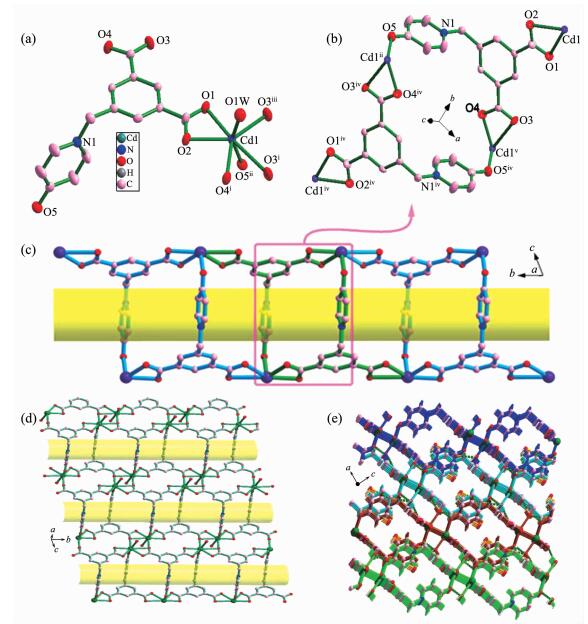 图2
(a) Metal coordination and atom labeling in compound 2; (b) Dinuclear metallamacrocycle unit;
(c) Side view of the tubular structure; (d) Tubular channels perpendicular to the ab plane of 2;
(e) Three dimensional supramolecular structure of 2
Figure2.
(a) Metal coordination and atom labeling in compound 2; (b) Dinuclear metallamacrocycle unit;
(c) Side view of the tubular structure; (d) Tubular channels perpendicular to the ab plane of 2;
(e) Three dimensional supramolecular structure of 2
图2
(a) Metal coordination and atom labeling in compound 2; (b) Dinuclear metallamacrocycle unit;
(c) Side view of the tubular structure; (d) Tubular channels perpendicular to the ab plane of 2;
(e) Three dimensional supramolecular structure of 2
Figure2.
(a) Metal coordination and atom labeling in compound 2; (b) Dinuclear metallamacrocycle unit;
(c) Side view of the tubular structure; (d) Tubular channels perpendicular to the ab plane of 2;
(e) Three dimensional supramolecular structure of 2
2.3 Thermal analyses and PXRD analyses
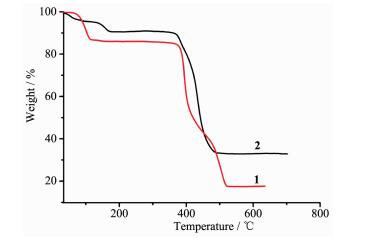
Thermal gravimetric analysis (TGA) was used to characterize the thermal stability of compounds, and the TGA of compounds 1~2 were carried out in nitrogen atmosphere (Fig. 3). For compound 1, the weight loss of 14.99% from 30 to 117 ℃ is assigned to the loss of two coordinated and 2.25 lattice water molecules (Calcd. 15.15%). There is no further weight loss from 117 to 362 ℃, After 362 ℃, the organic components start to decompose. In the TG curve of compound 2, the weight loss in the range of 30~209 ℃ (Obsd. 11.53%, Calcd. 12.35%) can be attributed to the removal of two lattice and one coordinated water molecules. The further weight losses represented the decomposition of the compound 2.
The synthesized products of 1~2 have been characterized by powder X-ray diffraction (PXRD). As shown in Fig. 4, the experimental PXRD patterns correspond well with the results simulated from the single crystal data, indicating the high purity of the synthesized samples and single phases of compounds 1~2.
2.4 Photochemical properties
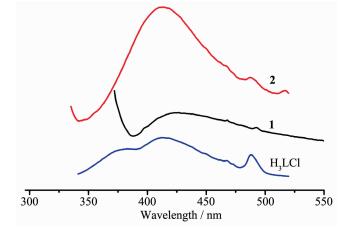
The solid-state photo-luminescent properties of the free H3LCl ligand, coordination polymers 1 and 2 have been investigated in the solid state at room temperature. The emission spectra of these compounds are shown in Fig. 5. An intense band is observed at 412 nm (λex=317 nm) for H2L. Compared to the H3LCl ligand, compound 1 results in red shift of 13 nm (425 nm, λex=364 nm). The shift of the emission maximum between 1 and H3LCl is considered to mainly originate from the influence of the coordination of the ligand to metal atom[23-24]. The compound 2 has same emission peak of 412 nm (λex=315 nm) with the free H2L, which indicates that it may originate from metal-perturbed intraligand charge transfers[25-27].
3 Conclusions
We have reported the syntheses, crystal structures and properties of two coordination polymers, {[Zn(L)(bpa)0.5(H2O)2]·2.25H2O}n (1) and {[Cd(L)(H2O)]·2H2O}n (2) with different structure derived from 5-(4-hydroxy-pyridinium-1-ylmethyl) isophthalic acid chloride ligand. Both compound 1 and 2 have a 3D supramole-cular architecture extended by hydrogen-bonding interactions, but the 3D network of compound 1 is constructed by an infinite 1D ladder-like chain, and that of compound 2 is formed by a 2D layer structure with tubular channels. In addition, the coordination modes of carboxylate groups in two compounds are different: μ2-η1:η1 mode for compound 1, μ3-η1:η2 mode for 2. Compared to the compounds based on the present ligand and reported in the literature[17], they have not only different structure, but also different coordination modes, which indicate that H3LCl ligand is a favorable and fashionable building block for compounds with abundant structural features and useful properties. Subsequent studies will focuse on the structures and properties of the novel functional coordination polymers constructed by the present ligand with a wide range of rare earth metals. Further investigations on this domain are underway.
-
-
[1]
Cao L H, Zang S Q, Li J B. Z. Anorg. Allg. Chem., 2011, 637:1427-1431 doi: 10.1002/zaac.v637.10
-
[2]
Zhu H F, Fan J, Okamura T A, et al. Inorg. Chem., 2006, 45(10):3941-3948 doi: 10.1021/ic051925o
-
[3]
Zhang L P, Ma J F, Yang J, et al. Cryst. Growth Des., 2009, 9(11):4660-4673 doi: 10.1021/cg900460k
-
[4]
乔宇, 尉兵, 王璐瑶, 等.无机化学学报, 2016, 32(7):1261-1266 doi: 10.11862/CJIC.2016.164QIAO Yu, WEI Bing, WANG Lu-Yao, et al. Chinese J. Inorg. Chem., 2016, 32(7):1261-1266 doi: 10.11862/CJIC.2016.164
-
[5]
Dong X Y, Zhang M, Pei R B, et al. Angew. Chem. Int. Ed., 2016, 55:2073-2077 doi: 10.1002/anie.201509744
-
[6]
Pan C, Nan J P, Dong X L, et al. J. Am. Chem. Soc., 2011, 133(32):12330-12333 doi: 10.1021/ja2031568
-
[7]
Han L, Valle H, Bu X H. Inorg. Chem., 2007, 46:1511-1513 doi: 10.1021/ic062015p
-
[8]
Qi Y, Luo F, Batten S R, et al. Cryst. Growth Des., 2008, 8:2806-2813 doi: 10.1021/cg701061q
-
[9]
Hu Y W, Li G H, Liu X M, et al. CrystEngComm, 2008, 10:888-893 doi: 10.1039/b800255j
-
[10]
Li S L, Lan Y Q, Qin J S, et al. Cryst. Growth Des., 2009, 9:4142-4146 doi: 10.1021/cg900371k
-
[11]
Li H Y, Cao L H, Wei Y L, et al. CrystEngComm, 2015, 17:6297-6307 doi: 10.1039/C5CE01008J
-
[12]
Kuai H W, Fan J, Liu Q, et al. CrystEngComm, 2012, 14:3708-3716 doi: 10.1039/c2ce25062d
-
[13]
Kuai H W, Hou C, Sun W Y. Polyhedron, 2013, 52:1268-1275 doi: 10.1016/j.poly.2012.06.020
-
[14]
Ji C, Li B, Ma M L, et al. CrystEngComm, 2012, 14:3951-3958 doi: 10.1039/c2ce06720j
-
[15]
Li J B, Dong X Y, Cao L H, et al. CrystEngComm, 2012, 14:4444-4453 doi: 10.1039/c2ce06705f
-
[16]
Zang S Q, Cao L H, Liang R, et al. Cryst. Growth Des., 2012, 12:1830-1837 doi: 10.1021/cg2013733
-
[17]
李付安, 徐伏, 杨维春, 等.无机化学学报, 2016, 32(9):1683-1691 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?flag=1&file_no=20160925&journal_id=wjhxxbcnLI Fu-An, XU Fu, YANG Wei-Chun, et al. Chinese J. Inorg. Chem., 2016, 32(9):1683-1691 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?flag=1&file_no=20160925&journal_id=wjhxxbcn
-
[18]
SMART and SAINT, Area Detector Control and Integration Software, Siemens Analytical X-Ray Systems, Inc. , Madison, WI, 1996.
-
[19]
Sheldrick G M. SADABS 2. 05, University of Göttingen, Germany, 1997.
-
[20]
Sheldrick G M. SHELXS-97, Program for the Solution of Crystal Structures, University of Göttingen, 1997.
-
[21]
Sheldrick G M. Acta Crystallogr. Sect. A, 2008, A64:112
-
[22]
Bernstein J, Davis R E, Shimoni L, et al. Angew. Chem. Int. Ed., 1995, 34:1555-1573 doi: 10.1002/(ISSN)1521-3773
-
[23]
Chang Z, Zhang A S, Hu T L, et al. Cryst. Growth Des., 2009, 9:4840-4846 doi: 10.1021/cg900659r
-
[24]
Guo J, Ma J F, Liu B, et al. Cryst. Growth Des., 2011, 11:3609-3621 doi: 10.1021/cg200581w
-
[25]
Zheng S L, Yang J H, Yu X L, et al. Inorg. Chem., 2004, 43:830-838 doi: 10.1021/ic034847i
-
[26]
Fang S M, Zhang Q, Hu M, et al. Cryst. Growth Des., 2010, 10:4773-4785 doi: 10.1021/cg100645p
-
[27]
Su Z, Fan J, Chen M, et al. Cryst. Growth Des., 2011, 11:1159-1169 doi: 10.1021/cg101365v
-
[1]
-
Figure 1 (a) Metal coordination and atom labeling in compound 1 with thermal ellipsoids at 50% probability level; (b) 1D ladder-like chain; (c) View of 3D supramolecular framework of 1 via hydrogen bonds (the dashed lines represent the hydrogen bonds); (d) Hydrogen bonds in compound 1
Hydrogen atoms and the solvent water molecule are omitted for clarity in (a); Symmetry codes: ⅰ 1+x, y, z; ⅱ 3-x, -y, -z; ⅲ 1-x, 1-y, 1-z; ⅳ 1-x, -y, 1-z; ⅴ -x, -y, 1-z; ⅵ -1+x, -1+y, z; ⅶ x, -1+y, -1+z; ⅷ -1+x, y, z; ⅸ -x, 1-y, 2-z; ⅹ 2-x, -y, 1-z; xi 1+ x, y, -1+z; ⅹⅱ -x, 1-y, 1-z; ⅹⅲ x, -1+y, z
Figure 2 (a) Metal coordination and atom labeling in compound 2; (b) Dinuclear metallamacrocycle unit; (c) Side view of the tubular structure; (d) Tubular channels perpendicular to the ab plane of 2; (e) Three dimensional supramolecular structure of 2
Thermal ellipsoids at 50% probability level in (a) and (b); Hydrogen atoms and the solvent water molecule are omitted for clarity; Symmetry codes: ⅰ x, 1+y, z; ⅱ 2-x, 1-y, 1-z; ⅲ 3-x, 1-y, -z; ⅳ 2-x, -y, 1-z; ⅴ x, -1+y, z; ⅵ 1+x, 1+y, -1+z; ⅶ 2-x, 2-y, -z; ⅷ 1-x, -y, 1-z; ⅸ 2-x, 1-y, -z
Table 1. Crystal data and structure refinement for compounds 1 and 2
Compound 1 2 Formula C20H23.5N2O9.25Zn C14HI5NO8Cd Formula weight 505.28 437.67 Crystal system Triclinic Triclinic Space group P1 P1 a/nm 0.969 30(8) 0.738 70(4) b/nm 1.066 54(9) 1.015 35(4) c/nm 1.227 35(11) 1.120 03(7) α/(°) 67.315(8) 70.191(4) β/(°) 78.218(7) 81.994(5) γ/(°) 67.830(8) 76.292(4) V/nm3 1.081 57(18) 0.766 25(7) Z 1 2 Dc/ (g·cm-3) 1.552 1.897 F(000) 523 436 Reflection collected 6 391 5 773 Independent reflection 3 811 2 687 Rint 0.058 9 0.024 9 GOF on F2 1.057 1.028 R1a[I>2σ(I)] 0.044 5 0.028 4 wR2b[I>2σ(I)] 0.110 7 0.068 9 $^{\text{a}}{R_1} = {\sum {\left| {\left| {{F_{\text{o}}}} \right|- \left| {{F_{\text{c}}}} \right|/\sum {\left| {{F_{\text{o}}}} \right|;} } \right.} ^{\text{b}}}w{R_2} = {\left[{\sum {w{{\left( {F_{\text{o}}^2-F_{\text{c}}^2} \right)}^2}} /\sum {w{{\left( {F_{\text{o}}^2} \right)}^2}} } \right]^{1/2}} $ Table 2. Selected bond lengths (nm) and bond angles (°) for compounds 1 and 2
Compound 1 Zn-O(1) 0.195 2(2) Zn-O(3)ⅰ 0.196 9(2) Zn-N(2) 0.205 5(2) Zn(1)-O(1W) 0.214 2(2) Zn(1)-O(2W) 0.229 1(2) O(1)-Zn(1)-N(2) 133.65(11) O(1)-Zn-O(3)ⅰ 125.29(10) O(1)-Zn(1)-O(1W) 88.55(10) O(3)ⅰ-Zn-N(2) 100.60(10) N(2)-Zn(1)-O(1W) 93.77(10) O(3)ⅰ-Zn(1)-О(1W) 95.37(11) O(3)ⅰ-Zn(1)-O(2W) 89.56(10) O(1)-Zn(1)-O(2W) 85.77(10) O(1W)-Zn(1)-O(2W) 173.98(9) N(2)-Zn(1)-0(2W) 88.72(10) Compound 2 Cd(1)-O(1) 0.232 3(2) Cd(1)-O(2) 0.239 8(2) Cd(1)-O(3)ⅰ 0.274 7(1) Cd(1)-O(4)ⅰ 0.228 2(3) Cd(1)-O(5)ⅱ 0.227 6(3) Cd(1)-O(1W) 0.234 2(3) Cd(1)-O(3)ⅲ 0.227 4(3) O(3)ⅰ-Cd(1)-O(4)ⅰ 50.396(2) O(5)ⅱ-Сd(1)-O(3)ⅰ 81.625(2) O(1W)-Cd(1)-O(3)ⅰ 79.926(2) O(1)-Cd(1)-O(3)ⅰ 173.275(3) O(5)ⅱ-Сd(1)-O(3)ⅲ 83.16(10) O(2)-Сd(1)-O(3)ⅰ 131.347(3) O(5)ⅱ-Cd(1)-O(4)ⅰ 93.32(10) 0(3)ⅲ-Сd(1)-O(4)ⅰ 131.81(9) O(1)-Cd(1)-O(3)ⅲ 91.73(10) Symmetry codes: ⅰ x+1, y, z for 1; ⅰ x, 1+y, z; ⅱ 2-x, 1-y, 1-z; ⅲ 3-x, 1-y, -z for 2. Table 3. Geometrical parameters of hydrogen bonds in compounds 1~2
D-H...A d(D-H)/nm d(H...A)/nm d(D...A)/nm ∠DHA/(°) Compound 1 O(1W)-Н(1WА)...O(5)ⅹⅰ 0.085 6 0.183 1 0.267 7 169.72 O(1W)-H(1WB)...O(4)ⅲ 0.086 6 0.213 7 0.289 0 144.98 O(2W)-H(2WA)...O(2)ⅹ 0.085 1 0.188 0 0.272 9 175.72 O(2W) -H(2WB)...O(3W)ⅰ 0.072 1 0.215 6 0.282 5 154.77 O(3W)-H(3WA)...O(4w) 0.085 8 0.194 8 0.280 6 178.94 O(3W)-H(3WB)...O (1)ⅷ 0.085 0 0.233 4 0.292 3 126.69 O(4W)-H(4WA)...O(4) 0.085 0 0.199 1 0.280 5 160.05 O(4W)-H(4WB)...O (5)ⅸ 0.072 0 0.207 5 0.275 8 158.52 Compound 2 O(1W)-Н(1WА)...O(1)ⅶ 0.083 2 0.227 6 0.298 9 143.85 O(1W)-Н(1WВ)...O(5)ⅵ 0.089 7 0.203 8 0.284 8 149.66 O(2W)-H(2WA)...N(1) 0.082 2 0.242 3 0.319 0 155.83 O(2W)-H(2WC)...O (1W)ⅸ 0.085 0 0.212 9 0.298 0 179.94 O(3W)-H(2WA)...O (2W) 0.085 0 0.234 0 0.318 8 175.81 O(3 W)-H(2WC)...O (2W)ⅷ 0.080 1 0.213 9 0.281 3 141.94 Symmetry codes: ⅰ 1+x, y, z; ⅲ 1-x, 1-y, 1-z; ⅷ -1+x, y, z; ⅸ -x, 1-y, 2-z; ⅹ 2-x, -y, 1-z; ⅹⅰ 1+x, y, -1+z for 1; ⅵ 1+x, 1+y, -1+z; ⅶ 2-x, 2-y, -z; ⅷ 1-x, -y, 1-z; ⅸ 2-x, 1-y, -z for 2. -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 1
- 文章访问数: 994
- HTML全文浏览量: 70



 下载:
下载:





 下载:
下载:

