 图Scheme1
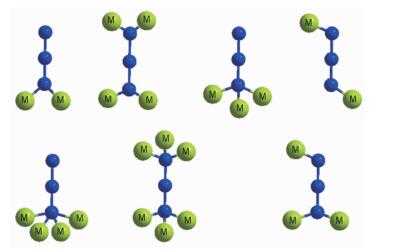
Coordination modes of bridging azido
Scheme1.
Coordination modes of bridging azido
图Scheme1
Coordination modes of bridging azido
Scheme1.
Coordination modes of bridging azido

呈现强链内铁磁耦合的EO-叠氮与羧基双重桥连一维铜链化合物:合成、晶体结构、磁性及DFT计算
English
One-Dimensional Cu(Ⅱ)Chain Compound with Simultaneous EO-Azido and Carboxylato Bridges Displaying Strong Ferromagnetic Coupling:Synthesis, Crystal Structure, Magnetic Properties with DFT Calculations
-
Key words:
- azido-copper
- / benzoate
- / crystal structure
- / magnetic property
- / DFT calculation
-
0 Introduction
The molecular magnetism has been a hot research topic, in which great progress has been achieved[1-2]. Research on the structural design and synthesis of molecule-based magnetic materials, inclu-ding single-molecule magnets (SMMs)[3] and single-chain magnets (SCMs)[4], has made tremendous strides as a significant intersection between chemical synth-esis and materials science and continues to be a productive area due to their intriguing structures, unique physical characteristics, and promising novel applications, such as magnetic sensors, magnetic swit-ches and multifunctional magnetic devices[5-6]. Since the first example of SCMs in 2001[4a], one-dimensional (1D) molecular assemblies have received considerable attention for the construction of new molecule-based magnets. Such materials are usually assembled by combining paramagnetic centers with suitable organic ligands that regulate the architectures and transmit magnetic exchange coupling[7].
A popular approach for constructing these types of materials is to employ short ligands capable of efficiently transmitting the magnetic coupling[8]. In this sense, the azido ligand with three donor atoms is able to link metal ions in different coordination modes (Scheme 1), which induces rich structural diversity as well as a range of different magnetic properties in the azido-metal complexes[9-14]. Especially, the azido-based Cu(Ⅱ) coordination polymers are among the most important kinds of azido-metal complexes owing to the superiority for understanding the fundamental nature of magnetic interactions and magneto-structural relationships on the molecular level[15]. It is well-established that μ-1, 3 (end-to-end, EE) modes usually propagate antiferromagnetic while μ-1, 1 (end-on, EO) modes are usually ferromagnetic in the azido-Cu(Ⅱ) cases, although the coupling between metal ions bridged by EO-azido ligands can be antiferromagnetic in the presence of other bridging ligands or for very large metal-N-metal angle[16]. It has been suggested that the strongest ferromagnetic coupling in the EO-azido linker occurs at a Cu-N-Cu bond angle close to 108°, and so an antiferromagnetic interaction would probably be found for larger Cu-N-Cu bond angles[17]. Consequently, an effective strategy for tuning the structures of azido-Cu(Ⅱ) compounds with notable magnetic properties is to introduce coligands into the systems. The most common coligands are carboxylate-containing organics in which the carboxyl links the Cu(Ⅱ) ions to generate various systems and adopts different bridging modes to transmit diverse superex-change interactions[18]. A series of azido/carboxylate/ Cu(Ⅱ) compounds involving the combinations of μ-1, 1-azido ligands and syn, syn-carboxylates have been previously prepared and performed intriguing struc-tures and magnetisms[19]. Most importantly, molecular orbital calculations dramatically support the counter complementary effect enforced by the carboxylate ligand, which weakens the effect of the antiferroma-gnetic azido ligand to the point where dominant ferromagnetic behavior is obtained[20]. It still remains significant challenge to design and configure the desired system for clarifying the complicated and crucial issues of azido-Cu(Ⅱ) compounds, such as the key factors influencing the magnetic interactions, the regulation of magneto-structural correlation, and the mechanism of magnetization.
Based on the considerations above, we report the synthesis, structural characterization, and magnetic properties of a 1D azido-Cu(Ⅱ) coordination polymer, [Cu(4-Fb)(N3)(H2O)]n (1) (4-Fb=4-formylbenzoate), in which the intrachain Cu(Ⅱ) ions are connected by a mixed-bridge of syn, syn-carboxylate and μ-1, 1-azido ligand with Cu-N-Cu angle of 113.34°. Magnetic investigations suggest that compound 1 shows strong ferromagnetic coupling between neighboring Cu(Ⅱ)ions, which is further explored by density functional theory (DFT) calculations as well.
1 Experimental
1.1 Physical measurements
Elemental analysis (C, H, N) was performed on a Perkin-Elmer 2400 CHN elemental analyzer. The FT-IR spectra were recorded in the range of 400~4 000 cm-1 using KBr pellets on an EQUINOX55 FT/IR spectrophotometer. The phase purity of the bulk or polycrystalline samples were verified by powder X-ray diffraction (PXRD) measurements performed on a Rigaku RU200 diffractometer at 60 kV, 300 mA and Cu Kα radiation (λ=0.071 073 nm), with a scan speed of 5°·min-1, a step size of 0.02° and a scan range of 5°~50°(2θ). Temperature-dependent magnetic measure-ment was obtained on poly-crystalline sample using a Quantum Design MPMS-XL7 SQUID magnetometer at temperatures range 1.9~300 K with an applied field of 1 000 Oe (restrained in eicosane to prevent torqueing at high fields). Magnetization measurements were taken at 2.0 K from 0 to 50 kOe. All data were corrected for diamagnetism estimated from Pascal′s constants, and an experimental correction for the sample holder was applied.
1.2 Materials and methods
All of the solvents and reagents for synthesis are of analytical grade and are commercially available. Cu(NO3)2·3H2O, 4-formylbenzoic acid (4-Fba) and NaN3 were purchased from commercial sources and used without further purification.
Caution! Although we have not experienced any problems in our experiments, azido and its compounds are potentially explosive; only a small amount of material should be prepared and handled with care.
1.3 Preparation of [Cu(4-Fb)(N3)(H2O)]n (1)
Compound 1 was hydrothermally synthesized under autogenous pressure. A mixture of Cu(NO3)2·3H2O (0.051 g, 0.3 mmol), 4-Fba (0.045 g, 0.3 mmol), NaN3 (0.033 g, 0.5 mmol) and H2O (8 mL) was sealed in a 15 mL Teflon-lined autoclave and heated to 120 ℃. After being maintained for 3 days, the reaction vessel was cooled to 20 ℃ in 12 h. Green crystals were collected (Yield: 80%, based on Cu). Anal. Calcd. for CuC8H7N3O4(%): C, 35.23; H, 2.58; N, 15.41. Found(%): C, 35.21; H, 2.57; N, 15.39. IR (KBr, cm-1): 3 414 (m), 2 094 (m), 1 662 (m), 1 608(s), 1 375(s), 1 317(w), 1 225(s), 1 179(s), 1 063(s), 1 037(s), 867(w), 774(w), 688(m), 584(w).
1.4 Crystallographic data collection and refinement
Suitable single crystal of the compound was mounted on glass fibers for X-ray measurements. Reflection data were collected at room temperature on a Bruker SMART APEX-CCD-based diffractometer using graphite mono-chromated Mo Kα radiation (λ=0.071 073 nm). An empirical absorption correction was applied using the SADABS program[21]. Data proc-essing was accomplished with the SAINT processing program. The structures were solved by the direct methods and refined with full-matrix least-squares on F2 using SHELXTL 97 program[22]. All non-hydrogen atoms were refined with anisotropic displacement parameters. Hydrogen atoms were placed in geome-trically calculated positions. Selected crystallographic data and structural refinement details for 1 are summarized in Table 1. Selected bond lengths and bond angles, and the hydrogen bonds of compound 1 are listed in Table S1 and S2, respectively.
Empirical formula C8H7CuN3O4 Formula weight 272.71 Crystal system Monoclinic Space group P21/c a / nm 0.758 02(8) b / nm 0.661 77(7) c / nm 2.008 2(2) β/(°) 97.707(2) V / nm3 0.998 30(18) Z 4 D / (Mg·m-3) 1.814 μ/ mm-1 2.19 F(000) 548 Unique reflections 2 080 Observed reflections 5 246 Rint 0.027 5 Data, restraint, parameter 2 080, 1, 150 Final R indices [I>2σ(I)] R1=0.024 9, wR2=0.066 8 R indices (all data) R1=0.028 9, wR2=0.068 4 Goodness-of-fit on F2 1.008 CCDC: 1496426.
1.5 Computational methodology
The following computational methodology was used to calculate the coupling constant in the title compound[23]. The spin Hamiltonian suggested originally by Heisenberg can be written as
$\hat H =-\sum {_{\left( {i > j} \right)}J{S_i}{S_j}} $ (where Si and Sj are the spin operators of the paramagnetic centers, Si=Sj=1/2 for Cu(Ⅱ) ion; and the J constant is the coupling constant between the paramagnetic spin carriers), which can be employed to express the exchange coupling between two transition metal ions, the full Hamiltonian matrix for the entire system can be established. The J value was calculated from the energy difference of the two spin states: the broken symmetry (BS) state and the triplet state (HS), the broken symmetry approach along with electron correlations has been widely used to investigate magnetic properties in a large number of magnetic systems[24]. The J value was calculated using the following equation:where EBS is the energy of the broken symmetry singlet state and EHS is the energy of the triplet state.
The DFT calculations are implemented with the ORCA 3.0.2 package[25]. The BP86 functional proposed by Becke[26] and Perdew[27a] and hybrid B3LYP func-tional built by Becke[27b] were applied in the calcula-tions, respectively. The double-ξ quality plus polariza-tion def2-SVP basis set and polarized triple-quality basis sets of def2-TZVP, TZVP, and TZV proposed by Ahlrichs and co-workers were respectively performed for all atoms[28]. The calculation model for the comp-ound was built from the experimental results.
2 Results and discussion
2.1 Crystal structure of 1
Single-crystal X-ray diffraction analysis reveals that compound 1 crystallizes in the monoclinic space group P21/c. The asymmetrical unit of compound 1 is composed by one Cu(Ⅱ) cation, one azido ligand, one 4-Fb ligand and one coordinated water molecule. The penta-coordinated Cu(Ⅱ) cation in the center presents a distorted tetragonal pyramid geometry (Fig. 1b). The bottom square is formed by two nitrogen atoms (Cu1-N1 0.199 38 nm, Cu1-N1i 0.199 46 nm) from two azido ligands and two oxygen atoms (Cu1-O1 0.194 08 nm, Cu1-O2i 0.195 66 nm) from two carboxylate groups of 4-Fb ligands. The apical position is occupied by one oxygen atom (Cu1-O3 0.233 92 nm) from coordinated water molecule (Fig. 1a). Adjacent Cu(Ⅱ) cations are mediated by EO-azido, μ2-bridging bidentate carboxylate groups, with a Cu-N-Cu angle of 113.34° and a Cu-Cu distance of 0.333 2 nm to yield a well-isolated 1D copper chain (Fig. 1c). And then, the linear metal chains are integrated by interchain hydrogen-bonding between the O atom in the coor-dinated water and the terminal N atom in the azido anion (O4…N3 0.268 nm) (Fig. 1d), constructing the supramolecular network of 1. In addition, the azido moieties are quasi linear with N1-N2-N3 angles of 178.8°, and the bond length of N1-N2 (0.121 8 nm) is slightly longer than N2-N3 (0.114 2 nm). The nearest distance of interchain Cu(Ⅱ) ions is 0.758 0 nm.
 图1
(a) Structure of 1 with 50% thermal ellipsoids; (b) Simplified tetragonal pyramidal geometry of the center Cu(Ⅱ) cation for 1; (c) 1D chain with carboxylate and azido bridges for 1; (d) Hydrogen bonding formed by azido and water molecules between adjacent chains in 1
Figure1.
(a) Structure of 1 with 50% thermal ellipsoids; (b) Simplified tetragonal pyramidal geometry of the center Cu(Ⅱ) cation for 1; (c) 1D chain with carboxylate and azido bridges for 1; (d) Hydrogen bonding formed by azido and water molecules between adjacent chains in 1
图1
(a) Structure of 1 with 50% thermal ellipsoids; (b) Simplified tetragonal pyramidal geometry of the center Cu(Ⅱ) cation for 1; (c) 1D chain with carboxylate and azido bridges for 1; (d) Hydrogen bonding formed by azido and water molecules between adjacent chains in 1
Figure1.
(a) Structure of 1 with 50% thermal ellipsoids; (b) Simplified tetragonal pyramidal geometry of the center Cu(Ⅱ) cation for 1; (c) 1D chain with carboxylate and azido bridges for 1; (d) Hydrogen bonding formed by azido and water molecules between adjacent chains in 1
2.2 Magnetic studies
The crystalline sample of 1 was all phase-pure, as confirmed by PXRD (Fig.S1). According to the obtained data, a dominant ferromagnetic coupling between the Cu(Ⅱ) cations in compound 1 can be suggested.
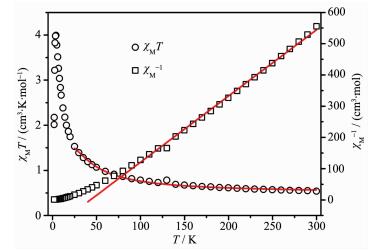
The magnetic properties of 1 are shown in Fig. 2 in the form of a χMT versus T plot (χM is the molar magnetic susceptibility per Cu(Ⅱ) cation). χMT values are observed as 0.54 cm3·K·mol-1 for 1 at 300 K, larger than the spin-only value (0.375 cm3·K·mol-1) for an isolated Cu(Ⅱ) cation (S=1/2). Upon cooling, the χMT values increase gradually, and firstly the value increases to 3.99 cm3·K·mol-1 at 4 K, suggesting ferromagnetic exchange between Cu(Ⅱ) cations and finally drops to 2.01 cm3·K·mol-1 at 1.9 K. The χMT vs T curve illustrates that strongly coupled ferroma-gnetic system accompanies with antiferromagnetic interaction between the azido-Cu(Ⅱ) chains in the compound 1, especially at low temperature. The parameters fitted by the Curie-Weiss law above are obtained to be C=0.472 cm3·K·mol-1 and θ=43.04 K. The positive θ value supports strong ferromagnetic coupling between the intrachain Cu(Ⅱ) ions. Consid-ering the mean-field approximation for interchain coupling zJ′ (Eq.2), the temperature-dependent magn-etic susceptibility data of 1 can be simulated with the formula proposed by Baker et al. (Eq.3)[29] for a ferro-magnetic Cu(Ⅱ) chain (S=1/2) which is achieved from the high temperature series expansion.
A=1.0+5.798 0x+16.902 7x2+29.376 9x3+29.832 9x4+14.036 9x5
B=1.0+2.798 0x+7.008 7x2+8.653 8x3+4.574 3x4
x=J/(2kT)
The best fit of the magnetic susceptibility data resulted in: g=2.27, J=72.1 cm-1, zJ′=-0.71 cm-1 and R=5.75×10-5 (g is the Zeeman factor of the metal ion, J describes the intrachain magnetic interaction, zJ′ describes the interchain antiferromagnetic interaction and R accounts for the agreement factor defined as R=∑[(χMT)obsd-(χMT)calcd]2/∑[(χMT)obsd]2). The large J value supports strong ferromagnetic coupling between the Cu(Ⅱ) centers. The small negative zJ′ value indicates the presence of interchain antiferromagnetic interac-tions, according with the drop in the χMT product at low temperature. In principle, it seems that intrachain antiferromagnetic coupling would be predicted for 1 due to the cooperation of EO-azido with the Cu-N-Cu angle of 113.34° (larger than the critical value of 108°) and the carboxylate group with syn-syn mode. However, according to the proposition from Thompson et al. and Escuer et al., the counter-complementarity function derived from two kinds of ligands may expound the strong ferromagnetic interaction, not just the total of the two isolated components[20].
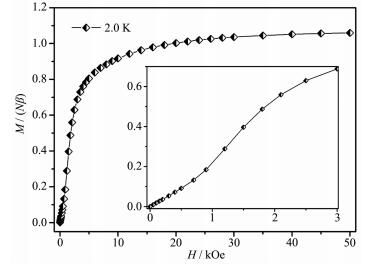
As shown in Fig. 3, the isothermal field-dependent magnetization M(H) values at 2 K and fields up to 50 kOe are measured for 1. The magnetization curve increases linearly under very low field, subsequently climbs up quickly until 10 kOe and rises up gradually to 50 kOe with an effective moment of 1.06Nβ at the high fields, slightly higher than the saturation value (1.0Nβ) of one Cu(Ⅱ) cation. Notably, the S-shaped curve emerges with a critical field of 1 200 Oe at low field (Fig. 3, inset), signifying weakly interchain antiferromagnetic exchange and 1 might sustain decoupling effect of external field.
The dc magnetization is determined at 2, 2.5, and 4 K within -7~7 kOe, which is shown in Fig.S2. When the temperature goes down to 2 K, no hysteresis loop emerges.The field-cooled (FC) and zero-field-cooled (ZFC) magnetization measurements were performed at a low applied field of 10 Oe below the temperature of 20 K (Fig.S3), the FC/ZFC plots with coincident pattern increase rapidly until the temperature drops to 2 K, indicating the absence of the ferromagnetic ordering. In addition, under the oscillating field of 3.5 Oe, the zero-field AC susceptibility experiments for 1 were determined in the range of 1.9~25 K at various frequencies of 1, 10, 33, 100, 333 and 1 000 Hz (Fig.S4). In-phase signal and no out-of-phase signal were observed until the temperature drops to 2 K, which further verifies intrachain antiferromagnetic coupling and implies that the slow dynamics of magnetization and long-rang ferromagnetic ordering are nonexistent in compound 1. The in-phase (χ′) component of the AC magnetic sus-ceptibility with a peak value at 4 K might explicate that compound 1 behaves as an antiferromagnet with extremely low Néel temperature.
2.3 Theoretical studies
In order to further clarify the ferromagnetic nature of the exchange interaction in compound 1, we performed a theoretical study of the isotropic coupling constants J between Cu(Ⅱ) ions based on DFT calculation at BP86 and B3LYP level with the aid of ORCA program. According to the structure of 1, supposing that the dominant magnetic exchange is mediated between adjacent two Cu(Ⅱ) ions through azido and carboxylate. The calculations for the compound were carried out with the model (for comparing) applied to the magnetic fitting by filling-in all the coordination sites of the Cu(Ⅱ) ions (Fig. 4).
The results of the theoretical calculation and the experimental fitting in terms of the coupling constants are listed in Table 2. Based on BP86 and B3LYP functions with def2-SVP, def2-TZVP, TZVP, and TZV basis sets, the calculated J values are unexceptionally found to be moderate positive values and close to the experimentally fitted values, which completely verifies that the strong ferromagnetic coupling is prevailing in compound 1. Obviously, the choices of the methods and basis sets for these calculations are simultan-eously suitable for the title compound. Although the calculated values deviate slightly from the fitting values, in all the cases the sign and the relative magnitudes of the exchange parameters agree very well with the experimental results. It is difficult to say in general which basis set works better, but it is clear that the results of theoretical calculation prove qualitatively and quantificationally the measuring data. Therefore, the tiny difference may result from the fact that the real compound is not scattered entities as has been modeled but is very complicated in the whole structures.
1 EHS/eV EBS/eV JDFT / cm-1 JExpt / cm-1 BP86 def2-tzvp -150 464.859 80 -150 464.849 39 83.54 72.1 def2-svp -150 381.266 57 -150 381.256 40 81.51 tzv -150 442.523 75 -150 442.513 24 84.23 tzvp -150 461.971 63 -150 461.960 85 86.60 B3LYP def2-tzvp -150 413.412 20 -150 413.401 33 87.63 def2-svp -150 329.346 82 -150 329.335 65 89.97 tzv -150 389.311 06 -150 389.299 97 89.39 tzvp -150 410.351 84 -150 410.340 80 88.96 2.4 Discussion
In compound 1, two consecutive Cu(Ⅱ) ions are bonded by syn-syn carboxylate and symmetric EO-azido bridges, constituting the 1D chain-like pattern. Intrachain Cu-Cu distances and Cu-N-Cu angles are 0.333 2 nm and 113.34°. The alternating 1D chains are interacted through interchain hydrogen-bonds derived from the water oxygen atom and the nitrogen atom of the azido group between nearby chains. The nearest interchain Cu…Cu separation is 0.758 0 nm. EO-azido is certainly one of the most interesting magnetic couplers in molecular magnetism, and the magnitude of the J parameter depends on several factors, but mainly the Cu-N-Cu angle (β). According to a number of studies on Cu(Ⅱ) systems with such bridges[30, 17a], the single EO-azido motif could also mediate ferromagnetic coupling, EO-azido bridging Cu(Ⅱ) ions with low β gives rise to ferromagnetic coupling, whereas the coupling is antiferromagnetic if the angle is above a critical β value which has been evaluated to be about 108°[19a]. In this sense, antiferromagnetic coupling would be expected for 1, owing to the presence of EO-azido bridges with a large Cu-N-Cu angle of 113.34°, together with the syn-syn carboxylate bridges featured in the chain. However, the fitting magnetic coupling parameter (J) confirms that the ferromagnetic interactions are enabled by the single EO-azido bridges in compound 1, probably due to the counter-complementarity effect from the syn-syn carboxylate bridge which usually transmit antiferromagnetic interactions between neighbouring metal ions.
To deduce a general magneto-structural relation-ship, we have made a comprehensive comparison of the Cu(Ⅱ)-azido-benzoate compounds reported in recent years, as shown in Table S3[31-32, 19a, 33-34]. EO-azido comp-ounds with Cu-N-Cu angles of 126.8°[31a], 108.2°[31a], 116.8°[31a], 109.4°[31c], 101.1°[31c], 111.9°[31b], and 105.5°[33d] also exhibit strong ferromagnetic coupling. Similarly, in our work, the Cu-N-Cu angle is 113.34°, and the corresponding compound shows ferromagnetic interac-tion. The coupling constant value (J=72.1 cm-1) for compound 1 is comparable to previous results in the literature[31b, 32a].
3 Conclusions
In present work, a new azido-copper compound with 4-formylbenzoic acid as coligand has been successfully isolated. Structural analyses indicate compound 1 features a 1D two-fold bridged copper chain in which the coordination geometry of center Cu(Ⅱ) ion is distorted tetragonal pyramid and the adjacent two Cu(Ⅱ) ions is bridged by mixed μ-1, 1(EO)-azido and syn-syn-carboxylate ligands. Magnetic investigations demonstrate that the compound is composed of ferromagnetically coupled ferromagnetic chains. The intrachain behavior reflects how the counter complementary effect imposed by the carboxylate bridge overcomes the antiferromagnetic effect of the azido bridge resulting in an overall ferromagnetic interaction. DFT calculations qualita-tively and quantificationally support the strong ferro-magnetic coupling between the Cu(Ⅱ) ions.
Supporting information is available at http://www.wjhxxb.cn
-
-
[1]
(a)Pei Y, Verdaguer M, Kahn O. J. Am. Chem. Soc. , 1986, 108: 7428-7430
(b)Miller J S, Calabrese J C, Rommelmann H, et al. J. Am. Chem. Soc. , 1987, 109: 769-781 -
[2]
(a)Wang X Y, Wang Z M, Gao S. Inorg. Chem. , 2008, 47: 5720-5726
(b)Wang X Y, Wang L, Wang Z M, et al. J. Am. Chem. Soc. , 2006, 128: 674-675
(c)Martín S, Barandika M G, Lezama L, et al. Inorg. Chem. , 2001, 40: 4109-4115 -
[3]
(a)Ungur L, Lin S Y, Tang J K, et al. Chem. Soc. Rev. , 2014, 43: 6894-6905
(b)Gatteschi D, Sessoli R. Angew. Chem. , Int. Ed. , 2003, 42: 268-297
(c)Wernsdorfer W, Aliaga-Alcalde N, Hendrickson D N, et al. Nature, 2002, 416: 406-409
(d)Woodruff D N, Winpenny R E, Layfield R A. Chem. Rev. , 2013, 113: 5110-5148
(e)Leng J D, Liu J L, Zheng Y Z, et al. Chem. Commun. , 2013, 49: 158-160
(f)Zhang P, Zhang L, Tang J K. Dalton Trans. , 2015, 44: 3923-3929 -
[4]
(a)Caneschi A, Gatteschi D, Lalioti N, et al. Angew. Chem. Int. Ed. , 2001, 40: 1760-1763
(b)Werner J, Rams M, Tomkowicz Z, et al. Inorg. Chem. , 2015, 54: 2893-2901
(c)Vaz M G, Cassaro R A A, Akpinar H, et al. Chem. -Eur. J. , 2014, 20: 5460-5467
(d)Wang Y Q, Cheng A L, Liu P P, et al. Chem. Commun. , 2013, 49: 6995-6997
(e)Pardo E, Ruiz-García R, Lloret F, et al. Chem. -Eur. J. , 2007, 13: 2054-2066
(f)Dhers S, Feltham H L, Brooker S. Coord. Chem. Rev. , 2015, 296: 24-44 -
[5]
(a)Neville S M, Halder G J, Chapman K W, et al. J. Am. Chem. Soc. , 2008, 130: 2869-2076
(b)Pardo E, Train C, Boubekeur K, et al. Inorg. Chem. , 2012, 51: 11582-11593
(c)Liu X Y, Qu X N, Zhang S, et al. Inorg. Chem. , 2015, 54: 11520-11525
(d)LI Hai-Qing(李海清), HUA Jing-Kun(华敬坤), ZHA Li-Qin(查丽琴), et al. Chinese J. Inorg. Chem. (无机化学学报), 2015, 31(7): 1417-1424 -
[6]
(a)Weng D F, Wang Z M, Gao S. Chem. Soc. Rev. , 2011, 40: 3157-3181
(b)Ferbinteanu M, Miyasaka H, Wernsdorfer W, et al. J. Am. Chem. Soc. , 2005, 127: 3090-3099
(c)Bogani L, Sangregorio C, Sessoli R, et al. Angew. Chem. Int. Ed. , 2005, 36: 5967-5971
(d)Sessoli R, Powell A K. Coord. Chem. Rev. , 2009, 253: 2328-2341
(e)Jeremies A, Gruschinski S, Meyer M, et al. Inorg. Chem. , 2016, 55: 1843-1853
(f)Liu X Y, Cen P P, Li F F, et al. RSC Adv. , 2016, 6: 96103-96108 -
[7]
(a)Ferlay S, Mallah T, Ouahes R, et al. Nature, 1995, 378: 701-703
(b)Entley W R, Girolami G S. Science, 1995, 268: 397-400
(c)Liu X Y, Sun L, Zhou H L, et al. Inorg. Chem. , 2015, 54: 8884-8886
(d)Chen M, Zhao H, Saudo E C, et al. Inorg. Chem. , 2016, 55: 3715-3717
(e)Liu X Y, Liu H X, Cen P P, et al. Inorg. Chim. Acta, 2016, 447: 12-17 -
[8]
(a)Lescouzec R, Toma L M, Vaissermann J, et al. Coord. Chem. Rev. , 2005, 249: 2691-2729
(b)Miyasaka H, Julve M, Yamashita M, et al. Inorg. Chem. , 2009, 48: 3420-3437
(c)Bernot K, Luzon J, Sessoli R, et al. J. Am. Chem. Soc. , 2008, 130: 1619-1627
(d)Ding M, Wang B, Wang Z, et al. Chem. -Eur. J. , 2012, 18: 915-924
(e)Reger D L, Pascui A E, Smith M D, et al. Inorg. Chem. , 2015, 54: 1487-1500
(f)SUN Lin(孙琳), LIU Huai-Xian(刘怀贤), ZHOU Hui-Liang (周惠良), et al. Chinese J. Inorg. Chem. (无机化学学报), 2015, 31(6): 1207-1214 -
[9]
Kahn O. Molecular Magnetism. New York:VCH, 1993.
-
[10]
(a)Ribas J, Escuer A, Monfort M, et al. Coord. Chem. Rev. , 1999, 1027: 193-195
(b)Zeng Y F, Hu X, Liu F C, et al. Chem. Soc. Rev. , 2009, 38: 469-480
(c)Adhikary C, Koner S. Coord. Chem. Rev. , 2010, 254: 2933-2958
(d)BAI Shi-Qiang(白士强), FANG Chen-Jie(房晨婕), YAN Chun-Hua(严纯华). Chinese J. Inorg. Chem. (无机化学学报), 2006, 22(12): 2123-2134 -
[11]
(a)Hong C S, Do Y. Angew. Chem. , Int. Ed. , 1999, 38: 193-195
(b)Liu T F, Fu D, Gao S, et al. J. Am. Chem. Soc. , 2003, 125: 13976-13977 -
[12]
Gao E Q, Bai S Q, Wang Z M, et al. J. Am. Chem. Soc., 2003, 125:4984-4985 doi: 10.1021/ja034129v
-
[13]
(a)Yoo H S, Kim I J, Yang N, et al. Inorg. Chem. , 2007, 46: 9054-9056
(b)Escuer A, Aroms G. Eur. J. Inorg. Chem. , 2006, 23: 4721-4736 -
[14]
(a)Cheng M, Ding Y S, Gao E Q, et al. Dalton Trans. , 2016, 45: 8028-8035
(b)Schweinfurth D, Sommer M G, Atanasov M, et al. J. Am. Chem. Soc. , 2015, 137: 1993-2005 -
[15]
(a)Liu J, Qin Y L, Qu M, et al. Dalton Trans. , 2013, 42: 11571-11575
(b)Hu K L, Kurmoo M, Wang Z, et al. Chem. -Eur. J. , 2009, 15: 12050-12064
(c)Li J R, Yu Q, Sanudo C, et al. Chem. Commun. , 2007, 25: 2602-2604
(d)Liu X Y, Cen P P, Li H, et al. Inorg. Chem. , 2014, 53: 8088-8097
(e)FAN Yan(范艳), WANG Chen-Min(汪晨敏), QU Zhi-Rong(瞿志荣). Chinese J. Inorg. Chem. (无机化学学报), 2016, 32(5): 864-870 -
[16]
(a)Kahn O, Sikorav S, Gouteron J, et al. Inorg. Chem. , 1983, 22: 2877-2883
(b)Cortes R, Urtiaga M K, Lezama L, et al. Dalton Trans. , 1993, 24: 3685-3694
(c)Thompson L K, Tandon S S. Comments Inorg. Chem. , 1996, 18: 125-144
(d)Zhang L, Zuo J L, Gao S, et al. Angew. Chem. Int. Ed. , 2000, 39: 3633-3635 -
[17]
(a)Ruiz E, Cano J, Alvarez S, et al. J. Am. Chem. Soc. , 1998, 120: 11122-11129
(b)Cabrero J, Graaf C, Bordas E, et al. Chem. -Eur. J. , 2003, 9: 2307-2315
(c)Triki S, García C J G, Ruiz E et al. Inorg. Chem. , 2005, 44: 5501-5508
(d)Mialane P, Dolbecq A, Marrot J, et al. Chem. -Eur. J. , 2005, 11: 1771-1778
(e)Nanda P K, Aromí G, Ray D. Chem. Commun. , 2006, 30: 3181-3183 -
[18]
(a)Shi W B, Cui A L, Kou H Z. ChemPlusChem, 2014, 79: 310-317
(b)Zhang S M, Chen Y H, Wang L H, et al. J. Solid State Chem. , 2015, 226: 201-205
(c)Wang Y Q, Tan Q H, Guo X Y, et al. RSC Adv. , 2016, 6: 72326-72332 -
[19]
(a)Yang L, Zhang S, Liu X Y, et al. CrystEngComm, 2014, 16: 4194-4201
(b)Stamatatos T C, Vlahopoulou G, Raptopoulou C P, et al. Eur. J. Inorg. Chem. , 2012, 19: 3121-3131
(c)Tangoulis V, Panagoulis D, Raptopoulou C P, et al. Dalton Trans. , 2008, 5: 1752-1760 -
[20]
(a)Thompson L K, Tandon S S, Lloret F, et al. Inorg. Chem. , 1997, 36: 3301-3306
(b)Escuer A, Vicente R, Mautner F A, et al. Inorg. Chem. , 1997, 36: 1233-1236 -
[21]
Sheldrick G M. SADABS, Program for Empirical Absorption Correction for Area Detector Data, University of Göttingen, Germany, 1996.
-
[22]
Sheldrick G M. SHELXS-97, Program for the Refinement of Crystal Structures, University of Göttingen, Germany, 1997.
-
[23]
(a)Ruiz E, Alemany P, Alvarez S, et al. J. Am. Chem. Soc. , 1997, 119: 1297-1303
(b)Ruiz E, Rodríguez-Fortea A, Cano J, et al. J. Comput. Chem. , 2003, 24: 982-989
(c)Ruiz E, Cano J, Alvarez S, et al. J. Comput. Chem. , 1999, 20: 1391-1400
(d)Ruiz E. Struct. Bonding, 2004, 113: 71-102 -
[24]
(a)Sarkar S, Datta A, Mondal A, et al. J. Phys. Chem. B, 2006, 110: 12-15
(b)Cremades E, Ruiz E. Inorg. Chem. , 2010, 49: 9641-9648
(c)Gole B, Chakrabarty R, Mukherjee S, et al. Dalton Trans. , 2010, 39: 9766-9778 -
[25]
(a)Neese F. ORCA-an ab initio, Density Functional and Semiempirical Program Package, Ver. 3. 0. 1, University of Bonn, Bonn, Germany, 2013.
(b)Neese F. WIREs Comput. Mol. Sci. , 2012, 2: 73-78 -
[26]
Becke A D. Phys. Rev. A, 1988, 38:3098-3100 doi: 10.1103/PhysRevA.38.3098
-
[27]
(a)Perdew J P. Phys. Rev. B, 1986, 33: 8822-8824
(b)Becke A D. J. Chem. Phys. , 1993, 98: 5648-5652 -
[28]
(a)Schäfer A, Horn H, Ahlrichs R. J. Chem. Phys. , 1992, 97: 2571-2577
(b)Schfer A, Huber C, Ahlrichs R. J. Chem. Phys. , 1994, 100: 5829-5835 -
[29]
Baker G A, Rushbrooke G S. Phys. Rev., 1964, 135:A1272 doi: 10.1103/PhysRev.135.A1272
-
[30]
(a)Tandon S S, Thompson L K, Manuel M E, et al. Inorg. Chem. , 1994, 33: 5555-5570
(b)Thompson L K, Tandon S S, Manuel M E. Inorg. Chem. , 1995, 34: 2356-2366
(c)Sikorav S, Bkouche-Waksman I, Kahn O. Inorg. Chem. , 1984, 23: 490-495 -
[31]
(a)Zhao J P, Hu B W, Saudo E C, et al. Inorg. Chem. , 2009, 48: 2482-2489
(b)Escuer A, Vicente R, Mautner F A, et al. Inorg. Chem. , 1997, 36: 1233-1236
(c)Zhang X M, Wang Y Q, Song Y, et al. Inorg. Chem. , 2011, 50: 7284-7294 -
[32]
(a)Kostakis G E, Mondal K C, Abbas G, et al. CrystEngComm, 2009, 11: 2084-2088
(b)Su Q J, Li S H, Wang L, et al. Inorg. Chem. Commun. , 2010, 13: 1210-1212
(c)Sengupta O, Gole B, Mukherjee S, et al. Dalton Trans. , 2010, 39: 7451-7465
(d)Li X B, Ma Y, Zhang X M, et al. Eur. J. Inorg. Chem. , 2011, 30: 4738-4744
(e)Mukherjee S, Patil Y P, Mukherjee P S. Dalton Trans. , 2012, 41: 54-64
(f)Mukherjee S, Mukherjee P S. Dalton Trans. , 2013, 42: 4019-4030 -
[33]
(a)Zeng Y F, Liu F C, Zhao J P, et al. Chem. Commun. , 2006, 21: 2227-2229
(b)Gu Z G, Song Y, Zuo J L, et al. Inorg. Chem. , 2007, 46: 9522-9524
(c)Liu F C, Zeng Y F, Zhao J P, et al. Inorg. Chem. , 2007, 46: 7698-7700
(d)Han Y F, Wang T W, Song Y, et al. Inorg. Chem. Commun. , 2008, 11: 207-209 -
[34]
(a)Sun W W, Qian X B, Tian C Y, et al. Inorg. Chim. Acta, 2009, 362: 2744-2748
(b)He Z, Wang Z M, Gao S, et al. Inorg. Chem. , 2006, 45: 6694-6705
(c)Zeng Y F, Zhao J P, Hu B W, et al. Chem. -Eur. J. , 2007, 13: 9924-9930
(d)Liu X Y, Chen S P, Grancha T, et al. Dalton Trans. , 2014, 43: 15359-15366
(e)Chakrabarty P P, Giri S, Schollmeyer D, et al. Polyhedron, 2015, 89: 49-54
(f)Setifi Z, Ghazzali M, Glidewell C, et al. Polyhedron, 2016, 117: 244-248
(g)Liu X Y, Li F F, Ma X H, et al. Dalton Trans. , 2017, 46: 1207-1217
-
[1]
-
Figure 1 (a) Structure of 1 with 50% thermal ellipsoids; (b) Simplified tetragonal pyramidal geometry of the center Cu(Ⅱ) cation for 1; (c) 1D chain with carboxylate and azido bridges for 1; (d) Hydrogen bonding formed by azido and water molecules between adjacent chains in 1
Hydrogen atoms are omitted for clarity in (a)~(c); Symmetry codes: ⅰ-x+1, y-1/2, -z+1/2
Table 1. Selected crystallographic data and structure refinement for compound 1
Empirical formula C8H7CuN3O4 Formula weight 272.71 Crystal system Monoclinic Space group P21/c a / nm 0.758 02(8) b / nm 0.661 77(7) c / nm 2.008 2(2) β/(°) 97.707(2) V / nm3 0.998 30(18) Z 4 D / (Mg·m-3) 1.814 μ/ mm-1 2.19 F(000) 548 Unique reflections 2 080 Observed reflections 5 246 Rint 0.027 5 Data, restraint, parameter 2 080, 1, 150 Final R indices [I>2σ(I)] R1=0.024 9, wR2=0.066 8 R indices (all data) R1=0.028 9, wR2=0.068 4 Goodness-of-fit on F2 1.008 Table 2. Comparison of the experimental (from fitting) and DFT studies
1 EHS/eV EBS/eV JDFT / cm-1 JExpt / cm-1 BP86 def2-tzvp -150 464.859 80 -150 464.849 39 83.54 72.1 def2-svp -150 381.266 57 -150 381.256 40 81.51 tzv -150 442.523 75 -150 442.513 24 84.23 tzvp -150 461.971 63 -150 461.960 85 86.60 B3LYP def2-tzvp -150 413.412 20 -150 413.401 33 87.63 def2-svp -150 329.346 82 -150 329.335 65 89.97 tzv -150 389.311 06 -150 389.299 97 89.39 tzvp -150 410.351 84 -150 410.340 80 88.96 -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 2
- 文章访问数: 1185
- HTML全文浏览量: 75


 下载:
下载:




 下载:
下载:

