 图1
XRD patterns of NaTaO3, Ag-NaTaO3, NaTaO3-RGO and Ag-NaTaO3-RGO with different Ag content
Figure1.
XRD patterns of NaTaO3, Ag-NaTaO3, NaTaO3-RGO and Ag-NaTaO3-RGO with different Ag content
图1
XRD patterns of NaTaO3, Ag-NaTaO3, NaTaO3-RGO and Ag-NaTaO3-RGO with different Ag content
Figure1.
XRD patterns of NaTaO3, Ag-NaTaO3, NaTaO3-RGO and Ag-NaTaO3-RGO with different Ag content

Ag-NaTaO3-RGO复合物的合成及其改进的光催化制氢性能
English
Ag-NaTaO3-RGO Composite: Synthesis and Improved Photocatalytic Hydrogen Production Property
-
Key words:
- photocatalysis
- / NaTaO3
- / graphene
- / hydrogen production
- / Ag nanoparticles
-
0 Introduction
In recent decades, energy deficiency and environ-ment pollution has been becoming severe issues and challenges for humanity. Because of its environmental benignity, recyclability, hydrogen energy has been considered as one of the most promising replacement energy resources and photocatalytic water splitting over photocatalysts to generate hydrogen has attracted intensive interest[1-5]. Therefore, the research of new highly active photocatalysts that could make full use of solar energy by transforming it to hydrogen energy has become a research focus in photocatalysis field. Due to their excellent photocatalytic properties, Alkali tantalite has attracted considerable attention in photocatalytic applications[6-9]. In particular, Perovskite NaTaO3 exhibited fairly high activity for splitting of pure water because of its appropriate bandgap. However, the efficiency of NaTaO3 was far from satisfaction due to its inherent high recombination rate of photogenerated electron-hole pairs. So far, various strategies to improve the photocatalytic activity of NaTaO3 have been developed, including deposition of noble metal[10-11], doping with metal ion[12-14], and coupling with semiconductor materials[15-16], etc. Among these strategies, coupling NaTaO3 with other materials to form a composite photocatalyst has been proved to be an effective way for improving the photoconversion efficiency[17].
Graphene, a single layer of graphite, shows many intriguing electronic and optoelectronic properties and has been regarded as ideal 2D supports for making various functional composite materials[18-20]. In parti-cular, graphene is widely recognized to serve as an electron collector and transporter to efficiently hinder electron-hole recombination and lengthen the lifetime of the photo-generated charge carriers from semicond-uctor[21-23]. Coupling graphene with photocatalyst for enhancing their photocatalytic performance has been paid extensive attention. For instance, Zhu et al.[24] recently demonstrated that ZnS nanoparticles exhibited a high photocatalytic H2-production activity by combining with graphene. Hou et al.[25] reported that nitrogen-doped graphene could improve the photoelectrochemical and photocatalytic activity of graphitic carbon nitride (g-C3N4) nanosheet. On the other hand, coupling with novel metal nanoparticles Ag also has been proven as a promising method to improve the photocatalytic activity of semiconductor[26]. For example, Yu et al.[27] reported that Ag-TiO2 nano-composites exhibit excellent performance in photo-catalytic degradation of dyes under the UV illumina-tion since silver particles could act as electron traps aiding electron-hole separation. According to their report, when the concentration of AgNO3 increased to 0.03 mol·L-1, the photocatalytic activity of Ag-TiO2 composite significantly increased and was 6.3 times higher than that of TiO2 thin films.
However, to the best of our knowledge, so far there is no report on coupling NaTaO3 with Ag and RGO at the same time. Herein, we report the preparation and characterization of Ag-NaTaO3-RGO composite through a three-step method for the first time. Such architecture provides improved separation rate of photo-generated electron-hole pairs. As a result, the composite is anticipated to exhibit enhanced photocatalytic activity under UV irradiation.
1 Experimental
1.1 Preparation of Ag-NaTaO3-RGO composite
All chemicals were analytical grade and were used without further purification. Deionized water (DI) was used in all experiments. Graphene oxide was prepared from natural graphite powders through the modified Hummers′ method[28]. NaTaO3 were synthes-ized by the hydrothermal method based on our previous work with modifications[29]. Typically, Ta2O5 (0.442 0 g), CH3COONa·3H2O (0.544 0 g), NaOH (0.120 0 g) and DI water (30 mL) were added into a beaker (100 mL) and stirred for 2 h then transferred to a 50 mL Teflon-lined stainless steel autoclave. Finally, the autoclave was maintained at 180 ℃ for 24 h and then allowed to cool to room temperature naturally. After filtering, washing, and drying, the NaTaO3 samples were obtained.
A typical synthesis of Ag-NaTaO3-RGO composite was described as follows: GO (0.015 0 g) and DI water (30 mL) were added into a beaker (100 mL) and sonicated for 30 min, a homogenous GO solution was obtained. Then, 2.000 0 g polyvinylpyrrolidone (PVP) was added into the GO water solution and heated in 60 ℃ water bath with vigorous stirring till the PVP being completely dissolved. After that 0.500 0 g NaTaO3 was added to the above aqueous solution and stirred for 2 h. Then, the suspension was heated in 60 ℃ water bath for 24 h while maintaining vigorous stirring after added calculated amount of AgNO3 (3 mL). The obtained Ag-NaTaO3-RGO composites were then washed by distilled water to get rid of residual PVP. Finally, the Ag-NaTaO3-RGO composites were freeze-dried at -50 ℃ for 24 h. A series of Ag-NaTaO3-RGO photocatalysts were prepared by changing the concentration of AgNO3 (0.1, 0.2, 0.4, 0.6 mol·L-1, respectively) and marked as e.g. 0.2Ag-NaTaO3-RGO.
1.2 Characterizations
X-ray powder diffraction (XRD) patterns were taken on a X-ray diffractometer (MSAL-XRDⅡ) using a Cu Kα radiation (λ=0.154 056 nm) at a scan rate of 8°·min-1 (40 kV, 20 mA, 2θ=10°~80°). Transmission electron microscopy (TEM) and high-resolution trans-mission electron microscopy (HRTEM) images were obtained using a PHILIPS TECNAI-10 microscope. Field emission scanning electron microscopy (FESEM) investigations were taken on a Zeiss Ultra 55 field scanning electron microscope at an accelerating voltage of 15 kV, the fracture surface of sample was coated with a thin layer of gold before analysis. The UV-Vis diffuse reflection spectra (DRS) were determined by a Shimadzu UV-2501PC UV-Vis spectrophotometer, equipped with an integration sphere attachment for their diffuse reflectance in the range of 200~800 nm. BaSO4 was used as the standard in all measurements. Photoluminescence (PL) emission spectra were recorded using a Hitachi F-4500. The excitation wavelength was 250 nm.
1.3 Evaluation of photocatalytic activity
The photocatalytic performance of the obtained samples were evaluated by photocatalytic water splitting hydrogen-evolution under UV irradiation and carried out in a photocatalytic reaction system (CEL-HX300, Beijing Chinese Education Au-Light Co., Ltd.). For photocatalytic hydrogen production, 0.1 g catalyst was well dispersed into deionized water (20 mL) placed in the photocatalytic reaction system, then 80 mL Na2S (0.1 mol·L-1) and Na2SO3 (0.04 mol·L-1) aqueous solution was added into the suspension as sacrificial agent. After purge the whole system with N2 for 30 min, the suspension was illuminated under a 280 W Hg-lamp (λ>250 nm) continuously and kept stirring. The obtained gas was quantitative analyzed by online analysis with a gas chromatograph (GC9800, Shanghai Kechuang Technology Co., Ltd.).
2 Results and discussion
2.1 XRD analysis
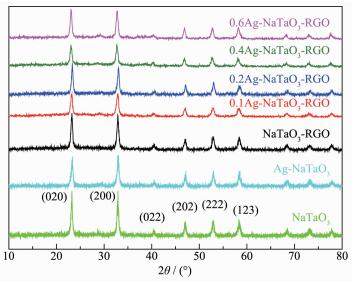
The XRD patterns of NaTaO3, Ag-NaTaO3, NaTaO3 -RGO and Ag-NaTaO3-RGO with different Ag content were shown in Fig. 1. As shown, the X-ray diffraction patterns of NaTaO3-RGO coincide well with that of pure NaTaO3. The peaks at 2θ of 22.85°, 32.55°, 40.23°, 46.67°, 52.58° and 58.40° are well assigned to the (020), (200), (022), (202), (222) and (123) crystal planes of cubic NaTaO3, respectively. Noticeably, the peaks for RGO are not observed in the diffraction patterns of NaTaO3-RGO and Ag-NaTaO3-RGO composites, which may be due to the low amount and relatively low diffraction intensity of RGO. However, the presence of RGO could be discerned by SEM and TEM, as discussed later. Moreover, the XRD patterns of Ag-NaTaO3-RGO with different Ag content are similar and no signal about silver can be detected. This may be due to the fact that the Ag nanoparticles were dispersedly loaded on the surface of the NaTaO3 and RGO.
 图1
XRD patterns of NaTaO3, Ag-NaTaO3, NaTaO3-RGO and Ag-NaTaO3-RGO with different Ag content
Figure1.
XRD patterns of NaTaO3, Ag-NaTaO3, NaTaO3-RGO and Ag-NaTaO3-RGO with different Ag content
图1
XRD patterns of NaTaO3, Ag-NaTaO3, NaTaO3-RGO and Ag-NaTaO3-RGO with different Ag content
Figure1.
XRD patterns of NaTaO3, Ag-NaTaO3, NaTaO3-RGO and Ag-NaTaO3-RGO with different Ag content
2.2 XPS analysis
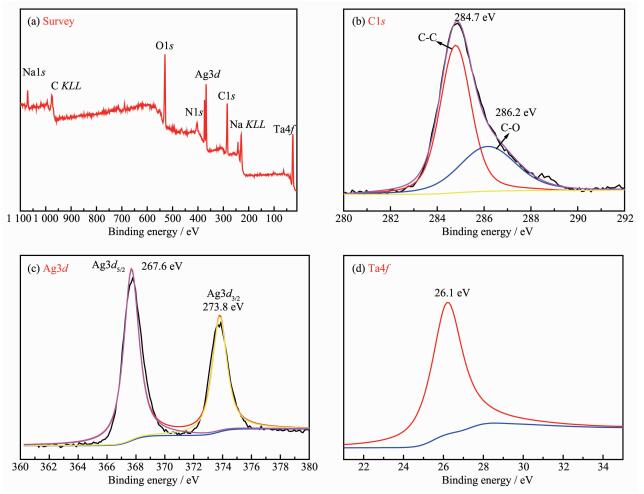
To further probe the chemical structure of the as-prepared samples, the XPS measurements were conducted. In Fig. 2a, the XPS survey spectrum of 0.2Ag-NaTaO3-RGO display the presence of C, N, O, Na, Ag and Ta elements. As shown in Fig. 2b, the C1s spectrum of 0.2Ag-NaTaO3-RGO could be deconvol-uted into two peaks. The peak at 284.78 and 288.29 eV could be attributed to the C-C and oxygen functional groups C-OH. This indicates that most of GO has been reduced to RGO. Two peaks in Fig. 2c centered at 367.6 and 373.8 eV could be attributed to Ag3d5/2 and Ag3d3/2, respectively. And the Ta4f peak for 0.2Ag-NaTaO3-RGO was shown in Fig. 2d, with the value of about 26.1 eV, confirming that Ta exists mainly in the Ta5+ chemical state on the sample surface.
2.3 Microtopography analysis
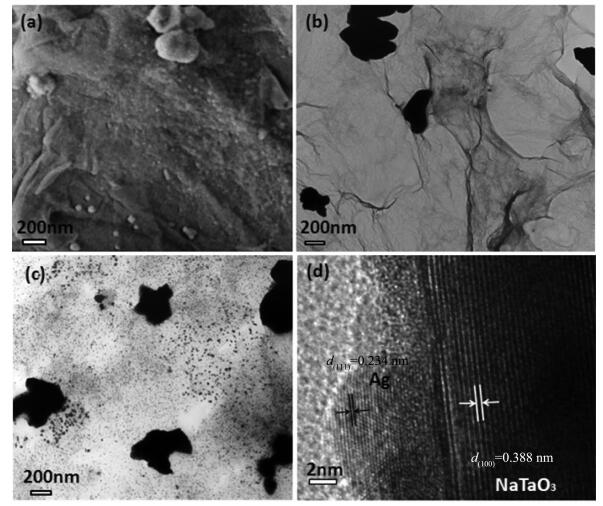
Fig. 3a is the SEM image of 0.2Ag-NaTaO3-RGO. As shown, the layered structure of the stacked RGO sheets can be clearly seen. The NaTaO3 particles and the Ag nanoparticles are well distributed on RGO nanosheets. The average size of NaTaO3 and Ag nanoparticles are about 50~100 nm and 10 nm, respectively. Obviously, the average size of NaTaO3 is much bigger than that of Ag, which is consistent with the result of TEM. Fig. 3b and Fig. 3c showed the TEM images of NaTaO3-RGO and 0.2Ag-NaTaO3-RGO composite. As shown in Fig. 3b, the light-gray thin RGO sheets are observed, and NaTaO3 particles are uniformly distributed on the graphene nanosheets. Fig. 3c further reveals that a large number of Ag nanoparticles spread uniformly and densely on the surface of graphene, which is consistent with the observation from SEM image. The HRTEM image in Fig. 3d reveals the well-defined lattice fringes of NaTaO3 and Ag nanoparticle with the spacings of 0.388 and 0.234 nm, corresponding to the (100) and (111) planes of NaTaO3 and Ag, respectively.
2.4 Spectra analyses
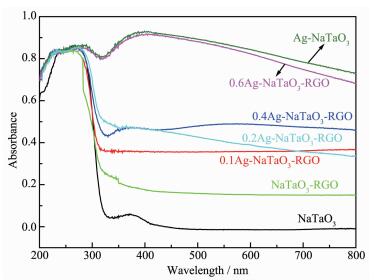
Fig. 4 exhibits the UV-Vis diffuse reflection spectra of NaTaO3, Ag-NaTaO3, NaTaO3-RGO and Ag-NaTaO3-RGO with different Ag content. The absorp-tion edge of NaTaO3 is at around 300 nm and which indicates that NaTaO3 hardly has photoresponse property under visible light, which is due to the intrinsic large band gap. Compared with pure NaTaO3, a red-shift of the absorption edge is observed for the NaTaO3-RGO composite, which may be attributed to the bonding effect between GO and NaTaO3. Moreover RGO loading enhances its light absorption over the entire range of wavelength, this is a typical behavior of graphene as previously reported[30]. Obviously, with the increase of Ag content, the absorption intensity of Ag-NaTaO3-RGO composites over visible light region increases. Such enhanced absorption in the visible light region may be attributed to the surface plasmon resonance (SPR) effect of Ag nanoparticles[11, 31]. How-ever, the influence of Ag nanoparticles on the band gap of NaTaO3 is almost negligible. So the increase of the visible light absorbance is not the main factor for the improvement of the photocatalytic activity.
PL is an effective strategy to characterize the separation and recombination rates of charge carriers of a photocatalyst. As shown in Fig. 5, the PL spectra of NaTaO3 exhibit a strong emission peaks centered at around 400 nm, while Ag-NaTaO3 and NaTaO3-RGO exhibit a weak fluorescence emission spectrum peak, implying that the photogenerated electrons and holes have better separation in Ag-NaTaO3 and NaTaO3-RGO composites. This result reveals that Ag and RGO play an important role in suppressing the recombination of the photogenerated carriers, which efficiently preve-nted the recombination of electrons and holes. In the case of 0.2Ag-NaTaO3-RGO, the photoluminescence intensity of peak at 400 nm further reduced. The results further indicat that the effect of Ag nanopar-ticles is quite similar to that of RGO, namely Ag can obviously enhance the separation of electrons and holes[10].
2.5 Photocatalytic activity analysis
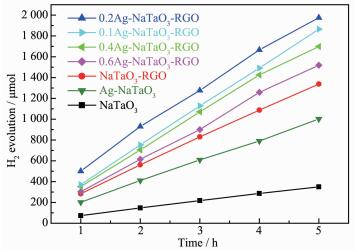
The photocatalytic activities of the samples were evaluated by photocatalytic water splitting hydrogen-evolution in a Na2S/Na2SO3 aqueous solution under UV irradiation. As shown in Fig. 6, the hydrogen production rate of NaTaO3 is about 70 μmol·h-1. The obvious increased of hydrogen production rate over Ag-NaTaO3 and NaTaO3-RGO (around 200.4 and 267.4 μmol·h-1) was observed. Obviously, all the Ag-NaTaO3-RGO composites have much higher hydrogen production rate under the identical conditions. Among all the Ag-NaTaO3-RGO samples, the as-prepared 0.2Ag-NaTaO3-RGO exhibits the highest H2 production rate (395 μmol·h-1) which is around 5.64, 1.97 and 1.48 times higher than that of pure NaTaO3, Ag-NaTaO3 and NaTaO3-RGO. We expect that the following factors may be responsible for the much greater photocatalytic activities of 0.2Ag-NaTaO3-RGO composites: (ⅰ) the efficient separation of electron-hole pairs originated from the excellent electron transfer property of graphene. (ⅱ) the further improvement of electron-hole separation rate due to Ag nanoparticle acted as electron traps. (ⅲ) the enhancement of light absorption over the entire range of wavelengths with the introduction of graphene[32-33]. In conclusion, graph-ene and Ag nanoparticles play very important roles for the improvement of photocatalytic activity of Ag-NaTaO3-RGO composite.
From all experimental results mentioned above, a possible reaction mechanism was tentatively illustrated in Fig. 7. Under ultraviolet illumination, the VB electrons of NaTaO3 are excited to the CB, creating holes in the VB. The holes react with H2O to form O2, and the photogenerated electrons in the CB of NaTaO3 transferr to graphene sheets since the redox potential of graphene/graphene- is lower than that of the CB of NaTaO3. During the process, graphene can greatly enhance separation rate of electron-hole pairs due to its excellent electron transfer property, and Ag nanoparticle can further improve the electron-hole separation rate because it can act as electron traps. As a result, the Ag-NaTaO3-RGO composites enhance photocatalytic H2 production activity as the recombination of photo-generated electron-hole pairs can be suppressed effectively due to the introduction of graphene and Ag nanoparticles.
3 Conclusions
In summary, we have rationally designed a ternary composite photocatalysts consisting of Ag, RGO and NaTaO3. In this unique ternary Ag-NaTaO3-RGO composite, RGO promotes the electron transfer from the CB of NaTaO3 to graphene and simultaneously enhances the light absorption over the entire range of wavelengths. At the same time, Ag nanoparticles act as electron traps which can further improve the separation of electron-hole pairs. As a result, Ag-NaTaO3-RGO enhances photocatalytic activity for water splitting hydrogen-evolution. This endeavor paves the way to build reliable triple-composites photo-catalysts for photocatalysis applications.
-
-
[1]
Wang X, Maeda K, Thomas A, et al. Nat. Mater., 2009, 8(1):76-80 doi: 10.1038/nmat2317
-
[2]
Ou H H, Lin L H, Zheng Y, et al. Adv. Mater., 2017, 29(22):1700008 doi: 10.1002/adma.v29.22
-
[3]
Yu J G, Qi L F, Jaroniec M. J. Phys. Chem. C, 2010, 114(30):13118-13125 doi: 10.1021/jp104488b
-
[4]
Liu J, Liu Y, Liu N Y, et al. Science, 2015, 347(6225):970-974 doi: 10.1126/science.aaa3145
-
[5]
Godin R, Wang Y, Zwijnenburg M A, et al. J. Am. Chem. Soc., 2017, 139(14):5216-5224 doi: 10.1021/jacs.7b01547
-
[6]
Kato H, Kudo A. J. Phys. Chem. B, 2001, 105(19):4285-4292 doi: 10.1021/jp004386b
-
[7]
Li X, Zang J L. J. Phys. Chem. C, 2009, 113(45):19411-19418 doi: 10.1021/jp907334z
-
[8]
Gömpel D, Tahir M N, Panthöfer M, et al. J. Mater. Chem. A, 2014, 2(21):8033-8040 doi: 10.1039/C4TA00183D
-
[9]
章薇, 谈国强, 夏傲, 等.硅酸盐学报, 2011, 39(11):1724-1728ZHANG Wei, TAN Guo-Qiang, XIA Ao, et al. J. Chin. Ceram Soc., 2011, 39(11):1724-1728
-
[10]
Xu D, Chen M, Song S, et al. CrystEngComm., 2014, 16(7):1384-1388 doi: 10.1039/c3ce41919c
-
[11]
Xu D B, Yang S B, Jin Y, et al. Langmuir, 2015, 31(35):9694-9699 doi: 10.1021/acs.langmuir.5b01294
-
[12]
Kato H, Asakura K, Kudo A. J. Am. Chem. Soc., 2003, 125(10):3082-3089 doi: 10.1021/ja027751g
-
[13]
Kudo A, Niishiro R, Iwase A, et al. Chem. Phys., 2007, 339(1/2/3):104-110
-
[14]
崔华楠, 石建英, 刘鸿.催化学报, 2015, 36(7):969-974CUI Hua-Nan, SHI Jian-Ying, LIU Hong. Chin. J. Catal., 2015, 36(7):969-974
-
[15]
Deng Y Y, Chen Y J, Chen B G, et al. J. Alloys Compd., 2013, 559:116-122 doi: 10.1016/j.jallcom.2013.01.069
-
[16]
Reddy K H, Martha S, Parida K M. RSC Adv., 2012, 2(25):9423-9436 doi: 10.1039/c2ra21083e
-
[17]
Meyer T, Priebe J B, da Silva R O, et al. Chem. Mater., 2014, 26(16):4705-4711 doi: 10.1021/cm500949x
-
[18]
Geim A K. Science, 2009, 324(5934):1530-1534 doi: 10.1126/science.1158877
-
[19]
Bai X J, Sun C P, Liu D, et al. Appl. Catal., B, 2017, 204:11-20 doi: 10.1016/j.apcatb.2016.11.010
-
[20]
Stankovich S, Dikin D A, Dommett G H, et al. Nature, 2006, 442(7100):282-286 doi: 10.1038/nature04969
-
[21]
Li B X, Liu T X, Wang Y F, et al. J. Colloid Interface Sci., 2012, 377(1):114-121 doi: 10.1016/j.jcis.2012.03.060
-
[22]
Li Q, Guo B D, Yu J G, et al. J. Am. Chem. Soc., 2011, 133(28):10878-10884 doi: 10.1021/ja2025454
-
[23]
李娟, 赵安婷, 邵姣婧, 等.无机化学学报, 2017, 33(7):1231-1235 doi: 10.11862/CJIC.2017.167LI Juan, ZHAO An-Ting, SHAO Jiao-Jing, et al. Chinese J. Inorg. Chem., 2017, 33(7):1231-1235 doi: 10.11862/CJIC.2017.167
-
[24]
Zhu B L, Lin B Z, Zhou Y, et al. J. Mater. Chem. A, 2014, 2(11):3819-3827 doi: 10.1039/C3TA14819J
-
[25]
Hou Y, Wen Z H, Cui S M, et al. Adv. Mater., 2013, 25(43):6291-6297 doi: 10.1002/adma.201303116
-
[26]
李晓芬, 陈梦莹, 梁诗景, 等.无机化学学报, 2016, 32(11):1987-1994 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?file_no=20161113&flag=1LI Xiao-Fen, CHEN Meng-Ying, LIANG Shi-Jing, et al. Chinese J. Inorg. Chem., 2016, 32(11):1987-1994 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?file_no=20161113&flag=1
-
[27]
Yu J G, Xiong J F, Cheng B, et al. Appl. Catal., B, 2005, 60(3/4):211-221
-
[28]
William S, Hummers J R, Offeman R E. J. Am. Chem. Soc., 1958, 80(6):1339-1339 doi: 10.1021/ja01539a017
-
[29]
黄浪欢, 产启中, 张斌, 等.催化学报, 2011, 32(11):1822-1830HUANG Lang-Huan, CHAN Qi-Zhong, ZHANG Bing, et al. Chin. J. Catal., 2011, 32(11):1822-1830
-
[30]
Xiang Q J, Yu J G, Jaroniec M. J. Am. Chem. Soc., 2012, 134(15):6575-6578 doi: 10.1021/ja302846n
-
[31]
Xu D B, Liu K L, Shi W D, et al. Ceram. Int., 2015, 41(3):4444-4451 doi: 10.1016/j.ceramint.2014.11.136
-
[32]
Xiang Q J, Yu J G, Jaroniec M. Chem. Soc. Rev., 2012, 41(2):782-796 doi: 10.1039/C1CS15172J
-
[33]
Hermann J M, Tahiri H, Ait-Ichou Y, et al. Appl. Catal., B, 1997, 13(3/4):219-228
-
[1]
-
-

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 5
- 文章访问数: 1731
- HTML全文浏览量: 175




 下载:
下载:






 下载:
下载:

