
一类六核三价铁簇化合物的合成及结构表征
English
Syntheses and Structures Characterization of a Family of Hexanuclear Iron(Ⅲ) Clusters
-
Key words:
- hexanuclear iron (Ⅲ) clusters
- / crystal structure
- / counter ions
-
0 Introduction
The polynuclear transition metal clusters have atracted numerous attention since they simultaneously display pleasing architectural aesthetics and fascinating magnetic properties[1-6]. The investigation of such magnetic materials is helpful in understanding the magnetio-structural correlations and provides a fertile platform for the research of the attractive mag-netic characteristics, such as ferromagnetic ordering[7], antiferromagnetic ordering[8], ferrimagnetic ordering[9-11], spin-canting[12-13], metamagnetic transition[14], spin-glass[15-17], spin-flop[18-20], and spin-frustrated[21-23], etc[24-26]. Although there is great success made in the practice, the design and synthesis of such materials still remain an elusive goal and a major challenge to systema-tically study the structure-property relationship[27]. To obtain a magnetic polynuclear cluster, incorporation of paramagnetic transition-metal ions with appropriate multidentate chelating ligands is a general strategy. In this respect, we chose Fe(Ⅲ) ion, with S=5/2, as the paramagnetic center due to the exchange anisotropy and the large spin ground state achieved[28-30]. As for the main organic ligand, we selected tripodal alcohol ligand, i.e., 1, 1, 1-tris(hydroxymethyl)propane (abbrev-iated H3tmp), which is a very attractive candidate for the construction of multinuclear clusters, due to its flexibility and diverse coordination modes as well as superexchange capacity reflected in interesting magnetic properties[31]. It is well known that nitrogen-containing heterocyclic groups generally act as a role of bridging ligands, due to their strong σ-electron donors to metals[32-33]. Furthermore, they can also perform counter cations because they are weak base and prone to protonate. Therefore, we have concentrated our efforts on the combination of nitrogen-containing heterocyclic ligands, which act as assistant role, with Fe(Ⅲ)-H3tmp system to explore the structures and investigate the resultant magnetic properties.
In this paper, we have successfully assembled and investigated three hexanuclear compounds, (C5N2H14)[Fe6(μ6-O)Cl6(tmp)4]·2H2O(·CH3OH (1), (C3N2H5)2[Fe6(μ6-O)Cl6(tmp)4] (2), and (C4N3H8)3(C2N3H4)[Fe12(μ6-O)2 Cl12(tmp)8]·3CH3OH (3). These three compounds possess different protonated nitrogen-containing heterocyclic counter cations, despite the same [Fe6(μ6-O)Cl6(tmp)4]2- core. Within the system of compounds 1 and 3, the nitrogen-containing heterocyclic ligands experience N-and C-alkylation reactions, which is validated by the crystallographic parameters, elemental analyses, and IR.
1 Experimental
1.1 Reagents and physical measurements
All chemicals and solvents were of reagent grade and used as commercially purchased without further purification.
Infrared spectra (400~4 000 cm-1) were measured on a Perkin-Elmer Fourier transform infrared (FTIR) spectrophotometer using KBr pellets (Fig.S1, Support-ing information). The elemental analyses (C, H and N) were performed on a Perkin-Elmer Model 240C elemental analyzer. The Fe and Cl elemental contents were determined by the Leaman inductively coupled plasma (ICP) spectrometer. Thermogravimetric analyses (TGA) were performed on a Perkin-Elmer TG-7 analyzer in flowing nitrogen atmosphere from room temperature to 800 ℃ with a heating rate of 10 ℃·min-1. Powder X-ray diffraction (PXRD) measurements were assessed on a Siemens D5005 diffractometer with Cu Kα (λ=0.154 184 nm) in 2θ range of 5°~50° at room temperature. The XRD accelerating voltage and emiss-ion current were 40 kV and 30 mA, respectively.
1.2 Syntheses of complexes 1~3
1.2.1 Synthesis of (C5N2H14)[Fe6(μ6-O)Cl6(tmp)4]·2H2O·CH3OH (1)
A mixture of FeCl3 (0.049 g, 0.3 mmol), H3tmp (0.027 g, 0.2 mmol), piperazine (0.026 g, 0.3 mmol), and methanol (15 mL) was placed in a 20 mL Teflon-lined autoclave and heated under autogenous pressure at 120 ℃ for 72 h. Then, the autoclave was cooled to ambient temperature at 3 ℃·h-1. Block-shaped red crystals of 1 were collected in ~80% yield (based on Fe). Anal. Calcd. for C30H66Cl6Fe6N2O16(%): C, 28.62; H, 5.25; N, 2.23; Fe, 26.64; Cl, 16.91. Found(%): C, 28.98; H, 5.14; N, 2.33; Fe, 26.51; Cl, 17.11. IR (KBr, cm-1): 3 489 (m), 2 966 (m), 2 914 (m), 2 868 (m), 1 623 (w), 1 472 (m), 1 453 (m), 1 398 (w), 1 356 (w), 1 304 (w), 1 198 (w), 1 111 (s), 1 034 (s), 941 (s), 771 (w), 593 (m), 527 (s), 473 (m), 437 (w).
1.2.2 Synthesis of (C3N2H5)2[Fe6(μ6-O)Cl6(tmp)4] (2)
A mixture containing FeCl3 (0.081 g, 0.5 mmol), H3tmp (0.027 g, 0.2 mmol), imidazole (0.014 g, 0.2 mmol), and methanol (15 mL) was heated in a Teflon-lined autoclave at 120 ℃ for 72 h and then allowed to cool to room temperature at the rate of 3 ℃·h-1. Needle-like red crystals of 2 were obtained in ~85% yield (based on Fe). Anal. Calcd. for C30H54Cl6Fe6N4O13(%): C, 29.36; H, 4.41; N, 4.57; Fe, 27.34; Cl, 17.35. Found(%): C, 29.05; H, 4.66; N, 4.39; Fe, 27.84; Cl, 17.64. IR (KBr, cm-1): 3 148 (m), 2 972 (m), 2 915 (m), 2 868 (m), 1 585 (m), 1 472 (m), 1 455 (w), 1 399 (w), 1 303 (w), 1 199 (w), 1 110 (s), 1 033 (s), 990 (w), 941 (s), 821 (w), 794 (w), 771 (w), 755 (w), 628 (m), 591 (m), 528 (s), 474 (m), 438 (w).
1.2.3 Synthesis of (C4N3H8)3(C2N3H4)[Fe12(μ6-O)2Cl12(tmp)8]·3CH3OH (3)
A mixture of FeCl3 (0.081 g, 0.5 mmol), H3tmp (0.041 g, 0.3 mmol), triazole (0.138 g, 2 mmol), and methanol (15 mL) was heated in Teflon-lined autoclave at 140 ℃ for 72 h. After the autoclave was cooled to room temperature at 3 ℃·h-1, red block-shaped crystals of 3 were afforded (Yield: 75% based on Fe). Anal. Calcd. for C65H128Cl12Fe12N12O29(%): C, 29.59; H, 4.856; N, 6.37; Fe, 25.43; Cl, 16.14. Found(%): C, 28.31; H, 4.921; N, 6.12; Fe, 25.49; Cl, 16.78. IR (KBr, cm-1): 3 464 (w), 3 075 (m), 2 964 (m), 2 911 (m), 2 866 (m), 1 628 (w), 1 584 (m), 1 470 (m), 1 452 (m), 1 398 (m), 1 302 (w), 1 198 (w), 1 165 (w), 1 112 (s), 1 038 (s), 942 (s), 771 (w), 733 (w), 659 (w), 625 (m), 592 (m), 526 (s), 473 (m).
1.3 Details of X-ray crystallography
Single-crystal X-ray diffraction data for complexes 1~3 were measured on a Bruker SMART APEX CCD area detector diffractometer using graphite monochro-mated Mo Kα radiation (λ=0.071 073 nm) at room temperature. Structure solution (direct methods) and the refinement of full-matrix least-squares were carried out using the SHELXTL software package[34]. All the non-hydrogen atoms were refined with anisotropic thermal parameters. The hydrogen atoms attached to carbon and nitrogen atoms were placed in geometrically calculated positions. For complex 1, the hydrogen atoms on the non-coordinated MeOH molecule, that is O(14), and on water molecules, that are O(1W) and O(2W), were not located. The atoms C(30), O(14), O(1W) and O(2W) were found disordered over two sites. For complex 2, the atom C(1) was highly disord-ered over two sites. For complex 3, the hydrogen atom on one non-coordinated MeOH molecule, that is O(14), was not located. The atoms C(13) and C(14) were highly disordered over two sites. The protonated degrees of all the counter cations are according to the charge balance. Crystallographic and refinement details for all compounds are summarized in Table 1. Selected bond lengths and angles are listed in Table S1~S3.
1 2 3 Formula C30H66Cl6Fe6N2O16 C30H54Cl6Fe6N4O13 C65H128Cl12Fe12N12O29 Formula weight 1 257.8 1 225.8 2 635.6 Space group Pbca R3c P1 Crystal system Orthorhombic Trigonal Triclinic a/nm 1.960 1(7) 2.746 9(3) 1.136 1(2) b/nm 1.922 3(7) 2.746 9 1.144 3(2) c/nm 2.550 4(9) 3.546 7(4) 2.195 5(4) α/(°) 96.408(2) β/(°) 101.976(3) γ/(°) 122.667(2) V/nm3 9.610(6) 23.176(3) 2.517 8(8) Z 8 18 1 Dc/(g·cm-3) 1.731 1.576 1.738 F(000) 5 117.2 11 155.9 1 348 θ range/(°) 1.60~25.04 1.48~25.00 1.94~25.21 Unique reflns (Rint) 8 506 (0.069 5) 4 555 (0.051 2) 9 078 (0.024 3) R1a, wR2b [I>2σ(I)] 0.043 9, 0.116 8 0.044 5, 0.146 4 0.051 7, 0.117 7 R1a, wR2b (all data) 0.082 3, 0.139 1 0.060 3, 0.156 3 0.099 4, 0.148 7 GOF on F2 1.072 1.09 1.027 aR1=∑||Fo|-|Fc||/∑|Fc|; bwR2=[∑w(|Fo|-|Fc|)2/∑w(FO2)]1/2; w=1/σ(Fo)2. CCDC: 980726, 1; 980727, 2; 980728, 3.
2 Results and discussion
2.1 Synthesis
Complex 1 was prepared by the solvothermal reaction of FeCl3, H3tmp, and piperazine in a 3:2:3 molar ratio in MeOH at 120 ℃ for 72 h. Interestingly, the piperazine ligand in such system experienced N-alkylation reaction to form methyl substituent pipera-zine (abbreviated as L1). Complex 3 was synthesized by the solvothermal reaction of FeCl3, H3tmp, and triazole in a 5:3:20 molar ratio in MeOH at 140 ℃ for 72 h. Part of triazole in such system went through C-and N-alkylation reactions to form dimethyl substituent triazole (abbreviated as L2). The reasons for such N/C-alkylation reactions in the systems 1 and 3 are unclear due to the complexity under the solvothermal condition. Complex 2 was obtained by the reaction of FeCl3 with H3tmp and imidazole in a 5:2:2 molar ratio in MeOH at 120 ℃ for 72 h.
2.2 X-ray crystal structures
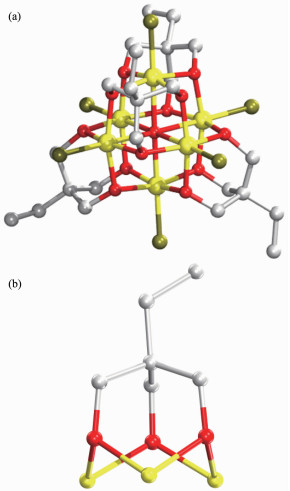
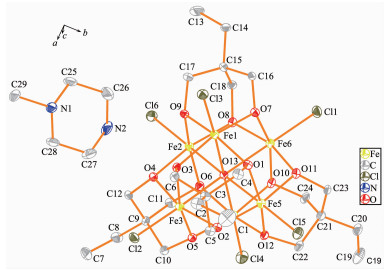
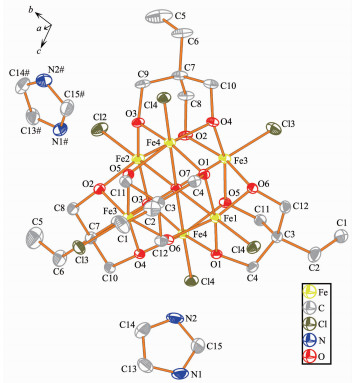
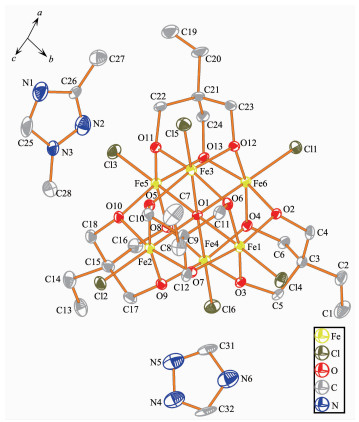
Single-crystal X-ray diffraction results of 1~3 reveal that each compound possesses same anionic structure unit composed of [Fe6(μ6-O)Cl6(tmp)4]2- core (Fig. 1), which exhibits marked similarities to previously reported compounds contained 1, 1, 1-tris(hydroxymet-hyl)ethane (H3thme)[35-36]. In the anionic core, the ligand H3tmp possesses the level of deprotonation tmp3- and adopts the η2:η2:η2:μ3 coordinated mode[31]. The central μ6-O approximately locates in the center of an octahedron formed by six Fe(Ⅲ) atoms. The Fe ions are all in +3 oxidation state, as determined from a combi-nation of charge balance considerations, inspection of bond lengths, and BVS calculations[37-39]. Each Fe atom exhibits distorted octahedral geometry, which coordin-ates to four oxygen atoms from two different tmp3- ligands, one μ6-O atom, and terminal chloride atom. It is worth notice that the charge balance is provided by the protonated nitrogen-containing heterocyclic cations. These counter cations provide not only the charge balance but also the hydrogen bond interactions to glue the counterpart of the structure. More structural details involving the packing patterns and the intracluster/intercluster distances are discussed as follows. The ellipsoid for the complexes 1~3 are shown in Fig. 2~4, respectively.
Compound 1 crystallizes in the orthorhombic system Pbca. The asymmetric unit consists of one [Fe6(μ6-O)Cl6(tmp)4]2- unit, one doubly-protonated L1 ligand, one non-coordinated methanol molecule, and two non-coordinated water molecules. The bond dista-nces of Fe-O (0.197 2~0.227 7 nm) and Fe-Cl (0.226 6 ~0.229 4 nm) are similar to those reported [Fe6] cluster containing H3thme ligand[35-36]. The intracluster Fe…Fe distances are observed in the range of 0.315 4~0.322 3 nm (Fig. S2a).
Compound 2 belongs to the trigonal system R3c. The asymmetric unit consists of half of [Fe6(μ6-O)Cl6(tmp)4]2- unit, and one mono-protonated imidazole cation. The Fe-O and Fe-Cl bond distance is in the range of 0.198 2~0.230 1 nm and 0.224 7~0.228 7 nm, respe-ctively. The intracluster Fe…Fe distances fall in the range of 0.318 5~0.321 9 nm (Fig.S2b). The [Fe6] units within the lattice of 2 connect to each other into a hydrophobic channel along c axis.
Compound 3 crystallizes in triclinic system P1. The asymmetric unit consists of one [Fe6(μ6-O)Cl6(tmp)4]2- unit, one and half of mono-protonated L2 ligands, half of mono-protonated triazole ligand, one and half of non-coordinated methanol molecules. The Fe-O and Fe-Cl bond distance is in the range of 0.198 0~0.228 2 nm and 0.227 1~0.230 6 nm, respectively. The intracluster Fe…Fe distances are within the range of 0.316 9~0.321 7 nm (Fig.S2c).
2.3 Thermal stability and PXRD analysis of 1~3
The thermogravimetric analyses (TGA) were performed to investigate the thermal stabilities of compounds 1~3 (Fig.S3). The TGA trace of compound 1 exhibits a weight loss of 5.13% from room temperature to 200 ℃ corresponding to the release of lattice water and methanol molecules (Calcd. 5.41%). Then two continuous weight loss steps are attributed to the loss of the counter-cation and the anion [Fe6] core. The remaining 37.84% above 735 ℃ corresponds to the final product of Fe2O3 (Calcd. 38.09%). The TGA profile of 2 does not lose any weight below 110 ℃ indicating there are no solvent molecules in the lattice. Then the weight loss is attributed to the gradual decomposition of the counter-cation and ligands. Finally, a plateau region is observed from 443 to 800 ℃. The final residue of 38.95% corresponds to the formation of Fe2O3 (Calcd. 39.08%). Compound 3 displays the first weight loss of approximately 3.76% from room temperature to 144 ℃ corresponding to the release of three methanol molecules (Calcd. 3.64%). And then, the release of the counter-cation and the decomposition of organic ligands occur through several continuous weight loss steps after 200 ℃. The final residuals were not characterized, which may be owing to the corrosive reactions of the final residuals with the TGA baskets made of Al2O3[40]. Viewing from the trend of the TGA curve, it is known that the decom-posing process is not achieved at the operating temperature limit (800 ℃) of our instrument.
The phase purity is confirmed by powder X-ray diffraction (PXRD) at room temperature. The experi-mental patterns of 1~3 are in fairly good agreement with the simulated ones generated from single-crystal diffraction data (Fig.S4~S6).
3 Conclusions
In summary, the use of different counter ions has led to the synthesis of a family of hexanuclear iron(Ⅲ) clusters 1~3. They have same anionic [Fe6(μ6-O)Cl6(tmp)4]2- core structures and different counter cations. The improved magnetic properties are in progress.
-
-
[1]
Milway V A, Tuna F, Farrell A R, et al. Angew. Chem. Int. Ed., 2013, 52:1949-1952 doi: 10.1002/anie.v52.7
-
[2]
Mannini M, Pineider F, Danieli C, et al. Nature, 2010, 468:417-421 doi: 10.1038/nature09478
-
[3]
Engelhardt L P, Muryn C A, Pritchard R G, et al. Angew. Chem. Int. Ed., 2008, 47:924-927 doi: 10.1002/(ISSN)1521-3773
-
[4]
Xiang S C, Hu S M, Sheng T L, et al. J. Am. Chem. Soc., 2007, 129:15144-15146 doi: 10.1021/ja0760832
-
[5]
Han S D, Song W C, Zhao J P, et al. Chem. Commun., 2013, 49:871-873 doi: 10.1039/C2CC37593A
-
[6]
赵素琴, 顾金忠.无机化学学报, 2016, 32(9):1611-1618 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?flag=1&file_no=20160916&journal_id=wjhxxbcnZHAO Su-Qin, GU Jin-Zhong. Chinese J. Inorg. Chem., 2016, 32(9):1611-1618 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?flag=1&file_no=20160916&journal_id=wjhxxbcn
-
[7]
Seki H, Hosaka Y, Saito T, et al. Angew. Chem. Int. Ed., 2016, 55:1360-1363 doi: 10.1002/anie.201509195
-
[8]
Fondo M, Doejo J, Garcia-Deibe A M, et al. CrystEngComm, 2016, 18:6673-6682 doi: 10.1039/C6CE01384H
-
[9]
Zhang Y J, Liu T, Kanegawa S, et al. J. Am. Chem. Soc., 2009, 131:7942-7943 doi: 10.1021/ja901539y
-
[10]
Ohba M, Kaneko W, Kitagawa S, et al. J. Am. Chem. Soc., 2008, 130:4475-4484 doi: 10.1021/ja7110509
-
[11]
Numata Y H, Inoue K, Baranov N, et al. J. Am. Chem. Soc., 2007, 129:9902-9909 doi: 10.1021/ja064828i
-
[12]
Zheng Y Z, Tong M L, Xue W, et al. Angew. Chem., 2007, 119:6188-6192 doi: 10.1002/(ISSN)1521-3757
-
[13]
Tian J M, Li W L, Li B, et al. Chem. Eur. J., 2013, 19:5097-5103 doi: 10.1002/chem.201204458
-
[14]
Wöhlert S, Boeckmann J, Wriedt M, et al. Angew. Chem. Int. Ed., 2011, 50:6920-6923 doi: 10.1002/anie.201007899
-
[15]
Ouellette W, Prosvirin A V, Whitenack K, et al. Angew. Chem. Int. Ed., 2009, 48:2140-2143 doi: 10.1002/anie.v48:12
-
[16]
Mahata P, Natarajan S, Panissod P, et al. J. Am. Chem. Soc., 2009, 131:10140-10150 doi: 10.1021/ja9017539
-
[17]
Ting L X, Yi W X, Xin Z W, et al. Adv. Mater., 2006, 18:2852-2856 doi: 10.1002/(ISSN)1521-4095
-
[18]
Folven E, Scholl A, Young A, et al. Nano Lett., 2012, 12:2386-2390 doi: 10.1021/nl300361e
-
[19]
Pękała M, Wolff-Fabris F, Fagnard J F, et al. J. Magn. Magn. Mater., 2013, 335:46-52 doi: 10.1016/j.jmmm.2013.01.036
-
[20]
Zhao J P, Zhao R, Yang Q, et al. Dalton Trans., 2012, 41:4852-4858 doi: 10.1039/c2dt12183b
-
[21]
Yoshida H, Yamaura J-i, Isobe M, et al. Nat. Commun., 2012, 3:860-864 doi: 10.1038/ncomms1875
-
[22]
Kenneth R P, Masaki A. Nat. Chem., 2011, 3:758-759 doi: 10.1038/nchem.1160
-
[23]
Aidoudi F H, Aldous D W, Goff R J, et al. Nat. Chem., 2011, 3:801-806 doi: 10.1038/nchem.1129
-
[24]
Lennartson A, Bond A D, Piligkos S, et al. Angew. Chem. Int. Ed., 2012, 51:11049-11052 doi: 10.1002/anie.201204207
-
[25]
Hoshino N, Iijima F, Newton G N, et al. Nat. Chem., 2012, 4:921-926 doi: 10.1038/nchem.1455
-
[26]
Miller J S. Chem. Soc. Rev., 2011, 40:3266-3296 doi: 10.1039/c0cs00166j
-
[27]
Bi Y F, Wang X T, Liao W P, et al. J. Am. Chem. Soc., 2009, 131:11650-11651 doi: 10.1021/ja9034939
-
[28]
Sanudo E C, Font-Bardia M, Solans X, et al. New J. Chem., 2011, 35:842-848 doi: 10.1039/c0nj00621a
-
[29]
Kershaw Cook L J, Kulmaczewski R, Mohammed R, et al. Angew. Chem. Int. Ed., 2016, 55:4327-4331 doi: 10.1002/anie.201600165
-
[30]
Li Z Y, Ohtsu H, Kojima T, et al. Angew. Chem. Int. Ed., 2016, 55:5184-5189 doi: 10.1002/anie.201511281
-
[31]
Brechin E K. Chem. Commun., 2005:5141-5153
-
[32]
Yang F, Li B, Xu W, et al. Inorg. Chem., 2012, 51:6813-6820 doi: 10.1021/ic300546z
-
[33]
吴小说, 汪鹏飞, 路朋朋, 等.无机化学学报, 2016, 32(9):1667-1675 http://www.cnki.com.cn/Article/CJFDTOTAL-SYQY201603027.htmWU Xiao-Shuo, WANG Peng-Fei, LU Peng -Peng, et al. Chinese J. Inorg. Chem., 2016, 32(9):1667-1675 http://www.cnki.com.cn/Article/CJFDTOTAL-SYQY201603027.htm
-
[34]
Sheldrick G M. SHELXS-97 and SHELXL-97, University of Göttingen, Germany, 1997.
-
[35]
Batchelor L J, Shaw R, Markey S J, et al. Chem. Eur. J., 2010, 16:5554-5557 doi: 10.1002/chem.201000222
-
[36]
Jiang G Q, Bai J F, Xing H, et al. Cryst. Growth Des., 2006, 6:1264-1266 doi: 10.1021/cg0600389
-
[37]
Dobson K D, McQuillan A J. Spectrochim. Acta Part A, 1999, 55:1395-1405 doi: 10.1016/S1386-1425(98)00303-5
-
[38]
Sun Z, Gantzel P K, Hendrickson D N. Inorg. Chem., 1996, 35:6640-6641 doi: 10.1021/ic9607200
-
[39]
Liu W, Thorp H H. Inorg. Chem., 1993, 32:4102-4105 doi: 10.1021/ic00071a023
-
[40]
Song J L, Yi F Y, Mao J G. Cryst. Growth Des., 2009, 9:3273-3277 doi: 10.1021/cg900010z
-
[1]
-
Table 1. Crystallographic data and structure refinement for complexes 1~3
1 2 3 Formula C30H66Cl6Fe6N2O16 C30H54Cl6Fe6N4O13 C65H128Cl12Fe12N12O29 Formula weight 1 257.8 1 225.8 2 635.6 Space group Pbca R3c P1 Crystal system Orthorhombic Trigonal Triclinic a/nm 1.960 1(7) 2.746 9(3) 1.136 1(2) b/nm 1.922 3(7) 2.746 9 1.144 3(2) c/nm 2.550 4(9) 3.546 7(4) 2.195 5(4) α/(°) 96.408(2) β/(°) 101.976(3) γ/(°) 122.667(2) V/nm3 9.610(6) 23.176(3) 2.517 8(8) Z 8 18 1 Dc/(g·cm-3) 1.731 1.576 1.738 F(000) 5 117.2 11 155.9 1 348 θ range/(°) 1.60~25.04 1.48~25.00 1.94~25.21 Unique reflns (Rint) 8 506 (0.069 5) 4 555 (0.051 2) 9 078 (0.024 3) R1a, wR2b [I>2σ(I)] 0.043 9, 0.116 8 0.044 5, 0.146 4 0.051 7, 0.117 7 R1a, wR2b (all data) 0.082 3, 0.139 1 0.060 3, 0.156 3 0.099 4, 0.148 7 GOF on F2 1.072 1.09 1.027 aR1=∑||Fo|-|Fc||/∑|Fc|; bwR2=[∑w(|Fo|-|Fc|)2/∑w(FO2)]1/2; w=1/σ(Fo)2. -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 2
- 文章访问数: 1439
- HTML全文浏览量: 79


 下载:
下载:



 下载:
下载:

