 图1
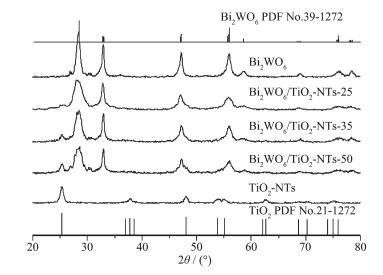
XRD patterns of the samples
Figure1.
XRD patterns of the samples
图1
XRD patterns of the samples
Figure1.
XRD patterns of the samples

Bi2WO6/TiO2纳米管异质结构复合材料的多模式下的光催化活性比较
English
Comparison of Photocatalytic Activity of Bi2WO6/TiO2 Nanotubes Heterostructures Composite under Multimode
-
Key words:
- TiO2 nanotubes
- / Bi2WO6
- / multimode degradation
- / photocatalysis
-
0 Introduction
Titanium dioxide (TiO2) has attracted a great deal of research attention because of their potential appli-cations in the photodegradation of organic pollutants, photocatalytic water splitting for hydrogen generation, dye-sensitized solar cells, and even gas sensors and biosensors, due to its low cost and abundant elements (Ti and O), long-term stability, and environmental-friendly characteristics[1-3]. However, its wide band gap and fast recombination of the photogenerated electron-hole (e--h+) are two limitations for its contemporary applications[4].
To overcome the above limitations, some measures have been taken. At present, one effective approach is to adjust TiO2 morphology. One-dimensional (1D) TiO2 nanomaterials have been receiving extensive interests[5-8]. Moreover, compared with other forms of TiO2, titania nanotubes possess the distinguishing features of nanotubes including large specific surface area, good electron/proton conductivity, and high aspect ratio. In addition, the open mesoporous morphology of TiO2 nanotubes can efficiently transfer the electrons along the 1D path without grain boundaries and junctions, while hollow space can capture scattered light to increase light harvesting as well as easier separation and recovery than TiO2 nanoparticles due to the length in the micrometer range[9].
Another strategy is to construct the heterostru-ctures by the wide band-gap semiconductor with a narrow band-gap semiconductor (with the proper band positions)[1, 10]. Yu and Li fabricated and reported Ag-based heterojunction[11-16], Au NPs loaded onto the α-Bi2O3/Bi2O2CO3[17], F-Bi2MoO6[18], anatase/rutile TiO2 particles[19], and MoS2/CdS composite[20], which are more efficient than individual component in photocatalytic properties. In particular, Qian and Ma groups successfully fabricated UCNPs/semiconductors for NIR-driven photocatalysis, such as UCNPs/TiO2 nano-fiber[21], UCNPs/TiO2/CdS nanofibers[22], NYF@TiO2-Au core@shell microspheres[23]. They show unique optical properties with wide absorption and enhanced photocatalytic abilities towards to organic dye removal efficiency under irradiation with NIR. Such synergistic interactions of heterojunction between two kinds of semiconductors are fairly powerful not only in improving the visible light harvesting ability but also in extending the lifetime of photoinduced electrons and holes via an internal charge transfer, facilitating the separation of electron-hole pairs and reducing the chance of recombination[24-26].
Among these, bismuth tungstate (Bi2WO6), as a typical Aurivillius oxide, has a layered structure with perovskite-like slabs of WO6 and [Bi2O2]2+ layer and has important physical and chemical properties such as ferroelectric piezoelectricity, catalytic behavior and nonlinear dielectric susceptibility[27-28]. More importan-tly, Bi2WO6 is a promising visible light-driven photo-catalyst with high photocatalytic activity[29-30]. However, the photocatalytic activity of pure Bi2WO6 is limited by difficult migration and high recombination proba-bility of photogenerated e--h+ pairs. Therefore, the combination of tubular morphology and heterostructure construction is a useful approach for designing hetero-structure photocatalysts with high charge separation efficiency.
In order to improve the photocatalytic activity, the construction of TiO2-Bi2WO6 heterostructures has become a hot research, and some achievement has been obtained in recent years. For instance, Wang et al. successfully synthesized TiO2-Bi2WO6 nanofibers by electrospinning technique[31]. Colón et al. and other groups have reported TiO2 modified flower[32]/sphere[33]/hollow tube-like Bi2WO6[34]. Wu and Luo et al. reported the preparation TiO2 nanobelts[35]/TiO2 nanotubes[36] grown on titanium (Ti) foil decorated with Bi2WO6 nanocrystals, respectively. These results indicate that the photocatalytic activities of TiO2-Bi2WO6 hetero-junctions show enhanced photocatalytic performance in comparison with individual components of Bi2WO6 or TiO2. To the best of our knowledge, much less notice has been taken of the preparation of TiO2 nanotubes synthesized by alkali hydrothermal treat-ment modified with Bi2WO6. Moreover, few investiga-tions were carried on the comparative mechanism of the enhanced photocatalytic activity for organic pollu-tants under multiple modes including UV, visible, and microwave-assisted photocatalysis. What is more, they lack direct evidence to explain photocatalytic mechan-ism under multiple modes that serve as background data for the environmental behavior of organic pollutants.
In this work, Bi2WO6/TiO2 nanotubes (Bi2WO6/TiO2-NTs) heterostructures were fabricated by multi-component assembly approach combined with hydro-thermal treatment, which is free from the usage of additives or surfactants. Subsequently, the photocatal-ytic activities of Bi2WO6/TiO2 nanotubes under multiple modes including UV, visible, and microwave-assisted photocatalysis were also studied in this work. Direct evidence to explain comparatively photocatalytic mechanism under multiple modes was supplied by free radical and hole trapping experiments. The relationship between the morphology, structure, optical properties and the photocatalytic activities of Bi2WO6/TiO2 heterostructures under multiple modes was investigated in detail.
1 Experimental
1.1 Preparation of Bi2WO6/TiO2 nanotubes
In a typical procedure[37], TiO2 nanotubes were dispersed in H2O (5 mL) under vigorously stirring for 0.5 h. Meanwhile, Bi(NO3)3·5H2O (0.972 g) and Na2WO4 ·2H2O (0.329 g) were dissolved in glacial acetic acid (HAc, 10 mL) and H2O (5 mL), respectively. Subse-quently, the above solutions were added into TiO2 nanotubes suspension to form a white suspension. After stirring for 2 h, the resulting mixture was suffered from hydrothermal treatment at 150 ℃ for 4 h, and the resulting precipitate was dried and washed with deionized water for three times. The obtained powder was further dried at 80 ℃ for 24 h. The final product was denoted as Bi2WO6/TiO2-NTs-x, where x represents the doping of TiO2 nanotubes (mass percentage).
1.2 Characterization of the catalyst
X-ray diffraction patterns were obtained on a Bruker-AXS (D8) X-ray diffractometer with Cu Kα radiation (λ=0.154 06 nm) at 40 kV and 40 mA in 2θ ranging from 20° to 80°. X-ray photoelectron spectros-copy (XPS) characterization was carried out on an ESCALAB 250Xi spectrometer equipped with Al Kα radiation at 300 W. N2 adsorption-desorption isotherm analyses of samples were obtained at 77 K using Micromeritics 3H-2000PS2. The morphologies of synthesized samples were analyzed using a scanning electron microscope (SEM) (HitachiS-4300) and trans-mission electron microscope (TEM) and high resolu-tion transmission electron microscope (HRTEM) (JEM-2100F). UV-Vis diffused absorption spectra (UV-Vis DRS) were recorded using a UV-Vis spectrophoto-meter (TU-1901) over the wavelength range of 200~800 nm and BaSO4 as a reference material.
1.3 Photocatalytic tests
Photocatalytic activities of the Bi2WO6/TiO2-NTs composite were studied by monitoring the degradation behaviors of rhodamine B (RhB) under multimode (including UV, visible, and microwave-assisted photo-catalysis mode). The 125 W high pressure mercury lamp (λ=313.2 nm), 400 W Xe lamp (λ=410.0 nm; moreover, the inner sleeve was made of No. 11 glass to filter out ultraviolet from the Xe lamp), and 15 W microwave electrodeless lamp (MEL, UV emission wavelength mainly located at 278 nm, U shape, 100 W output power of microwave reactor), were used as UV, visible light, and microwave-assisted photocata-lysis mode light source, respectively. The concetration of RhB was 50 mg·L-1. Moreover, the amounts of the catalyst (liquid volume) for the three modes (UV, visible, and microwave-assisted photocatalysis) were 100 mg (100 mL), 200 mg (220 mL), and 300 mg (500 mL), respectively.
The photocatalytic reaction was carried out in a quartz photoreactor. Prior to irradiation, the suspen-sion containing the solid catalyst and an aqueous solution of the contaminant was ultrasonicated for 10 min and then stirred for 1.5 h in the dark to ensure adsorption-desorption equilibrium. The reaction temp-erature was maintained at (30±2) ℃ by circulation of water through an external cooling jacket or by circulating solution to a cooler with the peristaltic pump. At certain time intervals, suspensions (5 mL) were sampled and centrifuged to remove the photo-catalyst particles. Decreases in the concentrations of RhB, methyl orange (MO), crystal violet (CV), and methylene blue (MB) were analyzed by TU-1901 UV-Vis spectrophotometer at λ=553, 464, 582, and 664 nm, respectively.
2 Results and discussion
2.1 Compositional and structural information
XRD was used to characterize the crystal structure of the as-prepared Bi2WO6/TiO2-NTs, as well as pure TiO2-NTs and Bi2WO6 (Fig. 1). The diffraction peaks of pure TiO2-NTs and Bi2WO6 are well matched with the standard patterns of anatase phase of TiO2 (PDF No.21-1272)[37] and orthorhombic phase of Bi2WO6 (PDF No.39-0256), respectively. After the coupling of Bi2WO6 and TiO2-NTs, when the TiO2-NTs loading increases from 25% to 50%, the diffraction peaks of TiO2 intensify gradually, whereas the peak intensities of Bi2WO6 decrease. No impurity peak is found in Bi2WO6/TiO2-NTs composites, suggesting that the composites exhibit a coexistence of both Bi2WO6 and TiO2 phases.
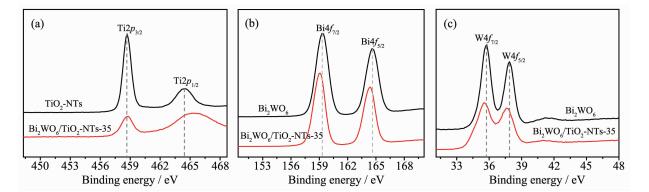
Valence states and the surface chemical comp-osition of the as-prepared samples were investigated by XPS technique. As shown in Fig. 2a, the peaks at 458.68 and 464.48 eV are attributed to Ti2p3/2 and Ti2p1/2, respectively, confirming the titanium species in the composite is Ti4+. After introduction of the Bi2WO6 into the TiO2 nanotubes, the binding energies of Ti2p3/2 and Ti2p1/2 shift to higher values (458.78 and 465.28 eV, respectively), which is attributed to diffusion of W6+ ions into the TiO2 lattice and further generation of WOTi bond linkage[35, 37]. As displayed in Fig. 2b and c, for pure Bi2WO6, the characteristic peaks at 164.58 and 159.28 eV are ascribed to Bi4f5/2 and Bi4f7/2 from Bi3+ in the lattice and the binding energy of W4f5/2 and W4f7/2 at 37.88 and 35.78 eV, respectively, are corresponded to W6+ [35]. In the XPS spectrum of Bi2WO6/TiO2-NTs, in contrast with Bi2WO6, the binding energy of Bi4f5/2 (164.38 eV) and Bi4f7/2 (159.08 eV) decreases by 0.2 eV while that of W4f5/2 (37.58 eV)and W4f7/2 (35.58 eV) decreases by 0.3 eV. The results suggest that the chemical environment surrounding Bi and W has changed, which is possibly influenced by TiO2-NTs. Thus, we can confirm that the TiO2-NTs successfully modified by Bi2WO6.
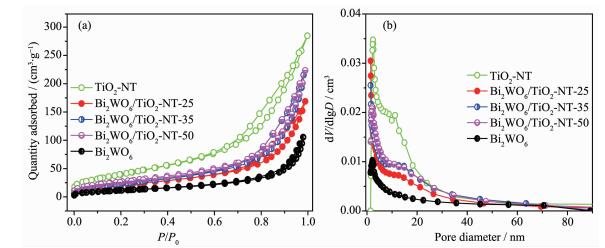
The porosity of the Bi2WO6/TiO2-NTs heterostru-ctures is investigated by N2 adsorption-desorption isotherms and the corresponding BJH pore size distri-bution. As shown in Fig. 3a, the isotherms exhibit type Ⅳ with an H3 hysteresis loop characteristic of meso-porous material[37], which is confirmed by the pore size distribution (Fig. 3b). Moreover, the formation of such mesoporous materials is attributed to the aggregation of the Bi2WO6 nanoparticles adhering to the surface of the TiO2 nanotubes. More importantly, as shown in Table 1, the measured BET surface areas of Bi2WO6/TiO2-NTs-25 (80 m2·g-1), Bi2WO6/TiO2-NTs-35 (88 m2·g-1) and Bi2WO6/TiO2-NTs-50 (101 m2·g-1) are greatly enhanced compared with that of Bi2WO6 (44 m2·g-1). Meanwhile, the specific surface areas of composite materials increase indeed together with the increase of TiO2-NTs contents from 25% to 50%.
Sample SBET/ (M2·g-1) Vp / (cm3·g-1) Dp / nm TiO2-NTs 151 0.44 8.50 Bi2WO6 44 0.16 10.44 Bi2WO6/TiO2-NTs-25 80 0.26 9.12 Bi2WO6/TiO2-NTs-35 88 0.33 10.20 Bi2WO6/TiO2-NTs-50 101 0.35 9.84 2.2 Morphology
The morphology and microstructure of the photocatalysts were also investigated. As shown in the SEM image (Fig. 4a), TiO2-NTs show the nanotubular morphology with an average diameter of 30 nm and length of 1 μm. While Bi2WO6 exhibits a typical structure of nanosheet consist of nanoparticles with the side length of 50~250 nm and thickness of 20~40 nm (Fig. 4b). As displayed in Fig. 4c~e, morphologies of TiO2 and Bi2WO6 change obviously after the combination by TiO2-NTs and Bi2WO6 through hydro-thermal treatment. The typical morphology structure of Bi2WO6/TiO2-NTs-25 consists of smooth TiO2 nano-tubes and curled Bi2WO6 flakes, which link mutually to each other. Moreover, the surface of TiO2 nanotubes becomes rough obviously after Bi2WO6 modification when TiO2 nanotubes loading increases from 35% to 50%. While Bi2WO6 changes from flakes to smaller nanoparticles. Furthermore, smaller Bi2WO6 nanoparti-cles homogeneously disperse on the surface of TiO2 nanotubes in-situ growth process. Compared with TiO2 -NTs and Bi2WO6, aggregation of Bi2WO6/TiO2-NTs has intensively alleviated with the loading of TiO2 nanotubes increasing from 0 to 50%.
In order to further confirm the Bi2WO6/TiO2-NTs heterostructures, HRTEM was used to investigate the detailed structure information of the Bi2WO6/TiO2-NTs. The corresponding HRTEM image displays two types of clear lattice fringes, as shown in Fig. 4f. The interplanar spacing of 0.35 and 0.315 nm corresponds to the (101) crystal plane of TiO2-NTs and the (131) crystal plane of the orthorhombic phase of Bi2WO6, respectively[18-19]. According to the results of XRD, XPS, SEM and HRTEM, we assume that Bi2WO6/TiO2-NTs heterostructures with Bi2WO6 nanoparticles on the surface of TiO2 nanotubes have been prepared successfully.
Based on the above results and discussion, we put forward the plausible formation of Bi2WO6/TiO2-NTs heterojunction. Considering Bi(NO3)3 with crystal water, Bi2O2(OH)NO3 is formed through the following hydrolysis and condensation reaction in the glacial acetic acid-water system (Eq.1~2). When Na2WO4 solution is added to the above reaction solution, Bi2WO6 nanoparticles are obtained (Eq.3)[38]. Then the intro-duction of TiO2-NTs into Bi2WO6 suspension, Bi2WO6 nanoparticles aggregate around TiO2-NTs. Subsequ-ently, at high temperature and high pressure, Bi2WO6 nanoparticles grow into curled flakes or smaller nanoparticles and homogeneously dispersed on the surface of TiO2 nanotubes in-situ growth process, resu-lting in the formation of Bi2WO6/TiO2-NTs heterojunc-tion[31].
2.3 Optical property
UV-Vis diffused absorption spectra (UV-Vis DRS) were carried out to investigate the optical properties of the photocatalysts. As shown in Fig. 5a, the pure TiO2-NTs and Bi2WO6 exhibit a fundamental absorption edge at around 388 and 450 nm, which originate from the charge transfer response of TiO2-NTs and Bi2WO6 from the valence band to the cond-uction band, respectively[39]. Compared with pure TiO2-NTs, the absorption edges of Bi2WO6/TiO2-NTs showed obvious red-shift to the longer wavelength within the range of visible light.
It is known that the optical absorption near the band edge of prepared samples obeys the following equation: (αhν)n=K(hν-Eg). In this equation, K, α, h, hν, Eg are constant, absorption coefficient, Planck constant, energy of the incident photon, band gap, respectively, and n is 0.5 and 1 for a direct and indirect band gap semi-conductor[38]. According to the formula, the calculated band gaps (Eg) of samples are 2.75 eV (Bi2WO6), 2.87 eV (Bi2WO6/TiO2-NTs-25), 2.94 eV (Bi2WO6/TiO2-NTs-35), 3.00 eV (Bi2WO6/TiO2-NTs-50), and 3.20 eV (TiO2-NTs), respectively.
The conduction band (CB) and valence band (VB) positions of the Bi2WO6 and TiO2 samples are estimated by the following equations: EVB=X-Ee+0.5Eg; ECB=EVB-Eg, where EVB and ECB are the VB and CB edge potentials, Ee is the energy of free electrons on the hydrogen scale (about 4.5 eV vs NHE). The X values for the Bi2WO6 and TiO2 materials are 6.21 and 5.81 eV, respectively[40-41]. The Eg of Bi2WO6 and TiO2-NTs are estimated to be 2.75 and 3.20 eV, respectively. Herein, the CB and VB edge potentials of Bi2WO6 and TiO2-NTs are calculated at 0.34 and 3.09 eV, and -0.29 and 2.91 eV, respectively.
2.4 Photocatalytic activity
The photocatalytic performance of the Bi2WO6/TiO2-NTs heterostructures in terms of photodegrada-tion of RhB molecules under multiple modes inclu-ding UV, visible, and microwave-assisted photocataly-sis was investigated.
Fig. 6a shows the photocatalytic activities of photocatalysts. Under UV light irradiation alone (without catalyst), only 3% RhB is degraded, which means the RhB can remain stability under long time irradiation. However, apparent changes in the concen-tration of RhB are observed in the existence of both light and catalyst. After irradiation for 90 min, 46.8%, 61.5%, 70.0%, 88.9%, 82.4% and 74.1% of the RhB is degraded by using the TiO2-NTs, Bi2WO6, Bi2WO6/TiO2-NTs-25, Bi2WO6/TiO2-NTs-35, Bi2WO6/TiO2-NTs-50, and P25, respectively.
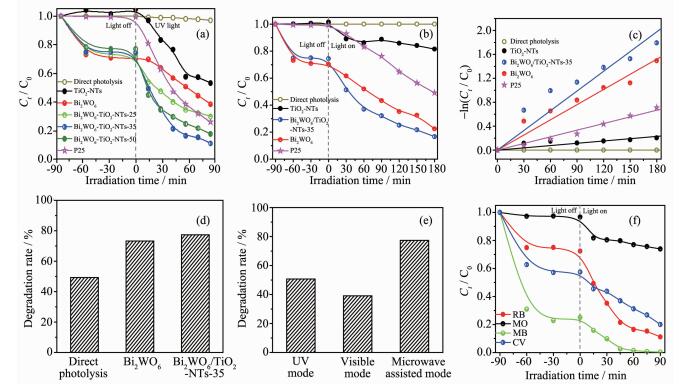 图6
Normalized decrease concentration of Ct /C0 of RhB solution containing different photocatalysts under UV (a) and visible (b) light irradiation; (c) -ln(Ct /C0) as a function of irradiation time for RhB degradation over photocatalysts; (d) Photocatalytic degradation RhB profiles obtained using different photocatalysts under microwave-assisted photocatalysis mode for 15 min; (e) Photocatalytic degradation RhB profiles by Bi2WO6/TiO2-NTs-35 obtained under multimode for 15 min; (f) Normalized decrease concentrations of Ct /C0 of different dyes using Bi2WO6/TiO2-NTs-35 under UV light irradiation
Figure6.
Normalized decrease concentration of Ct /C0 of RhB solution containing different photocatalysts under UV (a) and visible (b) light irradiation; (c) -ln(Ct /C0) as a function of irradiation time for RhB degradation over photocatalysts; (d) Photocatalytic degradation RhB profiles obtained using different photocatalysts under microwave-assisted photocatalysis mode for 15 min; (e) Photocatalytic degradation RhB profiles by Bi2WO6/TiO2-NTs-35 obtained under multimode for 15 min; (f) Normalized decrease concentrations of Ct /C0 of different dyes using Bi2WO6/TiO2-NTs-35 under UV light irradiation
图6
Normalized decrease concentration of Ct /C0 of RhB solution containing different photocatalysts under UV (a) and visible (b) light irradiation; (c) -ln(Ct /C0) as a function of irradiation time for RhB degradation over photocatalysts; (d) Photocatalytic degradation RhB profiles obtained using different photocatalysts under microwave-assisted photocatalysis mode for 15 min; (e) Photocatalytic degradation RhB profiles by Bi2WO6/TiO2-NTs-35 obtained under multimode for 15 min; (f) Normalized decrease concentrations of Ct /C0 of different dyes using Bi2WO6/TiO2-NTs-35 under UV light irradiation
Figure6.
Normalized decrease concentration of Ct /C0 of RhB solution containing different photocatalysts under UV (a) and visible (b) light irradiation; (c) -ln(Ct /C0) as a function of irradiation time for RhB degradation over photocatalysts; (d) Photocatalytic degradation RhB profiles obtained using different photocatalysts under microwave-assisted photocatalysis mode for 15 min; (e) Photocatalytic degradation RhB profiles by Bi2WO6/TiO2-NTs-35 obtained under multimode for 15 min; (f) Normalized decrease concentrations of Ct /C0 of different dyes using Bi2WO6/TiO2-NTs-35 under UV light irradiation
Fig. 6b displays the photocatalytic activity of prepared samples under the visible light irradiation. It is found that the photocatalytic performance of Bi2WO6/TiO2-NTs-35 to degrade RhB under visible light irradiation surpasses that of its individual counterparts.
At the same time, the kinetics of photocatalytic degradation of RhB is investigated by simplified Langmuir-Hinshelwood model. The pseudo-first-order rate constant (kapp) is calculated using the formula -ln(Ct/C0)=kappt, where C0 and Ct are the initial conce-ntration and concentration at reaction time t of RhB, respectively. From Fig. 6c, under visible irradiation, the rate constant over Bi2WO6/TiO2-NTs-35, Bi2WO6, P25, and TiO2-NTs is 1.10×10-2, 8.45×10-3, 3.71×10-3, and 1.27×10-3 min-1, respectively. Moreover, Bi2WO6/TiO2-NTs-35 shows the highest first-order rate constant, which is about 1.2 and 8.7 times greater than that of pure Bi2WO6 and TiO2-NTs, respectively.
Fig. 6d also exhibits the photocatalytic activity of different photocatalysts under microwave-assisted photocatalysis mode with electrodeless discharge lamp activated by microwaves as the light source. Bi2WO6/TiO2-NTs-35 shows highest photocatalytic activity towards RhB degradation under microwave-assisted photocatalysis mode. Moreover, Fig. 6e displays photo-catalytic activities of Bi2WO6/TiO2-NTs-35 under diff-erent modes after irradiation for 15 min. In contrast with UV and visible mode, the Bi2WO6/TiO2-NTs-35 shows higher activity under microwave-assisted photo-catalytic mode. In addition, different kinds of dyes were selected to evaluate the photocatalytic activity under UV light irradiation (Fig. 6f). The cationic dyes (CV, MB, and RhB) are effectively degraded, while the degradation of anionic dye (MO) is poor, which is attributed to the different structure and adsorption of dyes.
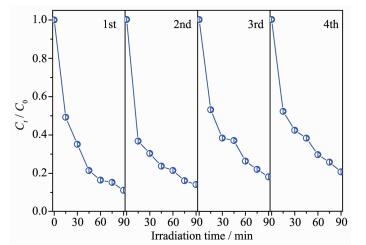
To evaluate the stability and reusability of Bi2WO6/TiO2-NTs-35 heterostructures for practical application, the photocatalytic degradation of RhB with the same photocatalyst is carried out for several times. As displayed in Fig. 7, degradation curve has no obvious decline after four cycles of RhB degradation reaction under UV light irradiation, which indicates Bi2WO6/TiO2-NTs-35 heterostructures maintain high stability.
2.5 Possible pathway of RhB degradation in Bi2WO6/TiO2-NTs system
The above photocatalytic tests indicate that: (ⅰ) the photocatalytic activity of pure TiO2-NTs can be further increased by introduction proper Bi2WO6 loading under multimode; (ⅱ) in contrast to UV and visible mode, Bi2WO6/TiO2-NTs showed higher photo-catalytic activity under microwave-assisted photo-catalysis mode. The influence factors towards the excellent photocatalytic activity of Bi2WO6/TiO2-NTs are discussed.
Firstly, Bi2WO6 modified TiO2 nanotubes play a major role in improving the photocatalytic activity of TiO2 nanotubes. On one hand, according to UV-Vis DRS analysis, Bi2WO6/TiO2-NTs heterostructures have a narrow band gap and exhibit enhanced UV and visible light absorption, consequently increases the utilization of light. On the other hand, the formed heterostructures between Bi2WO6 and TiO2-NTs photocatalysts can extend the lifetime of photoinduced electrons and holes via an internal charge transfer, further facilitate the separation of e--h+ pairs and reduce the chance of recombination. These well-separated electrons and holes can participate in the overall photocatalysis process.
Secondly, the open mesoporous morphology of nanotubes can enhance the contact between the substance and photocatalysts during the photocatalytic reaction. Meanwhile, the nanotubes provide an efficient transport channel for photogenerated electrons.
Thirdly, degradation mode influences the photo-catalytic activity of Bi2WO6/TiO2-NTs heterostructures. Compared with UV and visible mode, Bi2WO6/TiO2-NTs heterostructures display highest photocatalytic activity under microwave-assisted photocatalysis mode. Microwave enhances the reactants mobility and diffus-ion leading to increased exchange of reactants between catalyst surface and solution[42]. Moreover, more ·OH and ·O2- radicals are generated by photocatalysis with microwave irradiation than photocatalysis alone to enhance the separation of e--h+ pairs[43-45], which will be confirmed by the following trapping experiments.
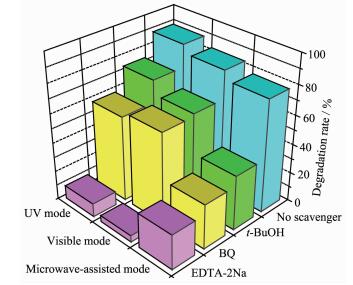
As shown in Fig. 8, the RhB degradation rate under UV degraded mode decreases obviously with the addition of disodium ethylenediaminetetraacetate (EDTA-2Na, 1 mmol·L-1) as scavenger for h+ (from 88.9% to 9.3%), is moderately reduced with the addi-tion of benzoquinone (BQ, 1 mmol·L-1) as scavenger for ·O2- (from 88.9% to 58.6%) and tert-butyl alcohol (t-BuOH, 1 mmol·L-1) as scavenger for ·OH (from 88.9% to 73.7%)[46-48]. Similar results are found in RhB photodegradation over Bi2WO6/TiO2-NTs-35 under visible mode. Compared with UV and visible mode, under microwave-assisted mode, there is a little difference. The degradation rate toward RhB exhibits a significant decrease when EDTA-2Na, BQ, and t-BuOH are introduced. Furthermore, RhB degradation rate is reduced from the original 77.4% to 23.9%, 30.7%, and 37.5%, respectively. These results suggest that: (ⅰ) under the three modes, the degradation of RhB is primarily driven by h+, ·OH, and ·O2-; (ⅱ) under the UV and visible mode, h+ is the dominant reactive oxidants; (ⅲ) under microwave-assisted mode, h+, ·OH and ·O2- make nearly equal contribution to RhB degradation. That is to say, more ·OH and ·O2- are generated under microwave-assisted photocatalysis mode compared with UV and visible mode.
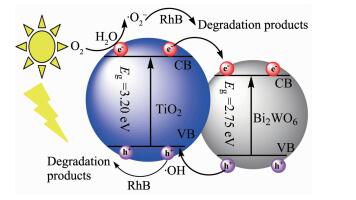
Based on the above results, the photocatalytic mechanism for Bi2WO6/TiO2-NTs heterostructures photocatalyst is tentatively proposed and schematically illustrated in Scheme 1. The conduction bands (CB) (the valence band (VB), band gap) of TiO2-NTs and Bi2WO6 are at -0.29 and 0.34 eV, respectively. Hence, under UV or MEL irradiation, both TiO2-NTs and Bi2WO6 are excited, and photogenerated electrons and holes are in their CB and VB, respectively. Subsequently, the photoexcited electrons in the CB of TiO2-NTs transfer to the CB of Bi2WO6, which is due to that ECB of TiO2-NTs is more negative than that of Bi2WO6. Simultaneously, the holes in the EVB of Bi2WO6 move to TiO2-NTs due to that the EVB of Bi2WO6 is more positive than that of TiO2-NTs. The hVB+ reacts with the absorbed H2O molecules or deoxidizes dioxygen dissolved in the aqueous solution to form ·OH radicals. In addition, the ECB can be easily oxidized by dioxygen to produce ·O2- radicals. With the help of ·OH, hVB+ and ·O2- species, RhB is degraded and then mineralized. The photocatalytic process under visible light irradiation is similar with UV (microwave-assisted photocatalytic mode) except TiO2-NTs are not excited.
3 Conclusions
In summary, Bi2WO6/TiO2-NTs heterostructures were fabricated by multicomponent assembly approach combined with hydrothermal treatment. Bi2WO6 flakes or nanoparticles dispersed on the surface of TiO2 nanotubes to form heterostructures. The prepared Bi2WO6/TiO2-NTs heterostructures exhibit considerably high photocatalytic activity towards the degradation of RhB under multimode including UV, visible and microwave-assisted photocatalysis. This enhanced photocatalytic activity is due to more efficient separa-tion of the e--h+ pairs, originating from the introduc-tion of Bi2WO6 modified TiO2-NTs, the nanotubular geometries, and the degradation mode. The h+, ·OH, and ·O2- radicals are the main active species during the photocatalysis process under multimode. Moreover, more ·OH and ·O2- radicals are generated by photo-catalyst with microwave-assisted irradiation. This work can provide important inspirations in developing the photocatalytic heterostructures materials.
Acknowledgments: This work is supported by the Natural Science Foundation of China (Grants No.21376126, 81403067), the Program for Young Teachers Scientific Research in Qiqihar University (Grant No.2014K-M03), and the Basic Business Special Scientific Research of Heilongjiang Province Education Department (Grant No.135109204).
-
-
[1]
Tian J, Zhao Z H, Kumar A, et al. Chem. Soc. Rev., 2014, 43:6920-6937 doi: 10.1039/C4CS00180J
-
[2]
Daghrir R, Drogui P, Robert D. Ind. Eng. Chem. Res., 2013, 52:3581-3599 doi: 10.1021/ie303468t
-
[3]
Sang L X, Zhao Y X, Burda C. Chem. Rev., 2014, 114:9283-9318 doi: 10.1021/cr400629p
-
[4]
张超颖, 王苹, 刘岩岩, 等.无机化学学报, 2017, 33(7):1132-1138 doi: 10.11862/CJIC.2017.170ZHANG Chao-Ying, WANG Ping, LIU Yan-Yan, et al. Chinese J. Inorg. Chem., 2017, 33(7):1132-1138 doi: 10.11862/CJIC.2017.170
-
[5]
Murciano L T, Lapkin A A, Chadwick D. J. Mater. Chem., 2010, 20:6484-6489 doi: 10.1039/c0jm01212b
-
[6]
Zhang Y L, Han C, Zhang G S, et al. Chem. Eng. J., 2015, 268:170-179 doi: 10.1016/j.cej.2015.01.006
-
[7]
Lee K, Mazare A, Schmuki P. Chem. Rev., 2014, 114:9385-9454 doi: 10.1021/cr500061m
-
[8]
Wang X D, Li Z D, Shi J, et al. Chem. Rev., 2014, 114:9346-9384 doi: 10.1021/cr400633s
-
[9]
Wehrenfennig C, Palumbiny C M, Snaith H J, et al. J. Phys. Chem. C, 2015, 119:9159-9168 doi: 10.1021/acs.jpcc.5b01827
-
[10]
Wang H L, Zhang L S, Chen Z G, et al. Chem. Soc. Rev., 2014, 43:5234-5244 doi: 10.1039/C4CS00126E
-
[11]
Li J, Fang W, Yu C L, et al. Appl. Surf. Sci., 2015, 358:46-56 doi: 10.1016/j.apsusc.2015.07.139
-
[12]
Yu C L, Zhou W Q, Yu J C, et al. Chin. J. Catal., 2014, 35:1609-1618 doi: 10.1016/S1872-2067(14)60170-4
-
[13]
Yu C L, Wei L F, Chen J C, et al. Ind. Eng. Chem. Res., 2014, 53:5759-5766 doi: 10.1021/ie404283d
-
[14]
Yu C L, Li G, Kumar S, et al. Adv. Mater., 2014, 26:892-898 doi: 10.1002/adma.v26.6
-
[15]
Yu C L, Wei L F, Zhou W Q, et al. Chemosphere, 2016, 157:250-261 doi: 10.1016/j.chemosphere.2016.05.021
-
[16]
Yu C L, Zhou W Q, Liu H, et al. Chem. Eng. J., 2016, 287:117-129 doi: 10.1016/j.cej.2015.10.112
-
[17]
Yu C L, Zhou W Q, Zhu L H, et al. Appl. Catal. B, 2016, 184:1-11 doi: 10.1016/j.apcatb.2015.11.026
-
[18]
Yu C L, Wu Z, Liu R Y, et al. Appl. Catal. B, 2017, 209:1-11 doi: 10.1016/j.apcatb.2017.02.057
-
[19]
Zhang J, Xu Q, Feng Z C, et al. Angew. Chem. Int. Ed., 2008, 47:1766-1769 doi: 10.1002/(ISSN)1521-3773
-
[20]
Zong X, Yan H J, Wu G P, et al. J. Am. Chem. Soc., 2008, 130:7176-7177 doi: 10.1021/ja8007825
-
[21]
Zhang F, Zhang C L, Peng H Y, et al. Part. Part. Syst. Char., 2016, 33:248-253 doi: 10.1002/ppsc.v33.5
-
[22]
Zhang F, Zhang C L, Wang W N, et al. ChemSusChem, 2016, 9:1449-1454 doi: 10.1002/cssc.v9.12
-
[23]
Xu Z H, Quintanilla M, Vetrone F, et al. Adv. Funct. Mater., 2015, 25:2950-2960 doi: 10.1002/adfm.201500810
-
[24]
Zhou F Q, Fan J C, Xu Q J, et al. Appl. Catal. B, 2017, 201:77-83 doi: 10.1016/j.apcatb.2016.08.027
-
[25]
Zhang F, Wang W N, Cong H P, et al. Part. Part. Syst. Char., 2017, 34(2):1600222(6 pages) doi: 10.1002/ppsc.v34.2
-
[26]
Min Y L, He G Q, Xu Q J, et al. J. Mater. Chem. A, 2014, 2:2578-2584 doi: 10.1039/c3ta14240j
-
[27]
Zhang L S, Wang H L, Chen Z G, et al. Appl. Catal. B, 2011, 106:1-13
-
[28]
张田, 邹正光, 何金云, 等.无机化学学报, 2017, 33(6):954-962 doi: 10.11862/CJIC.2017.093ZHANG Tian, ZOU Zheng-Guang, HE Jin-Yun, et al. Chinese J. Inorg. Chem., 2017, 33(6):954-962 doi: 10.11862/CJIC.2017.093
-
[29]
Huang J, Tan G Q, Ren H J, et al. ACS Appl. Mater. Interfaces, 2014, 6:21041-21050 doi: 10.1021/am505817h
-
[30]
Zhang L W, Zhu Y F. Catal. Sci. Technol., 2012, 2:694-706 doi: 10.1039/c2cy00411a
-
[31]
Zhang Y P, Fei L F, Jiang X D, et al. J. Am. Ceram. Soc., 2011, 94:4157-4161 doi: 10.1111/j.1551-2916.2011.04905.x
-
[32]
López S M, Hidalgo M C, Navío J A, et al. J. Hazard. Mater., 2011, 185:1425-1434 doi: 10.1016/j.jhazmat.2010.10.065
-
[33]
Liu Z, Liu X Z, Lu D Z, et al. Mater. Lett., 2014, 130:143-145 doi: 10.1016/j.matlet.2014.05.097
-
[34]
Hou Y F, Liu S J, Zhang J H, et al. Dalton Trans., 2014, 43:1025-1031 doi: 10.1039/C3DT52046C
-
[35]
Li Y, Wu W J, Wu M Z, et al. Mater. Res. Bull., 2014, 55:121-125 doi: 10.1016/j.materresbull.2014.04.001
-
[36]
Deng F, Liu Y, Luo X B, et al. Sep. Purif. Technol., 2013, 120:156-161 doi: 10.1016/j.seppur.2013.09.032
-
[37]
Ma F Y, Geng Z, Yang X, et al. RSC Adv., 2015, 5:46677-46685 doi: 10.1039/C5RA05404D
-
[38]
Chen S F, Tang W M, Hu Y F, et al. CrystEngComm, 2013, 15:7943-7950 doi: 10.1039/c3ce41036f
-
[39]
Di J, Xia J X, Ge Y P, et al. Appl. Catal. B, 2015, 168-169:51-61
-
[40]
Li L, Huang X D, Hu T Y, et al. New J. Chem., 2014, 38:5293-5302 doi: 10.1039/C4NJ01002G
-
[41]
Dai K, Lu L H, Liang C H, et al. Appl. Catal. B, 2014, 156-157:331-340 doi: 10.1016/j.apcatb.2014.03.039
-
[42]
Zhang X W, Li G T, Wang Y Z. Dyes Pigm., 2007, 74:536-544 doi: 10.1016/j.dyepig.2006.03.012
-
[43]
Genuino H C, Hamal D B, Fu Y J, et al. J. Phys. Chem. C, 2012, 116:14040-14051
-
[44]
Zhang Z H, Yu F Y, Huang L R, et al. J. Hazard. Mater., 2014, 278:152-157 doi: 10.1016/j.jhazmat.2014.05.064
-
[45]
Zhang X W, Sun D D, Li G T, et al. J. Photochem. Photobiol. A, 2008, 199:311-315 doi: 10.1016/j.jphotochem.2008.06.009
-
[46]
Xiao J D, Xie Y B, Cao H B, et al. Catal. Commun., 2015, 66:10-14 doi: 10.1016/j.catcom.2015.03.004
-
[47]
Lin S L, Liu L, Hu J S, et al. Appl. Surf. Sci., 2015, 324:20-29 doi: 10.1016/j.apsusc.2014.10.101
-
[48]
Ma F Y, Shi T, Gao J, et al. Colloids Surf. A, 2012, 401:116-125 doi: 10.1016/j.colsurfa.2012.03.036
-
[1]
-
Figure 6 Normalized decrease concentration of Ct /C0 of RhB solution containing different photocatalysts under UV (a) and visible (b) light irradiation; (c) -ln(Ct /C0) as a function of irradiation time for RhB degradation over photocatalysts; (d) Photocatalytic degradation RhB profiles obtained using different photocatalysts under microwave-assisted photocatalysis mode for 15 min; (e) Photocatalytic degradation RhB profiles by Bi2WO6/TiO2-NTs-35 obtained under multimode for 15 min; (f) Normalized decrease concentrations of Ct /C0 of different dyes using Bi2WO6/TiO2-NTs-35 under UV light irradiation
Table 1. Textural parameters of various TiO2-based materials
Sample SBET/ (M2·g-1) Vp / (cm3·g-1) Dp / nm TiO2-NTs 151 0.44 8.50 Bi2WO6 44 0.16 10.44 Bi2WO6/TiO2-NTs-25 80 0.26 9.12 Bi2WO6/TiO2-NTs-35 88 0.33 10.20 Bi2WO6/TiO2-NTs-50 101 0.35 9.84 -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 8
- 文章访问数: 1765
- HTML全文浏览量: 198




 下载:
下载:








 下载:
下载:

