
邻异丙硫基苯甲酰基和苯硫甲基铁衍生物的合成与反应
English
Synthesis and Reactivity of ortho-Isopropylthiobenzoyl and Phenylthiomethyl Iron Derivatives
-
Key words:
- thioether
- / iron carbonyl
- / metal-acyl complex
- / pyridine
- / phosphine ligand
-
The investigation on the metal-acyl complexes continues to be an active research area in organometa-llic chemistry since these complexes show excellent catalytic activity in many organic transformations[1-4], and this category of complexes are proposed as the key active intermediates in numerous catalytic processes[5-8]. Recent studies have proven the practicability that the metal-acyl moiety was introduced into the main chain of polymers to form potential functional organometallic materials[9], and the organometallic aqueous colloids were formed through the self-assembly of the metal-acyl complexes[10], which markedly broaden their application fields. Among the metal-acyl complexes, the acyl iron complexes have drawn more and more attention in recent years because of the successful elucidation of the structure of [Fe]-hydrogenase[11-13], which suggests that an acyl-iron ligates to the active site of [Fe]-hydrogenase[14]. Since then a number of acyl iron complexes have been synthesized and character-ized[15-20], which promotes the rapid development of acyl iron complexes. To match the electronic and steric properties of biomimetic models for the active site of [Fe]-hydrogenase, several kinds of donor-functionalized acyl ligands, such as acylphosphine[21], carbamoylpyri-dine[19], acylmethylpyridine[16], and acylmethylpyra-zole[22-23], have been used to synthesize acyl iron comp-lexes. Because of the introduction of the chelation-assisted donor atoms bearing different electron-donating ability, the corresponding acyl iron complexes exhibit versatile reactivity. In addition, the incorporation of the sulfur-containing ligand to the metal center of model complexes for [Fe]-hydrogenase is also important, since the sulfur atom possibly plays a role in the catalytic cycle of hydrogen activation by [Fe]-hydro-genase[24-25]. However, iron complexes with sulfur-functionalized acyl ligand[26-27] are rare in known synth-etic model complexes of [Fe]-hydrogenase, in which phenylthiolate or 2-pyridinethiolate ligand is usually used to occupy the cysteine sulfur position. Other structural models with sulfur-functionalized acyl ligand should be conducive to gaining a deeper understanding of the structure and catalytic function of [Fe]-hydrogenase owing to the strong influence of the stereo-electronic environment of the iron center on the ability of the biomimic to bind hydrogen molecule. Our previous investigations showed that the different chelation-assisted donor atoms significantly affected the structure and reactivity of the corresponding acyl iron complexes[22-23]. As an extension of our investiga-tions on this subject, herein we describe the synthesis and reactivity of thioether-based iron complexes.
1 Experimental
Solvents were dried and freshly distilled prior to use according to standard procedures. All reactions were carried out under an atmosphere of argon. NMR spectra were recorded on a Bruker 400 spectrometer using CDCl3 as solvent unless otherwise noted, and the chemical shifts were reported with respect to the reference (internal SiMe4 for 1H and 13C NMR spectra, external 85% H3PO4 aqueous solution for 31P NMR spectra). IR spectra were recorded as KBr pellets on a Tensor 27 spectrometer. Elemental analyses were carried out on an Elementar Vario EL analyzer.
1.1 Synthesis of [(o-iPrS)C6H4COFe(CO)2I]2 (1)
A hexane solution of n-BuLi (1.6 mol·L-1, 1.3 mL, 2 mmol) was added to the solution of N, N, N′, N′-tetramethylethylenediamine (TMEDA) (0.2 mL, 2 mmol) in hexane (10 mL) at room temperature. After 30 min, isopropylthiobenzene (0.32 mL, 2 mmol) was added to the above-mentioned solution. The resulting mixture was stirred for 4 h at room temperature, during which time a solid was precipitated out. Then, the reaction mixture was cooled to -78 ℃, and THF (20 mL) was added, After the solid was completely dissolved, Fe(CO)5 (0.26 mL, 2 mmol) was added. The reaction mixture was continuously stirred at -78 ℃ for 30 min, and allowed to reach room temperature slowly and stirred for 2 h. A solution of I2 (0.51 g, 2 mmol) in THF (10 mL) was added dropwise. After completion of addition, the reaction mixture was stirred overnight. The solvent was removed under reduced pressure, and the residue was purified by column chromatography on silica with CH2Cl2 as the eluent. The red eluate was concentrated to dryness to give 1 as a red solid. Yield: 0.33 g (38%). 1H NMR: δ 1.43 (d, J=5.3 Hz, 6H, CH3), 1.49 (d, J=5.0 Hz, 6H, CH3), 3.20~3.35 (m, 2H, CH), 7.55~7.65 (m, 4H, C6H4), 7.74~7.80 (m, 4H, C6H4). 13C NMR: δ 22.1 (CH3), 22.4 (CH3), 48.7 (CH), 125.2, 130.8, 131.7, 132.1, 136.4, 150.5 (C6H4), 195.8, 205.0 (C≡O), 246.9 (C=O). IR(cm-1): ν(C≡O) 2 090, 2 030, 2 004, 1 975; ν(C=O) 1 628. Anal. Calcd. for C24H22Fe2I2O6S2(%): C 34.48, H 2.65; Found(%): C 34.35, H 3.10.
1.2 Synthesis of (o-iPrS)C6H4COFe(CO)2(SPy) (2)
Sodium hydride (12 mg, 0.5 mmol) was added to a solution of 2-mercaptopyridine (56 mg, 0.5 mmol) in CH2Cl2 (20 mL). The reaction mixture was stirred for 1 h at room temperature, followed by the addition of a solution of 1 (0.21 g, 0.25 mmol) in CH2Cl2 (10 mL). The reaction mixture was continuously stirred for 6 h. The solvent was removed under reduced pressure, and the residue was purified by column chromatography on silica with CH2Cl2 as the eluent. The red eluate was concentrated to dryness to afford 0.16 g (72%) of 2. This complex consisted of two isomers (2A/2B) in solution. The relative ratio of isomers was ca. 1: 1 in CDCl3 and 2: 1 in acetone-d6, respectively, according to the integral of the corresponding protons of the methine and methyl groups. 2A: 1H NMR: δ 1.38 (d, J=6.6 Hz, CH3), 1.49 (d, J=6.6 Hz, CH3), 3.35~3.46 (m, CH), 3.50~3.66 (m, CH), 6.55~6.61 (m), 6.82~6.90 (m), 7.00~7.16 (m), 7.29 ~7.35 (m), 7.36~7.41 (m), 7.61~7.70 (m), 7.88~7.95 (m) (phenyl and pyridyl protons). 13C NMR: δ 21.5, 22.0, 22.1, 22.6 (CH3), 46.3, 46.9 (CH), 117.5, 117.9, 122.2, 122.5, 125.4, 127.4, 130.3, 130.5, 130.8, 131.4, 131.7, 131.8, 135.5, 136.1, 138.1, 138.6, 148.5, 148.7, 149.7, 151.0, 177.3, 180.8 (phenyl and pyridyl carbons), 208.8, 210.2, 210.7, 211.6 (C≡O), 260.3, 262.2 (C=O). 2B: 1H NMR (acetone-d6): δ 1.39 (d, J=6.8 Hz), 1.41 (d, J=6.7 Hz), 1.53 (d, J=6.7 Hz), 3.55~3.65 (m), 3.68~3.83 (m), 6.77 (t, J=6.2 Hz), 6.89 (dd, J1=2.8 Hz, J2=8.3 Hz), 7.00~7.03 (m), 7.09~7.24 (m), 7.42 (dd, J1=1.0 Hz, J2=7.6 Hz), 7.89 (t, J=8.1 Hz), 8.20 (d, J=5.3 Hz). The other signals in the range of 7.48~7.70 overlapped seriously. IR(cm-1): ν(C≡O) 2 023, 1 967, 1 957; ν(C=O) 1 615. Anal. Calcd. for C17H15FeNO3S2(%): C 50.88, H 3.77, N 3.49; Found(%): C 50.66, H 4.23, N 3.46.
1.3 Synthesis of C6H5SCH2Fe(CO)3I (3)
This complex was similarly obtained using thioanisole instead of isopropylthiobenzene as above-mentioned synthesis of 1. Yield: 39%. 1H NMR: δ 3.95 ~4.00 (m, 2H, CH2), 7.39 (s, br, 3H, C6H5), 7.53 (s, br, 2H, C6H5). 13C NMR: δ 38.7 (CH2), 129.8, 130.0, 130.1, 135.8 (C6H5), 203.8, 209.4, 210.2 (C≡O). IR: ν(C≡O) 2 077, 2 019, 1 996 cm-1. Anal. Calcd. for C10H7FeIO3S(%): C 30.80, H 1.81; Found(%): C 30.92, H 2.14.
1.4 Synthesis of C6H5SCH2Fe(CO)2(PPh3)I (4)
PPh3 (26 mg, 0.1 mmol) was added to a solution of 3 (39 mg, 0.1 mmol) in THF (20 mL). The reaction mixture was stirred overnight at room temperature. The solvent was removed under reduced pressure, and the residue was purified by column chromatography on silica with CH2Cl2/hexane (1: 2, V/V) as the eluent to give a red eluate, which was concentrated to dryness to afford 20 mg (32%) of 4 as a red solid. 1H NMR: δ 3.92~4.11 (m, 2H, CH2), 7.03~7.60 (m, 20H, C6H5). 13C NMR: δ 36.0 (d, JP-C=20.2 Hz, CH2), 128.4 (d, JP-C=9.5 Hz), 128.1, 129.0, 129.3, 130.2, 133.6, 133.9 (d, JP-C =10.1 Hz), 135.0, 135.4, 136.0 (C6H5), 214.9 (d, JP-C=17.5 Hz), 218.1 (C≡O). 31P NMR: δ 40.1. IR: ν(C≡O) 2 004, 1 950 cm-1. Anal. Calcd. for C27H22FeIO2PS(%): C 51.95, H 3.55; Found(%): C 51.80, H 3.89.
1.5 Reaction of 1 with PPh3
Reaction of 1 with PPh3, like that of 3 with PPh3, resulted in the decomposition of the starting material. No characterizable product was obtained.
1.6 Crystal structure determination
Crystals of 1, 2A, 3 and 4 suitable for X-ray analyses were obtained by slow diffusion of hexane into their CH2Cl2 solutions at -18 ℃. All intensity data were collected on a Rigaku Saturn CCD detector using Mo Kα radiation (λ=0.071 073 nm). Semi-empirical absorption corrections were applied using the Crystal-clear program[28]. The structures were solved by direct methods and difference Fourier map using SHELXS of the SHELXTL package and refined with SHELXL[29] by full-matrix least-squares on F2. All non-hydrogen atoms were refined anisotropically. Hydrogen atoms were added geometrically and refined with riding model position parameters. A summary of the fundamental crystal data for 1, 2A, 3 and 4 is listed in Table 1.
Complex 1 2A 3 4 Formula C24H22Fe2l2O6S2 C17H15FeNO3S2 C10H7FeIO3S C27H22FeIO2PS Formula weight 836.03 401.27 389.97 624.23 Crystal size/mm 0.20×0.18×0.12 0.20×0.18×0.12 0.24×0.22×0.16 0.20×0.18×0.12 Crystal system Monoclinic Monoclinic Monoclinic Monoclinic Space group C2/c P21/n P21/n P21/c a/nm 2.267 3(5) 1.166 2(2) 1.229 12(13) 1.273 57(18) b/nm 0.961 08(19) 1.250 4(3) 0.710 29(7) 1.049 13(15) c/nm 1.649 6(3) 1.188 6(2) 1.433 60(14) 1.871 3(2) β/(°) 127.86(3) 95.379(2) 97.984(2) 92.063(3) T/K 113(2) 113(2) 113(2) 113(2) V/nm3 2.837 8(13) 1.725 5(6) 1.239 4(2) 2.498 7(6) Z 4 4 4 4 Dc/(g·cm-3) 1.957 1.545 2.090 1.659 F(000) 1 616 824 744 1 240 μ/mm-1 3.381 1.130 3.863 2.009 θ range/(°) 2.40~25.02 2.34~36.37 2.05~28.71 3.20~27.52 Measured reflection 14 131 30 684 16 369 31 228 Unique reflection (Rint) 2 513 (0.076 8) 8 160 (0.019 9) 3 210 (0.019 9) 5 749 (0.046 5) Observed reflection with [I > 2σ(I)] 2 004 7 136 3 082 5 039 Parameter 165 219 145 298 GOF 1.219 1.014 1.056 0.998 Residuals R, wR2 0.076 9, 0.160 5 0.024 7, 0.063 5 0.011 5, 0.028 1 0.023 7, 0.060 0 CCDC: 1545834, 1; 1545835, 2A; 1545836, 3; 1545837, 4.
2 Results and discussion
2.1 Synthesis and reactivity of ortho-isopropyl-thiobenzoyl iron complex
It is known that isopropylthiobenzene is deproto-nated with n-BuLi in the presence of TMEDA to give the ortho-lithiation product[30]. Herein, we find that reaction of this ortho-lithiation product with iron carbonyl followed by iodine yields a dimeric ortho-isopropylthiobenzoyl iron complex 1 (Scheme 1). This result is significantly different from the similar reac-tions of (pyrazol-1-yl)methyllithium[23] and (2-pyridyl)methyllithium[31], which only give mononuclear acyl iron complexes, revealing that different chelation-assisted donors strongly affect the fundamental structures of metal-acyl complexes. The substitution of iodide in 1 by 2-pyridinethiolate is successful, which gives mono-nuclear complex 2. However, reaction of 1 with PPh3 leads to the decomposition of the starting material. Complexes 1 and 2 are air-stable solids and their solution could be manipulated in air for a short time without notable decomposition. They have been characterized by spectroscopic methods.
The IR spectra of 1 and 2 show that the acyl carbonyl peak occurrs at 1 628 and 1 615 cm-1, respe-ctively, lower than those in polydentate acyl ligated iron complexes[22-23], but comparable to those in mono-dentate acyl ligated iron complexes[23]. The correspon-ding acyl carbon resonates at 246.9 in the 13C NMR spectrum of 1. The 1H and 13C NMR spectra of 2 contain two sets of proton and carbon signals, indica-ting that this complex possibly exists in solution as mixture of two geometric isomers (Scheme 1). The main structural difference of two isomers arises from the relative position of ortho-isopropylthiobenzoyl with 2-pyridinethiolate. Furthermore, the relative ratio of two isomers is variable in the CDCl3 and acetone-d6 solutions, showing that the interconversion between two isomers is possible in solution with the help of solvent, through partial dissociation of the ligands and succedent association process. Similar isomerism and interconversion have been observed in (pyrazol-1-yl)acyl iron complexes[23].
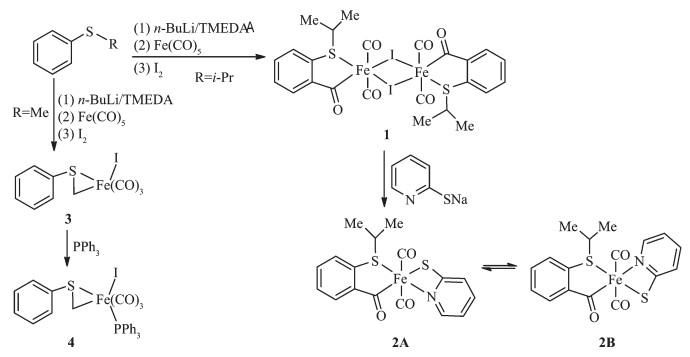
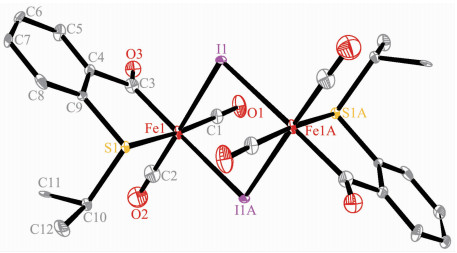
The structures of 1 and 2A were confirmed by X-ray crystallography, and are shown in Fig. 1 and 2, respectively. The selected bond distances and angles are listed in Table 2. Fig. 1 shows that complex 1 consists of two unequal iodide bridges between two benzoyl iron fragments. The Fe-I(1) bond distance (0.266 8(2) nm) is slightly shorter than the Fe-I(1A) bond distance (0.273 4(2) nm). These values are similar to those in bidentate acyl ligated iron complexes, but shorter than those in monodentate acyl ligated iron complexes[23]. Fig. 2 shows that the pyridyl nitrogen atom in 2A occupies the cis-position to the acyl group with the C(8)-Fe(1)-N(1) angle of 94.54(3)°. Complexes 1 and 2A possess analogous Fe-Cacyl and CCacyl bond distances, which are also comparable to the correspon-ding values reported for monodentate acyl iron complexes[23].
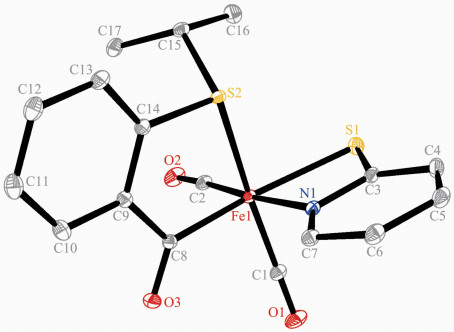
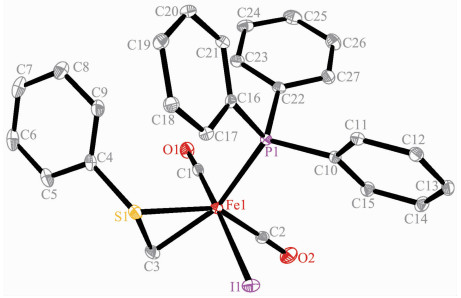
Complex 1 Fe(1)-I(1) 0.266 8(2) Fe(1)-I(1A) 0.273 4(2) Fe(1)-S(1) 0.229 8(3) Fe(1)-C(1) 0.178 1(13) Fe(1)-C(2) 0.172 6(15) Fe(1)-C(3) 0.194 8(15) C(1)-O(1) 0.113 7(16) C(3)-O(3) 0.126 3(16) C(3)-C(4) 0.151 9(19) Fe(1)-I(1)-Fe(1A) 91.82(6) I(1)-Fe(1)-I(1A) 88.18(6) C(2)-Fe(1)-I(1) 176.4(5) C(3)-Fe(1)-I(1A) 174.8(4) C(3)-Fe(1)-S(1) 87.6(4) Fe(1)-C(3)-O(3) 124.6(11) Fe(1)-S(1)-C(9) 99.1(4) C(4)-C(3)-O(3) 118.5(13) C(11)-C(10)-S(1) 113.9(9) Complex 2A Fe(1)-S⑴ 0.241 60(5) Fe(1)-S(2) 0.227 92(4) Fe(1)-N(1) 0.200 22(8) Fe(1)-C(1) 0.178 97(9) Fe(1)-C(2) 0.177 37(9) Fe(1)-C(8) 0.194 77(9) C(1)-O(1) 0.114 1(1) C(8)-O(3) 0.122 4(1) C(8)-C(9) 0.151 6(1) C(8)-Fe(1)-S(1) 164.20(2) C(8)-Fe(1)-S(2) 88.00(3) C(8)-Fe(1)-N(1) 94.54(3) Fe(1)-C(3)-N(1) 101.95(5) Fe(1)-C(8)-O(3) 125.67(7) Fe(1)-S(1)-C(3) 77.19(3) Fe(1)-S(2)-C(14) 99.47(3) S(1)-Fe(1)-S(2) 87.75(1) N(1)-Fe(1)-S(1) 69.78(2) N(1)-Fe(1)-S(2) 81.67(2) C(17)-C(15)-S(2) 114.68(6) Complex 3 Fe(1)-I(1) 0.265 17(3) Fe(1)-S(1) 0.226 51(4) Fe(1)-C(1) 0.178 6(1) Fe(1)-C(4) 0.201 9(1) C(4)-S(1) 0.176 1(1) S(1)-C(4)-Fe(1) 73.28(4) C(1)-Fe(1)-C(4) 99.60(5) C(2)-Fe(1)-S(1) 96.58(4) C(2)-Fe(1)-I(1) 176.90(4) C(4)-Fe(1)-I(1) 89.43(4) C(4)-Fe(1)-S(1) 48.11(3) Fe(1)-S(1)-C(4) 58.61(4) Fe(1)-S(1)-C(5) 111.75(4) Complex 4 Fe(1)-P(1) 0.229 44(6) Fe(1)-S(1) 0.226 54(6) Fe(1)-I(1) 0.268 21(4) Fe(1)-C(1) 0.175 6(2) Fe(1)-C(3) 0.201 7(2) C(3)-S(1) 0.176 7(2) C(1)-Fe(1)-C(3) 87.94(9) C(1)-Fe(1)-I(1) 171.26(6) C(2)-Fe(1)-S(1) 148.87(7) C(3)-Fe(1)-S(1) 48.30(6) C(3)-Fe(1)-P(1) 154.97(6) C(3)-S(1)-Fe(1) 58.47(7) C(16)-P(1)-C(22) 103.62(8) C(16)-P(1)-Fe(1) 113.49(6) S(1)-C(3)-Fe(1) 73.22(7) S(1)-Fe(1)-P(1) 106.88(2) S(1)-Fe(1)-I(1) 85.15(2) P(1)-Fe(1)-I(1) 92.87(2) Symmetry codes: A: -x+2, -y+1, -z+1 for 1. 2.2 Synthesis and reactivity of phenylthiomethyl iron complex
The lithiation process of thioanisole with n-BuLi is different from that of isopropylthiobenzene[30, 32]. Thioan-isole was deprotonated with n-BuLi to give the methyl metalation product instead of the ortho lithiation product of phenyl group[32]. Reactions of α-thiocarban-ions with iron carbonyl have been investigated, which generated acyl iron intermediates and subsequently transferred to various organic and organometallic derivatives[33-34]. However, we find that reaction of phenylthiomethyllithium with iron carbonyl followed by iodine only affords phenylthiomethyl iron complex 3 (Scheme 1). No acyl iron derivatives are obtained. Reactivity of 3 is significantly different from that of 1. For example, similar treatment of 3 with 2-pyridinethiolate results in the decomposition of 3, rather than the substitution of iodide. In addition, reaction of alkylmetal carbonyl compounds with nucleophilic ligands, such as phosphine (PR3), has been extensively used to form acyl metal derivatives through alkyl/carbonyl migratory insertion reaction[9], but reaction of 3 with PPh3 only give the carbonyl substitution product 4. No induced carbonyl insertion reaction took place, though such reaction should be able to reduce the ring strain in 3. Complexes 3 and 4 also are air-stable solids. Their IR and NMR spectra are highly consistent with the alkyl iron structure. These two complexes only exhibit three (for 3) or two (for 4) absorption bands in the range of 2 077~1 950 cm-1 for terminal carbonyl ligands, and no acyl carbonyl absorption band is observed in their IR spectra.
Complexes 3 and 4 were also characterized by single crystal X-ray structural determination. Fig. 3 and 4 reveal that the fundamental frameworks in complexes 3 and 4 are similar to each other, except that PPh3 locates on the trans-position of the methyl carbon in 4 instead of one carbonyl group like in 3. The phenylthiomethyl group bonds to the iron atom in a bidentate κ2-[C, S] chelating fashion in these two complexes, yielding a three-membered metallocyclic structure. Complexes 3 and 4 possess analogous Fe-S, Fe-I and Fe-Cmethyl bond distances (Table 2). The Fe-S and Fe-I bond distances are also comparable to the corresponding values in 1 and 2.
3 Conclusions
In summary, ortho-isopropylthiobenzoyl and phenylthiomethyl iron derivatives have been obtained by the reaction of ortho-isopropylthiophenyllithium or phenylthiomethyllithium with iron carbonyl followed by treatment with iodine. These complexes also show markedly different reactivities upon treatment with nucleophilic ligands.
-
-
[1]
Almenara N, Ibarlucea L, Mendicute-Fierro C, et al. Dalton Trans., 2016, 45:18502-18509 doi: 10.1039/C6DT03778J
-
[2]
Panthi B D, Gipson S L, Franken A. Inorg. Chim. Acta, 2015, 425:176-181 doi: 10.1016/j.ica.2014.09.035
-
[3]
Wotal A C, Ribson R D, Weix D J. Organometallics, 2014, 33:5874-5881 doi: 10.1021/om5004682
-
[4]
Hermanson J R, Figley T M, Seibert A L, et al. J. Organomet. Chem., 2008, 693:2061-2064 doi: 10.1016/j.jorganchem.2008.02.028
-
[5]
Omae I. Coord. Chem. Rev., 2011, 255:139-160 doi: 10.1016/j.ccr.2010.08.001
-
[6]
Joe C L, Doyle A G. Angew. Chem. Int. Ed., 2016, 55:4040-4043 doi: 10.1002/anie.201511438
-
[7]
苏吕, 肖含兵, 苑雨萌, 等.有机化学, 2017, 37:630-635 http://www.cnki.com.cn/Article/CJFDTOTAL-SYQY201603027.htmSU Lü, XIAO Han-Bing, YUAN Yu-Meng, et al. Chin. J. Org. Chem., 2017, 37:630-635 http://www.cnki.com.cn/Article/CJFDTOTAL-SYQY201603027.htm
-
[8]
崔龙, 周喜, 杨先贵, 等.无机化学学报, 2014, 30(7):1600-1608 http://www.cnki.com.cn/Article/CJFDTOTAL-SYQY201603027.htmCUI Long, ZHOU Xi, YANG Xian-Gui, et al. Chinese J. Inorg. Chem., 2014, 30(7):1600-1608 http://www.cnki.com.cn/Article/CJFDTOTAL-SYQY201603027.htm
-
[9]
Wang X, Cao K, Liu Y, et al. J. Am. Chem. Soc., 2013, 135:3399-3402 doi: 10.1021/ja400755e
-
[10]
Murshid N, Rahman M A, Wang X. J. Organomet. Chem., 2016, 819:109-114 doi: 10.1016/j.jorganchem.2016.06.031
-
[11]
张天永, 盛了, 李彬, 等.化学研究与应用, 2015, 27(11):1609-1618 doi: 10.3969/j.issn.1004-1656.2015.11.001ZHANG Tian-Yong, SHENG Liao, LI Bin, et al. Chem. Res. Appl., 2015, 27(11):1609-1618 doi: 10.3969/j.issn.1004-1656.2015.11.001
-
[12]
Corr M J, Murphy J A. Chem. Soc. Rev., 2011, 40:2279-2292 doi: 10.1039/c0cs00150c
-
[13]
Wright J A, Turrell P J, Pickett C J. Organometallics, 2010, 29:6146-6156 doi: 10.1021/om1008567
-
[14]
Hiromoto T, Ataka K, Pilak O, et al. FEBS Lett., 2009, 583:585-590 doi: 10.1016/j.febslet.2009.01.017
-
[15]
Hu B, Chen X, Gong D, et al. Organometallics, 2016, 35:2993-2998 doi: 10.1021/acs.organomet.6b00524
-
[16]
Xu T, Yin C J M, Wodrich M D, et al. J. Am. Chem. Soc., 2016, 138:3270-3273 doi: 10.1021/jacs.5b12095
-
[17]
Turrell P J, Hill A D, Ibrahim S K, et al. Dalton Trans., 2013, 42:8140-8146 doi: 10.1039/c3dt50642h
-
[18]
Royer A M, Salomone-Stagni M, Rauchfuss T B, et al. J. Am. Chem. Soc., 2010, 132:16997-17003 doi: 10.1021/ja1072228
-
[19]
Turrell P J, Wright J A, Peck J M T, et al. Angew. Chem. Int. Ed., 2010, 49:7508-7511 doi: 10.1002/anie.v49:41
-
[20]
Chen D, Scopelliti R, Hu X. J. Am. Chem. Soc., 2010, 132:928-929 doi: 10.1021/ja9100485
-
[21]
Royer A M, Rauchfuss T B, Gray D L. Organometallics, 2009, 28:3618-3620 doi: 10.1021/om9004059
-
[22]
Zhao D W, Liu X L, Zhang X Y, et al. J. Organomet. Chem., 2015, 780:56-62 doi: 10.1016/j.jorganchem.2014.12.039
-
[23]
Zhao D W, Xu Y, Guo Y W, et al. J. Organomet. Chem., 2015, 791:303-310 doi: 10.1016/j.jorganchem.2015.06.008
-
[24]
Yang X, Hall M B. J. Am. Chem. Soc., 2009, 131:10901-10908 doi: 10.1021/ja902689n
-
[25]
Murray K A, Wodrich M D, Hu X, et al. Chem. Eur. J., 2015, 21:3987-3996 doi: 10.1002/chem.201405619
-
[26]
Song L C, Xu K K, Han X F, et al. Inorg. Chem., 2016, 55:1258-1269 doi: 10.1021/acs.inorgchem.5b02490
-
[27]
Durgaprasad G, Xie Z L, Rose M J. Inorg. Chem., 2016, 55:386-389 doi: 10.1021/acs.inorgchem.5b01733
-
[28]
CrystalStructure 3. 7. 0, Crystalclear 1. 36: Crystal Structure Analysis Package, Rigaku and Rigaku/MSC, TX, 2000.
-
[29]
Sheldrick G M. Acta Crystallogr. Sect. A, 2008, A64:112-114 http://citeseerx.ist.psu.edu/viewdoc/summary?doi=10.1.1.526.2513
-
[30]
Horner L, Lawson A J, Simons G. Phosphorus Sulfur Silicon Relat. Elem., 1982, 12(3):353-356 doi: 10.1080/03086648208078968
-
[31]
Chen D, Scopelliti R, Hu X. Angew. Chem. Int. Ed., 2010, 49:7512-7515 doi: 10.1002/anie.v49:41
-
[32]
Seebach D, Gabriel J, Hässig R. Helv. Chim. Acta, 1984, 67:1083-1099 doi: 10.1002/hlca.19840670421
-
[33]
Alper H, Fabre J L. Organometallics, 1982, 1:1037-1040 doi: 10.1021/om00068a006
-
[34]
Lotz S, Dillen J L M, van Dyk M M. J. Organomet. Chem., 1989, 371:371-382 doi: 10.1016/0022-328X(89)85232-5
-
[1]
-
Table 1. Crystal data and refinement parameters for complexes 1, 2A, 3 and 4
Complex 1 2A 3 4 Formula C24H22Fe2l2O6S2 C17H15FeNO3S2 C10H7FeIO3S C27H22FeIO2PS Formula weight 836.03 401.27 389.97 624.23 Crystal size/mm 0.20×0.18×0.12 0.20×0.18×0.12 0.24×0.22×0.16 0.20×0.18×0.12 Crystal system Monoclinic Monoclinic Monoclinic Monoclinic Space group C2/c P21/n P21/n P21/c a/nm 2.267 3(5) 1.166 2(2) 1.229 12(13) 1.273 57(18) b/nm 0.961 08(19) 1.250 4(3) 0.710 29(7) 1.049 13(15) c/nm 1.649 6(3) 1.188 6(2) 1.433 60(14) 1.871 3(2) β/(°) 127.86(3) 95.379(2) 97.984(2) 92.063(3) T/K 113(2) 113(2) 113(2) 113(2) V/nm3 2.837 8(13) 1.725 5(6) 1.239 4(2) 2.498 7(6) Z 4 4 4 4 Dc/(g·cm-3) 1.957 1.545 2.090 1.659 F(000) 1 616 824 744 1 240 μ/mm-1 3.381 1.130 3.863 2.009 θ range/(°) 2.40~25.02 2.34~36.37 2.05~28.71 3.20~27.52 Measured reflection 14 131 30 684 16 369 31 228 Unique reflection (Rint) 2 513 (0.076 8) 8 160 (0.019 9) 3 210 (0.019 9) 5 749 (0.046 5) Observed reflection with [I > 2σ(I)] 2 004 7 136 3 082 5 039 Parameter 165 219 145 298 GOF 1.219 1.014 1.056 0.998 Residuals R, wR2 0.076 9, 0.160 5 0.024 7, 0.063 5 0.011 5, 0.028 1 0.023 7, 0.060 0 Table 2. Selected bond distances (nm) and angles (°) for complexes 1, 2A, 3 and 4
Complex 1 Fe(1)-I(1) 0.266 8(2) Fe(1)-I(1A) 0.273 4(2) Fe(1)-S(1) 0.229 8(3) Fe(1)-C(1) 0.178 1(13) Fe(1)-C(2) 0.172 6(15) Fe(1)-C(3) 0.194 8(15) C(1)-O(1) 0.113 7(16) C(3)-O(3) 0.126 3(16) C(3)-C(4) 0.151 9(19) Fe(1)-I(1)-Fe(1A) 91.82(6) I(1)-Fe(1)-I(1A) 88.18(6) C(2)-Fe(1)-I(1) 176.4(5) C(3)-Fe(1)-I(1A) 174.8(4) C(3)-Fe(1)-S(1) 87.6(4) Fe(1)-C(3)-O(3) 124.6(11) Fe(1)-S(1)-C(9) 99.1(4) C(4)-C(3)-O(3) 118.5(13) C(11)-C(10)-S(1) 113.9(9) Complex 2A Fe(1)-S⑴ 0.241 60(5) Fe(1)-S(2) 0.227 92(4) Fe(1)-N(1) 0.200 22(8) Fe(1)-C(1) 0.178 97(9) Fe(1)-C(2) 0.177 37(9) Fe(1)-C(8) 0.194 77(9) C(1)-O(1) 0.114 1(1) C(8)-O(3) 0.122 4(1) C(8)-C(9) 0.151 6(1) C(8)-Fe(1)-S(1) 164.20(2) C(8)-Fe(1)-S(2) 88.00(3) C(8)-Fe(1)-N(1) 94.54(3) Fe(1)-C(3)-N(1) 101.95(5) Fe(1)-C(8)-O(3) 125.67(7) Fe(1)-S(1)-C(3) 77.19(3) Fe(1)-S(2)-C(14) 99.47(3) S(1)-Fe(1)-S(2) 87.75(1) N(1)-Fe(1)-S(1) 69.78(2) N(1)-Fe(1)-S(2) 81.67(2) C(17)-C(15)-S(2) 114.68(6) Complex 3 Fe(1)-I(1) 0.265 17(3) Fe(1)-S(1) 0.226 51(4) Fe(1)-C(1) 0.178 6(1) Fe(1)-C(4) 0.201 9(1) C(4)-S(1) 0.176 1(1) S(1)-C(4)-Fe(1) 73.28(4) C(1)-Fe(1)-C(4) 99.60(5) C(2)-Fe(1)-S(1) 96.58(4) C(2)-Fe(1)-I(1) 176.90(4) C(4)-Fe(1)-I(1) 89.43(4) C(4)-Fe(1)-S(1) 48.11(3) Fe(1)-S(1)-C(4) 58.61(4) Fe(1)-S(1)-C(5) 111.75(4) Complex 4 Fe(1)-P(1) 0.229 44(6) Fe(1)-S(1) 0.226 54(6) Fe(1)-I(1) 0.268 21(4) Fe(1)-C(1) 0.175 6(2) Fe(1)-C(3) 0.201 7(2) C(3)-S(1) 0.176 7(2) C(1)-Fe(1)-C(3) 87.94(9) C(1)-Fe(1)-I(1) 171.26(6) C(2)-Fe(1)-S(1) 148.87(7) C(3)-Fe(1)-S(1) 48.30(6) C(3)-Fe(1)-P(1) 154.97(6) C(3)-S(1)-Fe(1) 58.47(7) C(16)-P(1)-C(22) 103.62(8) C(16)-P(1)-Fe(1) 113.49(6) S(1)-C(3)-Fe(1) 73.22(7) S(1)-Fe(1)-P(1) 106.88(2) S(1)-Fe(1)-I(1) 85.15(2) P(1)-Fe(1)-I(1) 92.87(2) Symmetry codes: A: -x+2, -y+1, -z+1 for 1. -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 2
- 文章访问数: 1356
- HTML全文浏览量: 208



 下载:
下载:




 下载:
下载:

