
无铅和稀土的大尺寸分子基绿光荧光晶体:(C5H13ClN)2[MnCl4]
English
Lead/Rare Earth-Free Green-Light-Emitting Crystal of Molecular-Based Hybrid Compound: (C5H13ClN)2[MnCl4] with Large Crystal Size
-
Key words:
- organic-inorganic hybrid
- / crystal structure
- / luminescence
-
0 Introduction
Luminescent materials recently are receiving increasing attention due to their promising applica-tions in lighting, flat panel display, data storage and so on[1-4]. However, the traditional luminescent materials were usually prepared by introducing rare-earth ions as dopants, which suffered from cumbersome prepara-tion process and low light-emitting efficiency[5-9]. With the flourishing development of (CH3NH3)[PbI3], the scientific research interest on luminescent materials has been gradually shifted to organic-inorganic hybrid materials which possess richness of structural trans-formation and diversity of physical properties as well as potential practical applications[10-20]. In particular, the organic-inorganic hybrid materials with chemical formula (A)2[MX4] (A=organic cation, M=transition divalent metal ion and X=halide ion), possess various structures, modulated phases, magnetic properties and thermochromic behavior[21-27]. Moreover, the organic-inorganic hybrid compounds, which overcome the drawbacks of traditional rare earth-doped luminescent materials and own facile synthesis and mechanical flexibility, would provide a new strategy for the fabrication of luminescent devices[5].
From the viewpoint of the preservation of environment and the utilization of cheap natural resources, the research on luminescent materials on the basis of lead-free compounds has attracted growing attention. Mn2+ is well known to display inter-esting photophysical properties such as fluorescence and phosphorescence, and an essential feature of members of these groups is that they could emit either green (tetrahedrally coordinated Mn2+) or red to pink (octahedrally coordinated Mn2+) luminescence depend-ing on their coordination environment[28-30]. Especially, the tetrahedrally coordinated manganese(Ⅱ) organic-inorganic hybrid complexes, constitute a special interest and might be promising phosphor materials due to their simple synthetic methods, interesting solid-state properties as well as enlarged applications in fluorescent tubes, radiation detectors, cathode-ray tubes, and X-ray imaging screens as well as in signs, toys, and so on[28, 31-38]. Besides, comparing with the green-emitting rare and rare-earth precious metal ions Tb, the inexpensive/abundant manganese would possess an increasingly predominant superiority in the preparation of luminescent materials. In the past few years, the optical properties of Mn-based compounds with various methylamine or pyridine and their deriva-tive cations have been studied extensively both in experiment and theory. For instance, Xiong's group reported a series of organic-inorganic hybrid lumine-scent ferroelectrics on the basis of pyrrolidinium base[5, 36, 39-42], which has inspired our research interest.
In search of new hybrids with interesting optical properties, we designed the luminescent organic-inorganic hybrid material, (C5H13ClN)2[MnCl4], which exhibits intense greenish fluorescent emission (520 nm) and high thermal stability. The synthesis and characterization of 1 were also disclosed through the X-ray single-crystal diffraction, powder X-ray diffraction, etc. Structure analysis, thermal analysis and optical analysis were used together to demonstrate the crystal structure as well as the PL emission characteristics of 1. It was hoped that this study would shed new light on the development of emitting materials based on organic-inorganic hybrid compounds and open new opportunities for probing novel emitting materials.
1 Experimental
1.1 Preparation of crystal
All reagents were purchased from Aladdin Co., Ltd. and used without further purification. (C5H13ClN)2[MnCl4] was prepared through the following process: a solution of 2-dimethylaminoisopropyl chloride hydro-chloride (3.16 g, 0.02 mol) in methanol was slowly added to the methanol solution of anhydrous mang-anese chloride (1.26 g, 0.01 mol) under stirring. And slow evaporation of the mixed solution at 333 K yielded green crystals. Anal. Calcd. for C10H26Cl6MnN2(%): C 27.17, H 5.93, N 6.34; Found(%): C 27.21, H 5.96, N 6.37.
1.2 Single crystal X-ray diffraction analysis
The X-ray diffraction data collection was carried out on a Rigaku Saturn 724 diffractometer equipped with graphite monochromated Mo Kα radiation (λ=0.071 073 nm) at 293 K. Data collection and cell refine-ment were performed using Crystalclear software package (Rigaku, 2005). The structure was solved by the direct method and full-matrix least-squares refinement based on F2 was accomplished through SHELXL-97[43]. All hydrogen atoms on carbon/nitrogen atoms were generated geometrically and refined using a "riding" model with Uiso(H)=1.2Ueq. The anisotropi-cally refinements for non-hydrogen atoms were taken using all reflections with I>2σ(I). Details concerning crystal data and refinement could be found in Table 1. Selected bond lengths/angles and hydrogen bonds are available in Table S1.
Empirical formula C10H26Cl6MnN2 Formula weight 441.97 Crystal system Monoclinic Space group P21/c a/nm 1.142 4(2) b/nm 1.256 2(3) c/nm 1.463 3(3) β/(°) 99.310(3) Volume/nm3 2.072 3(7) Z 4 Dc/(g·cm3) 1.417 F(000) 908 Collected reflection 9 913 Unique reflection 3 601 Rint 0.095 Observed reflection 2 714 Goodness-of-fit on F2 1.116 R1, wR2[I>2σ(I)] 0.080, 0.307 CCDC: 1570589.
1.3 IR and powder X-ray diffraction measurements
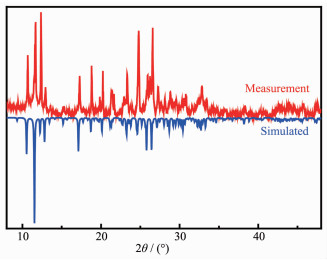
IR spectrum was recorded on a Shimadzu model IR-60 spectrometer at room temperature, with the sample prepared into KBr diluted pellet (Fig.S1). The powder X-ray diffraction measurement of 1 was carried out on a PANalytical X'Pert PRO X-ray diffractometer with Cu Kα radiation (λ=0.154 18 nm) and a fixed power source (30.0 kV, 10.0 mA) at 293 K. The diffraction pattern was collected in the 2θ range of 5°~50° with a step size of 0.02°. And the powder X-ray diffraction (PXRD) pattern of 1 matched pretty well with the pattern simulated from the single crystal structure, which indicated the phase purity (Fig. 1).
1.4 Luminescence measurements
Excitation and emission spectra measurements were carried out on Pluorolog 3-TCSPC spectrofluo-rometer (Horiba Jobin Yvon Inc). This kind of spectrofluorometer is equipped with a 450 W xenon lamp as an exciting source, and an emission monochromator, a double-excitation monochromator, and a photomultiplier as detection system. Measure-ments were performed at room temperature.
1.5 Thermogravimetric (TGA) analysis
Thermogravimetric analysis (TG) was performed on a TA type SDT-Q600 thermo-analyzer with a heating rate of 10 K·min-1 from 300 to 1 073 K, and the sample was under nitrogen atmosphere in the process of measurement.
2 Results and discussion
2.1 Crystal structure of 1
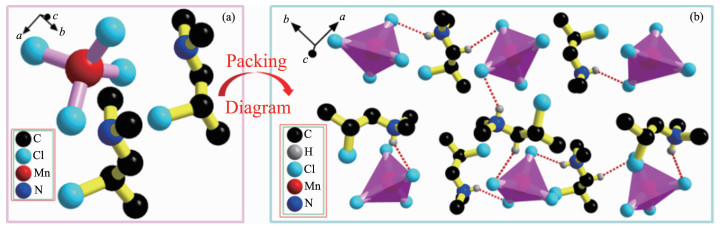
To understand the mechanism of emitting better, a precise single-crystal structure measurement for 1 was performed at room temperature. Compound 1 crystallizes in the monoclinic crystal system with the centrosymmetric space group P21/c and point group 2/m. As shown in Fig. 2a, the asymmetric unit of 1 consists of two (C5H12ClN)+ cations and one isolated [MnCl4]2- anion, where the environment around Mn could be described as a distorted tetrahedral. Under room temperature, the Mn-Cl bond lengths varied from 0.236(2) to 0.238(2) nm, giving the mean value of 0.237(2) nm (Tables S1), which were within normal range and in accordance with other structurally similar compounds. The Cl-Mn-Cl angles were in the range from 102.82(8)° to 115.79(8)°, distorted from the ideal tetrahedral geometry of 109.471°. Moreover, the packing diagram of 1 was presented in Fig. 2b. As shown in Fig. 2b, one layer of cations was embedded in the free space between two neighboring inorganic [MnCl4]2- layer, forming an alternating well-known organic-inorganic hybrid structure. Moreover, as presented in Fig. 2b, the packing was stabilized by N-H…Cl hydrogen bonds and each anion forms two N-H…Cl hydrogen bonds with two nearest cations, and the distances of H1D…Cl2 and H2D…Cl4i were 0.247 and 0.243 nm, respectively.
2.2 Thermal property of 1
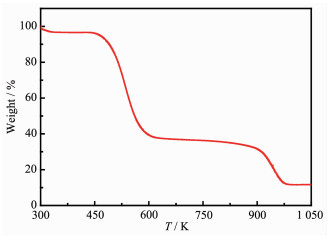
For characterizing the thermal stability of compound 1, TGA measurement was performed from 300 to 1 073 K. As shown in Fig. 3, the sample has a perfect thermal stability up to T=473 K. Besides, the decomposition process of 1 could be divided into the following stages. Firstly (473~614 K), 1 decomposed into metal(Ⅱ) chloride and carbon[21, 44-50]. Secondly (910 ~1 027 K), the mixture of metal(Ⅱ) chloride and elemental carbon decomposed continuously[21].
2.3 Optical property of 1
As shown in Fig. 4a, the yellow green crystal and powder of (C5H13ClN)2[MnCl4] emitted strong green light while illuminated by UV lamp (365 nm). And in order to better characterize the luminescence property of 1, the absorption, excitation as well as emission spectra analyses were depicted as follows. The absorption spectrum of the powder sample includes two strong absorption peaks (358 and 444 nm) in the ultraviolet region, which are related to the 6A1→4T1 electronic transition of Mn2+. The excitation spectrum was recorded under room temperature and exhibited two dominant absorption peaks at around 357 and 450 nm (Fig. 4b), which coincided with the UV results. These excitation peaks were also related to the electronic transition of Mn2+ from the 6A1 ground state to different excited spin-quartet states, which can be attributed to tetrahedral crystal-field transition based on the related irreps in Td-symmetry notation[5, 37, 51-52]. And the photoluminescence (PL) emission spectra were recorded by exciting the crystal using 360 and 460 nm excitation wavelength (Fig. 4c and 4d). Intere-stingly, under the selected two excitation wavelength, a strong emission peak centered at around 521 nm could be observed, confirming the special emission property of compound 1. The emission lifetime of 1 was as long as 83 μs. Besides, we also estimated the luminescence quantum yield of compound 1, and a relatively high QE of 15% was obtained.
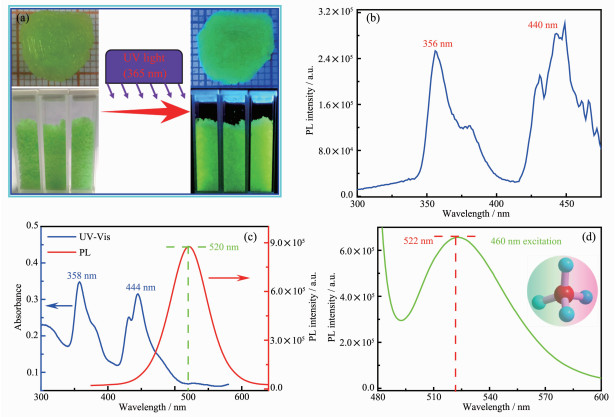 图4
(a) Yellow green crystal and powder of 1 under sunlight and UV light (365 nm), and the size of crystal 1 was about 1 cm×1 cm×0.3 cm; (b) Excitation spectrum of 1 at room temperature; (c) Absorption (blue line) and emission (red line) spectra of 1 at room temperature; (d) Emission spectrum of 1 at room temperature under 460 nm excitation wavelength
Figure4.
(a) Yellow green crystal and powder of 1 under sunlight and UV light (365 nm), and the size of crystal 1 was about 1 cm×1 cm×0.3 cm; (b) Excitation spectrum of 1 at room temperature; (c) Absorption (blue line) and emission (red line) spectra of 1 at room temperature; (d) Emission spectrum of 1 at room temperature under 460 nm excitation wavelength
图4
(a) Yellow green crystal and powder of 1 under sunlight and UV light (365 nm), and the size of crystal 1 was about 1 cm×1 cm×0.3 cm; (b) Excitation spectrum of 1 at room temperature; (c) Absorption (blue line) and emission (red line) spectra of 1 at room temperature; (d) Emission spectrum of 1 at room temperature under 460 nm excitation wavelength
Figure4.
(a) Yellow green crystal and powder of 1 under sunlight and UV light (365 nm), and the size of crystal 1 was about 1 cm×1 cm×0.3 cm; (b) Excitation spectrum of 1 at room temperature; (c) Absorption (blue line) and emission (red line) spectra of 1 at room temperature; (d) Emission spectrum of 1 at room temperature under 460 nm excitation wavelength
3 Conclusions
In summary, through deliberate selection to the organic cations, we successfully synthesized an organic -inorganic hybrid compound (C5H13ClN)2[MnCl4], which exhibits unusual thermal stability and excellent green light emission under external stimulus. Moreover, 1 was prepared through the simple and convenient solution method and its crystal structure and fluorescence property had been discussed in this paper. These perfect characteristics together with economic and environment friendly preparation process enable 1 to show potential applications as an emitting material. It is not only useful for the fabrication of low-cost emitting devices, but also advantageous to promote the development of emitting materials in display and sensing fields.
Supporting information is available at http://www.wjhxxb.cn
-
-
[1]
Xu L J, Sun C Z, Xiao H, et al. Adv. Mater., 2017, 29(10):1605739 doi: 10.1002/adma.201605739
-
[2]
Huang Q, Meerheim R, Fehse K, et al. SID International Symposium Digest of Technical Papers, 2007, 38(1):1282-1285 doi: 10.1889/1.2785546
-
[3]
Jou J-H, Kumar S, Agrawal A, et al. J. Mater. Chem. C, 2015, 3(13):2974-3002 doi: 10.1039/C4TC02495H
-
[4]
Volz D, Wallesch M, Fléchon C, et al. Green Chem., 2015, 17(4):1988-2011 doi: 10.1039/C4GC02195A
-
[5]
Jiang C L, Zhong N, Luo C H, et al. Chem. Commun., 2017, 53(44):5954-5957 doi: 10.1039/C7CC01107E
-
[6]
Blazek K, Krasnikov A, Nejezchleb K, et al. Phys. Status Solidi B, 2005, 242(6):1315-1323 doi: 10.1002/(ISSN)1521-3951
-
[7]
Kan D, Kanda R, Kanemitsu Y, et al. Appl. Phys. Lett., 2006, 88(19):191916 doi: 10.1063/1.2202750
-
[8]
Shimizu Y, Sakagami S, Goto K, et al. Mater. Sci. Eng. B, 2009, 161(1):100-103 http://www.sciencedirect.com/science/article/pii/S0921510708005965
-
[9]
Yamamoto H, Okamoto S, Kobayashi H. J. Lumin., 2002, 100(1):325-332 http://www.sciencedirect.com/science/article/pii/S0022231302004325
-
[10]
Mostafa M F, El Dean T S, Tammam A K. Mater. Chem. Phys., 2016, 180:373-382 doi: 10.1016/j.matchemphys.2016.06.020
-
[11]
Xu C, Zhang W Y, Chen C, et al. J. Mater. Chem. C, 2017, 5(28):6945-6953 doi: 10.1039/C7TC02087B
-
[12]
Lv X H, Liao W Q, Wang Z X, et al. Cryst. Growth Des, 2016, 16(7):3912-3916 doi: 10.1021/acs.cgd.6b00480
-
[13]
Fu X Q, Hang T, Ye Q, et al. Inorg. Chem. Commun., 2011, 14(1):281-284 doi: 10.1016/j.inoche.2010.11.014
-
[14]
Zhao M M, Ge J Z, Qu Z R. Inorg. Chem. Commun., 2010, 13(10):1152-1155 doi: 10.1016/j.inoche.2010.06.034
-
[15]
王国喜, 李波, 熊仁根, 等.无机化学学报, 2007, 23(11):1997-1998 doi: 10.3321/j.issn:1001-4861.2007.11.028WANG Guo-Xi, LI Bo, XIONG Ren-Gen, et al. Chinese J. Inorg. Chem., 2007, 23(11):1997-1998 doi: 10.3321/j.issn:1001-4861.2007.11.028
-
[16]
徐海军, 付大伟.无机化学学报, 2008, 24(8):1352-1355XU Hai-Jun, FU Da-Wei. Chinese J. Inorg. Chem., 2008, 24(8):1352-1355
-
[17]
Lv X H, Liao W Q, Li P F, et al. J. Mater. Chem. C, 2016, 4(9):1881-1885 doi: 10.1039/C5TC04114G
-
[18]
Szafranski M. Cryst. Growth Des, 2016, 16(7):3771-3776 doi: 10.1021/acs.cgd.6b00279
-
[19]
陆新荣, 赵颖, 刘建, 等.无机化学学报, 2015, 31(9):1678-1686LU Xin-Rong, ZHAO Ying, LIU Jian, et al. Chinese J. Inorg. Chem., 2015, 31(9):1678-1686
-
[20]
左锣, 冯建华, 郭莉, 等.无机化学学报, 2013, 29(9):1979-1984ZUO Luo, FENG Jian-Hua, GUO Li, et al. Chinese J. Inorg. Chem., 2013, 29(9):1979-1984
-
[21]
Busi S, Fröhlich R, Lahtinen M, et al. Z. Naturforsch. B:Chem. Sci., 2007, 62(1):35-43 http://www.degruyter.com/view/j/znb.2007.62.issue-1/znb-2007-0106/znb-2007-0106.xml
-
[22]
Matsumoto T, Miyazaki Y, Albrecht A S, et al. J. Phys. Chem. B, 2000, 104(43):9993-10000 doi: 10.1021/jp0020081
-
[23]
Hammar P R, Dender D C, Reich D H, et al. J. Appl. Phys., 1997, 81(8):4615-4617 doi: 10.1063/1.365180
-
[24]
Xiao Z L, Chen H Z, Shi M M, et al. Mater. Sci. Eng. B, 2005, 117(3):313-316 doi: 10.1016/j.mseb.2004.12.052
-
[25]
Gesi K. J. Phys. Soc. Jpn., 2002, 71(5):1376-1380 doi: 10.1143/JPSJ.71.1376
-
[26]
Nicholson R, Fujii Y, Chen X, et al. J. Phys. Chem. Solids, 2001, 62(5):933-940 doi: 10.1016/S0022-3697(00)00257-2
-
[27]
Willett R, Wei M. J. Phys. Chem. Solids, 2000, 61(12):2025-2028 doi: 10.1016/S0022-3697(00)00201-8
-
[28]
Pitula S, Mudring A V. Chem. Eur. J., 2010, 16(11):3355-3365 doi: 10.1002/chem.v16:11
-
[29]
Orgel L. J. Chem. Phys., 1955, 23(10):1958-1958 doi: 10.1063/1.1740614
-
[30]
Lawson K E. J. Chem. Phys., 1967, 47(9):3627-3633 doi: 10.1063/1.1712432
-
[31]
Seddon K, Earle M. UK Patent, WO2006043110A1. 2006-04-27.
-
[32]
Balsamy S, Natarajan P, Vedalakshmi R, et al. Inorg. Chem., 2014, 53(12):6054-6059 doi: 10.1021/ic500400y
-
[33]
Cotton F A, Daniels L M, Huang P. Inorg. Chem., 2001, 40(14):3576-3578 doi: 10.1021/ic0101836
-
[34]
Chandra B, Khokhar M, Gupta R, et al. Pramana, 1987, 29(4):399-407 doi: 10.1007/BF02845778
-
[35]
Hardy G E, Zink J I. Inorg. Chem., 1976, 15(12):3061-3065 doi: 10.1021/ic50166a026
-
[36]
Barreda-Argueso J A, Nataf L, Rodríguez-Lazcano Y, et al. Inorg. Chem., 2014, 53(19):10708-10715 doi: 10.1021/ic501854g
-
[37]
Rodríguez-Lazcano Y, Nataf L, Rodríguez F. Phys. Rev. B, 2009, 80(8):085115 http://adsabs.harvard.edu/abs/2009PhRvB..80h5115R
-
[38]
Gesi K, Ozawa K. J. Phys. Soc. Jpn., 1984, 53(2):627-634 doi: 10.1143/JPSJ.53.627
-
[39]
Lee J-H P, Lewis B D, Mendes J M, et al. J. Coord. Chem., 2003, 56(16):1425-1442 doi: 10.1080/00958970310001642591
-
[40]
Zhang Y, Liao W Q, Fu D W, et al. J. Am. Chem. Soc., 2015, 137(15):4928-4931 doi: 10.1021/jacs.5b01680
-
[41]
Zhang Y, Liao W Q, Fu D W, et al. Adv. Mater., 2015, 27(26):3942-3946 doi: 10.1002/adma.201501026
-
[42]
Ye H Y, Zhou Q H, Niu X H, et al. J. Am. Chem. Soc., 2015, 137(40):13148-13154 doi: 10.1021/jacs.5b08290
-
[43]
Sheldrick G M. SHELX-97, Program for the Solution and the Refinement of Crystal Structures, University of Göttingen, Germany, 1997.
-
[44]
Emam S M, AbouEl-Enein S A, Emara E M. J. Therm. Anal. Calorim., 2017, 127(2):1611-1630 doi: 10.1007/s10973-016-5835-6
-
[45]
AbouEl-Enein S A, Emam S M, Polis M W, et al. J. Mol. Struct., 2015, 1099(2):567-578
-
[46]
Emam S M, El-Sayed I E T, Nassar N. Spectrochim. Acta. A Mol. Biomol. Spectrosc., 2015, 138(5):942-953 http://www.ncbi.nlm.nih.gov/pubmed/24867072
-
[47]
El-Metwally N M, Arafa R, El-Ayaan U. J. Therm. Anal. Calorim., 2014, 115(3):2357-2367 doi: 10.1007/s10973-013-3065-8
-
[48]
Gaber M, El-Ghamry H A, Fathalla S K. Spectrochim. Acta Part A, 2015, 139:396-404 doi: 10.1016/j.saa.2014.12.057
-
[49]
AbouEl-Enein S. J. Therm. Anal. Calorim., 2008, 91(3):929-936 doi: 10.1007/s10973-006-8281-z
-
[50]
El-Boraey H A. J. Therm. Anal. Calorim., 2005, 81(2):339-346 doi: 10.1007/s10973-005-0789-0
-
[51]
Sugano S, Tanabe Y, Kamimura H. Multiplets of Transition-Metal Ions in Crystals. New York and London:Academic Press, 1970:331
-
[52]
Griffith J S. The Theory of Transition-Metal Ions. New York:Cambridge University Press, 1961:455
-
[1]
-
Figure 4 (a) Yellow green crystal and powder of 1 under sunlight and UV light (365 nm), and the size of crystal 1 was about 1 cm×1 cm×0.3 cm; (b) Excitation spectrum of 1 at room temperature; (c) Absorption (blue line) and emission (red line) spectra of 1 at room temperature; (d) Emission spectrum of 1 at room temperature under 460 nm excitation wavelength
Emission spectrum in (c) was obtained by excited the powder sample under 360 nm excitation wavelength; strong green-emission at around 520 nm was associated with the electronic transition of Mn2+ in [MnCl4]2- tetrahedron in (c) and (d)
Table 1. Crystal data and structure refinement for F
Empirical formula C10H26Cl6MnN2 Formula weight 441.97 Crystal system Monoclinic Space group P21/c a/nm 1.142 4(2) b/nm 1.256 2(3) c/nm 1.463 3(3) β/(°) 99.310(3) Volume/nm3 2.072 3(7) Z 4 Dc/(g·cm3) 1.417 F(000) 908 Collected reflection 9 913 Unique reflection 3 601 Rint 0.095 Observed reflection 2 714 Goodness-of-fit on F2 1.116 R1, wR2[I>2σ(I)] 0.080, 0.307 -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 3
- 文章访问数: 1678
- HTML全文浏览量: 202


 下载:
下载:



 下载:
下载:

