
基于PbBr2的有机-无机杂化化合物的合成和晶体结构
-
关键词:
- 溴化铅
- / 有机-无机杂化化合物
- / 晶体结构
English
Syntheses and Crystal Structures of Two Homogeneous Organic-Inorganic Compounds Based on PbBr2
-
Key words:
- lead bromide
- / organic-inorganic hybrid compounds
- / crystal structure
-
0 Introduction
Haloplumbate-based hybrids have been received considerable research interests due to their tunable structures from the discrete mononuclear or polynuclear species (zero-dimensional; abbr. 0D) to the infinite variety with higher dimensionality[1-13] (one-dimensional[1, 3-9], two-dimensional[10-11] or three-dimen-sional[2]; hereafter abbr. as 1D, 2D and 3D, respectively) and the wide range of novel physical properties, beneficial in optics[14-20] and electronics[21-22].
In the context of haloplumbate-based hybrids, the perovskite-type ones have attracted tremendous research interest. The 3D haloplumbate-based perovskites, (CH3NH3)PbI3-xClx, with much lower exciton binding energies and intense light absorption over the whole visible light region have been employed as absorbers in solar cells. It is remarkable that the records of certified power conversion efficiencies have being constantly updated and over merely a few years, the power conversion efficiency has been enhanced to 22.1%[20]. Most recently, the (CH3NH3)PbI3-xClx perovs-kites have been found to show amazing bipolar and bistable resistive switching behavior with small on-off voltage (< 1.0 V) in a simple metal-dielectric-metal capacitor configuration device memory field[23]. The 2D haloplumbate-based hybrids, (N-MEDA)[PbBr4-xClx] (N-MEDA=N-1-methylethane-1, 2-diammonium, x=0~1.2), are single-phase white-light emitters, and their broadband emission across the entire visible spectrum arises from corrugated lead halide sheets. Interes-tingly, the emission is tunable through halide substitu-tion to afford both "warm" and "cold" white light in such haloplumbate-based wide-band gap semicon-ductors[14]. The 1D iodoplumbate-based hybrids were reported to display ferroelectricity, wherein the polarization is switchable under an alternating current electrical field[24].
In addition, a 3D open-framework hybrid, {(EDAMP)2[Pb7I18]·4H2O}n (EDAMP2+=Et2NHC6H4-CH2C6H4NHEt2), in which the inorganic framework is built from purely octahedral PbI6 units and behaves as a quantum-wire array, shows a fascinating wavelength-dependent photochromic behavior[25]. Its color changes from yellow to olive green under illumination by light with λ=500 nm and further to dark green by light with λ < 500 nm. Most interestingly, the reversion of the color for the hybrid can be accomplished by heating, indicating that this hybrid possesses switchable photochromic nature. It is well known that a material with switchable functionality through external stimuli, such as thermally-triggered, irradiation-induced and applied pressure, is very useful for application in the fields of sensors, memory and data storage[26-29].
In this study, we report the syntheses and crystal structures of two haloplumbate-based hybrids, {(4-CH3-Bz-4-Ph-Py)[PbBr3]}n (1) and {(4-F-Bz-4-Ph-Py)[PbBr3]}n (2).
1 Experimental
1.1 Materials and general methods
All chemicals and solvents were reagent grade and used without further purification. (4-CH3-Bz-4-Ph-Py)Br and (4-F-Bz-4-Ph-Py)Br were synthesized according to a similar procedure described in the literature[30]. Elemental analyses for C, H and N were performed with an Elementar Vario EL Ⅲ analytic instrument. Powder X-ray diffraction (PXRD) data for 1 and 2 were collected on a Rigaku/max-2550 diffra-ctometer with Cu Kα radiation (λ=0.154 18 nm) at room temperature. The acceleration voltage was 40 kV with a 40 mA current flux. The data were collected in the 2θ range from 5° to 50°.
1.2 Synthesis of {(4-CH3-Bz-4-Ph-Py)[PbBr3]}n (1)
A mixture of (4-CH3-Bz-4-Ph-Py)Br, KBr and PbBr2 with a molar ratio of 1:1:1 in DMF (25 mL) was placed in an oven and slowly evaporated at 55 ℃ for 10~14 days to produce light yellow needle-shaped crystals in ca. 95% yield. Elemental analysis calculated for C38H36N2Pb3Br8(%):C 25.62, H 2.04, N 1.57; Found(%): C 25.60, H 2.03, N 1.55.
1.3 Synthesis of {(4-F-Bz-4-Ph-Py)[PbBr3]}n (2)
The synthesis of 2 was the same as that of 1 except that (4-F-Bz-4-Ph-Py)Br was used instead of (4-CH3-Bz-4-Ph-Py)Br. Yellow needle-shaped crystals were gained in ca. 95% yield. Elemental analysis calculated for C36H30N2Pb3Br8F2(%): C 24.16, H 1.69, N 1.57; Found(%): C 24.11, H 1.65, N 1.54.
1.4 X-ray crystallography
Two block single crystals of 1 and 2 with dimen-sions of both 0.19 mm×0.18mm×0.17mm were selected under an optical microscope and glued to thin glass fibers, respectively. X-ray diffraction intensity data were collected on a Bruker APEX ⅡCCD diffra-ctometer equipped with a graphite monochromated Mo Kα (λ=0.071 073 nm) using the φ-ω scan mode at 296(2) K. Data reductions and absorption corrections were performed with the SAINT and SADABS software packages[31], respectively. Structures were solved by direct methods and refined by full matrix least-squares with SHELXL-2014/7 software package[32]. The non-hydrogen atoms were anisotropically refined using the full-matrix least-squares method on F2. All hydrogen atoms were placed at the calculated positions and refined riding on the parent atoms. The details about data collection, structure refinement and crystallo-graphy are summarized in Table 1.
Compound 1 2 Formula C38H36N2Pb3Br8 C36H30N2Pb3Br8F2 Formula weight 1 781.54 1 911.29 Space group P21212 P21212 Crystal system Orthorhombic Orthorhombic a / nm 2.101 3(2) 2.137 9(2) b / nm 2.349 6(3) 2.286 2(2) c / nm 0.442 11(5) 0.435 81(4) V /nm3 2.182 8(4) 2.130 1(3) Z 2 2 Dc / (g·cm-3) 2.711 2.980 F(000) 1 608 1 712 μ / mm-1 18.893 19.368 range for data collection / (°) 1.73-27.66 1.30-27.44 Index ranges -23 ≤ h ≤27, -30 ≤ k ≤ 30, -5 ≤ l ≤ 5 -27 ≤h ≤ 27, -28 ≤ k ≤ 29, -5 ≤ l ≤ 5 Rint 0.086 7 0.036 3 Flack 0.340(13) 0.453(10) Independent reflection, restraint, parameter 5 045, 0, 105 4 848, 0, 103 Goodness of fit on F2 1.059 1.061 R1, wR2a [I > 2σ(I)] R1=0.062 7, wR2=0.163 7 R1=0.054 8, wR2=0.147 3 R1, wR2a [all data] R1=0.084 7, wR2=0.178 7 R1=0.064 8, wR2=0.159 2 (Δρ)max, (Δρ)min / (e·nm-3) 4 402, -4 931 4 416, -3 827 a R1=∑||Fo|-|Fc||/|Fo|, wR2=[∑w(∑Fo2-Fc2)2/∑w(Fo2)2]1/2 CCDC: 1572941, 1; 1572942, 2.
2 Results and discussion
2.1 Description of crystal structure
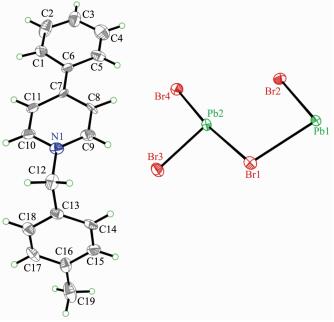
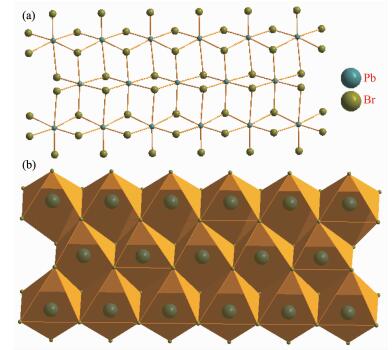
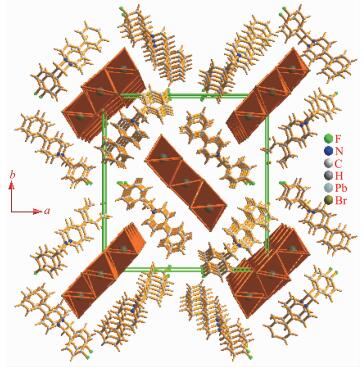
Compound 1 crystallizes in the P21212 space group at room temperature and no chiral separation was done. The asymmetric unit, as shown in Fig. 1, consists of one Pb2+ ion and three different Br- anions together with one 4-CH3-Bz-4-Ph-Py+ cation. The Pb2+ ion is located at an inversion center and coordinated with six Br- to form the slightly distorted PbI6 octahe-dron. The Pb-Br lengths range from 0.271 38(23) to 0.349 15(20) nm and the Br-Pb-Br angles fall within the range of 83.754(46)°~172.861(50)° at 296 K, these geometry parameters within the coordination octahed-ron are comparable to other haloplumbates. Three different Br- ions are adopted in the μ3-bridged model to connect three neighboring Pb2+ ions. The adjacent PbBr6 coordination octahedral are connected together via the edge-sharing mode to form a uniform [Pb3Br9]n chain along the a-axis direction (Fig. 2).
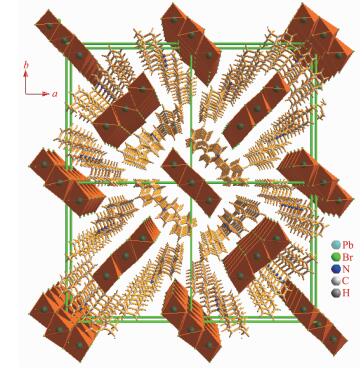
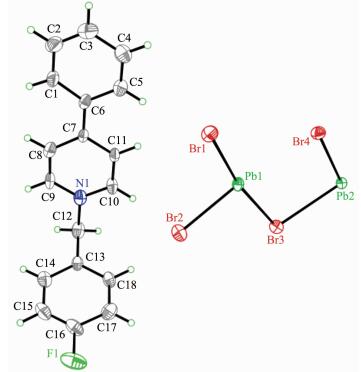
The cation is composed of a 4-phenylpyridine and a 4-methylbenzyl, and the neighboring cations are aligned into quadrilateral-shaped 1D channels, and the inorganic [Pb3Br9]n chains reside in the channels (Fig. 3). Charged-assisted H-bonding interactions appear between the CH2 groups in the cations and the Br-ions in the inorganic chains. Compound 2 (Fig. 4~5) is isostructural with compound 1.
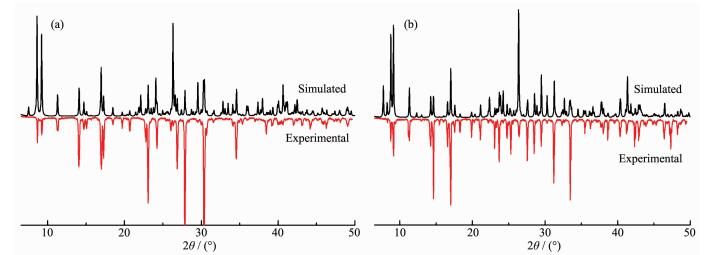
2.2 Powder X-ray diffraction (PXRD)
Powder X-ray diffraction (PXRD) analyses were carried out for 1 and 2 at room temperature to characterize their purity. As shown in Fig. 6, the measured peak positions closely match the simulated peak positions, indicative of pure products.
-
-
[1]
Zhao S P, Ren X M. Dalton Trans., 2011, 40:8261-8272 doi: 10.1039/c0dt01806f
-
[2]
Krautscheid H, Vielsack. Angew. Chem. Int. Ed., 1995, 34:2035-2037 doi: 10.1002/(ISSN)1521-3773
-
[3]
Krautscheid H, Lode C, Vielsack F, et al. J. Chem. Soc. Dalton Trans., 2001:1099-1104
-
[4]
Tang Z, Guloy A M. J. Am. Chem. Soc., 1999, 121:452-453 doi: 10.1021/ja982702i
-
[5]
Liu J J, Guan Y F, Jiao C, et al. Dalton Trans., 2015, 44:5957-5960 doi: 10.1039/C4DT03785E
-
[6]
Zhang Z J, Xiang S C, Zhang Y F, et al. Inorg. Chem., 2006, 45:1972-1977 doi: 10.1021/ic051350v
-
[7]
She Y J, Zhao S P, Ren X M, et al. Inorg. Chem. Commun., 2014, 46:29-32 doi: 10.1016/j.inoche.2014.04.030
-
[8]
Tong Y B, Ren L T, Ren X M, et al. Dalton Trans., 2015, 44:17850-17858 doi: 10.1039/C5DT02739J
-
[9]
Duan H B, Yu S S, Ren X M, et al. Dalton Trans., 2016, 45:4810-4818 doi: 10.1039/C5DT04594K
-
[10]
Lemmerer A, Billing D G. CrystEngComm, 2012, 14:1954-1966 doi: 10.1039/c2ce06498g
-
[11]
Willett R D, Maxcy K R, Twamley B. Inorg. Chem., 2002, 41:7024-7030 doi: 10.1021/ic020455k
-
[12]
SUN Cai(孙财), WANG Ming-Sheng(王明盛), GUO Guo-Cong(郭国聪). Proceedings of 9th Chinese Inorganic Chem-istry Conference(中国化学会第九届全国无机化学学术会议论文集). Nanchang: [s. n. ], 2015.
-
[13]
福建物质结构研究所.硅酸盐通报, 2016, 2:477Fujian Institute of Research on the Structure of Matter. Bulletin of the Chinese Ceramic Society, 2016, 2:477
-
[14]
Dohner E R, Hoke E T, Karuadasa H I. J. Am. Chem. Soc., 2014, 136:1718-1721 doi: 10.1021/ja411045r
-
[15]
Wehrenfenging C, Liu M, Snaith H J, et al. J. Phys. Chem. Lett., 2014, 5:1300-1306 doi: 10.1021/jz500434p
-
[16]
Guloy A M, Tang Z J, Miranda P B, et al. Adv. Mater., 2001, 13:833-837 doi: 10.1002/1521-4095(200106)13:11<833::AID-ADMA833>3.0.CO;2-T
-
[17]
Fujisawa J I, Ishihara T. Phys. Rev. B:Condens. Matter., 2004, 70:113203 doi: 10.1103/PhysRevB.70.113203
-
[18]
Liu G N, Shi J R, Han X J, et al. Dalton Trans., 2015, 44:12561-12575 doi: 10.1039/C5DT00687B
-
[19]
陆新荣, 赵颖, 刘建, 等.无机化学学报, 2015, 31(9):1678-1686 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?file_no=20150904&flag=1LU Xing-Rong, ZHAO Ying, LIU Jian, et al. Chinese J. Inorg. Chem., 2015, 31(9):1678-1686 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?file_no=20150904&flag=1
-
[20]
Yang W S, Park B W, Jung E H, et al. Science, 2017, 356:1376-1379 doi: 10.1126/science.aan2301
-
[21]
Liu M, Johnston M B, Snaith H J. Nature, 2013, 501:395-398 doi: 10.1038/nature12509
-
[22]
Xing G, Mathews N, Sun S, et al. Science, 2013, 18:344-347
-
[23]
Yoo E J, Lyu M, Yun J H, et al. Adv. Mater., 2015, 27:6170-6175 doi: 10.1002/adma.201502889
-
[24]
Zhao H R, Li D P, Ren X M, et al. J. Am. Chem. Soc., 2010, 132:18-19 doi: 10.1021/ja907562m
-
[25]
Zhang Z J, Xiang S C, Guo G C, et al. Angew. Chem., Int. Ed., 2008, 47:4149-4152 doi: 10.1002/(ISSN)1521-3773
-
[26]
Maldonado P, Kanungo S, Saha-Dasgupta T, et al. Phys. Rev. B:Condens. Matter., 2013, 88:020408 doi: 10.1103/PhysRevB.88.020408
-
[27]
Kahn O, Martinez C J. Science, 1999, 279:44-48
-
[28]
Liang J, Chen Z, Xu L, et al. J. Mater. Chem. C, 2014, 2:2243-2250 doi: 10.1039/c3tc31638f
-
[29]
Dong X Y, Li B, Ma B B, et al. J. Am. Chem. Soc., 2013, 135:10214-10217 doi: 10.1021/ja403449k
-
[30]
袁国军, 刘光祥, 时超, 等.无机化学学报, 2017, 33(10):1855-1860 doi: 10.11862/CJIC.2017.225YUAN Guo-Jun, LIU Guang-Xiang, SHI Chao, et al. Chinese J. Inorg. Chem., 2017, 33(10):1855-1860 doi: 10.11862/CJIC.2017.225
-
[31]
SMART and SAINT, Siemens Analytical X-ray Instrument Inc., Madison, WI, 1996.
-
[32]
Sheldrick G M. Acta Crystallogr. Sect. C:Cryst. Struct. Commun., 2015, C71:3-8
-
[1]
-
Table 1. Crystallographic and structure refinement data for 1 and 2
Compound 1 2 Formula C38H36N2Pb3Br8 C36H30N2Pb3Br8F2 Formula weight 1 781.54 1 911.29 Space group P21212 P21212 Crystal system Orthorhombic Orthorhombic a / nm 2.101 3(2) 2.137 9(2) b / nm 2.349 6(3) 2.286 2(2) c / nm 0.442 11(5) 0.435 81(4) V /nm3 2.182 8(4) 2.130 1(3) Z 2 2 Dc / (g·cm-3) 2.711 2.980 F(000) 1 608 1 712 μ / mm-1 18.893 19.368 range for data collection / (°) 1.73-27.66 1.30-27.44 Index ranges -23 ≤ h ≤27, -30 ≤ k ≤ 30, -5 ≤ l ≤ 5 -27 ≤h ≤ 27, -28 ≤ k ≤ 29, -5 ≤ l ≤ 5 Rint 0.086 7 0.036 3 Flack 0.340(13) 0.453(10) Independent reflection, restraint, parameter 5 045, 0, 105 4 848, 0, 103 Goodness of fit on F2 1.059 1.061 R1, wR2a [I > 2σ(I)] R1=0.062 7, wR2=0.163 7 R1=0.054 8, wR2=0.147 3 R1, wR2a [all data] R1=0.084 7, wR2=0.178 7 R1=0.064 8, wR2=0.159 2 (Δρ)max, (Δρ)min / (e·nm-3) 4 402, -4 931 4 416, -3 827 a R1=∑||Fo|-|Fc||/|Fo|, wR2=[∑w(∑Fo2-Fc2)2/∑w(Fo2)2]1/2 -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 5
- 文章访问数: 1740
- HTML全文浏览量: 295


 下载:
下载:





 下载:
下载:

