
2-(5-甲基-1, 3, 4-噻二唑)-硫乙酸锌(Ⅱ)配合物的合成、晶体结构和性质
-
关键词:
- 2-(5-甲基-1, 3, 4-噻二唑)-硫乙酸
- / 晶体结构
- / π-π相互作用
- / 氢键
English
Syntheses, Crystal Structures and Properties of 2-(5-Methyl-1, 3, 4-thiadiazol-2-ylthio Zinc(Ⅱ)Coordination Compounds
-
0 Introduction
The construction of supramolecular architectures are currently of great interest owing to their intriguing structures[1-4] and potential applications in catalysts, electrochemistry, optics, magnetism, gas sorption and so on[5-9]. However, how to construct appropriate crystal structures is the primary issue. The carboxylate ligands are widely employed in the design of coordination frameworks for the following reasons: Firstly, because their various coordination modes and flexible molecular backbones can provide a variety of coordination polymers with appealing structures and properties[10]. Secondly, the inherent negative charge of the carboxylate groups can compensate for the charge induced by the metal centers[11-12]. Thirdly, hydrogen bond formation by carboxylate groups reinforces whole architectures[13-15]. On the other hand, the N-donor ligands, such as 4, 4′-bipyridine (4, 4′-bipy) and 1, 2-bis(4-pridyl)ethane (bpe) have also been proven to be useful connectors to formulate high dimensional compounds[16]. Recently, we have successfully synthes-ized some supramolecular compounds by using 2-(5-methyl-1, 3, 4-thiadiazol-2-ylthio) acetic acid(Hmtyaa) ligand[17-19]. Herein, we successfully obtained two new coordination compound based on Hmtyaa ligand and N-donor ligands (bpe), namely, [Zn(mtyaa)2(H2O)4]·4H2O (1) and [Zn(bpe)(mtyaa)2(H2O)2]n (2).
1 Experimental
1.1 Reagents and physical measurements
Hmtyaa ligand was synthesized as literature reported[20]. All other chemicals were of reagent grade quality from commercial sources and were used without further purification. The IR absorption spectra of the compounds were recorded in the range of 400~4 000 cm-1 by means of a Nicolet (Impact 410) spec-trometer with KBr pellets (5 mg of sample in 500 mg of KBr). C, H and N analyses were carried out with a Perkin Elmer 240C elemental analyser. XRD measure-ments were performed on a Philips X′pert MPD Pro X-ray diffractometer using Cu Kα radiation (λ=0.154 18 nm, 2θ=5°~50°), in which the X-ray tube was operated at 40 kV and 40 mA. The as-synthesized samples were characterized by thermogravimetric analysis (TGA) on a Perkin Elmer thermogravimetric analyser Pyris 1 TGA up to 1 023 K using a heating rate of 20 K·min-1 under a N2 atmosphere.
1.2 Synthesis
1.2.1 Synthesis of [Zn(mtyaa)2(H2O)4]·4H2O (1)
A solution of ZnCl2 (54 mg, 0.4 mmol) in H2O (3 mL) was added to a solution of Hmtyaa (38 mg, 0.2 mmol) in H2O (5 mL) which was adjusted to pH≈7.0 with dilute sodium hydroxide (1 mol·L-1), and the mixture was stirred at room temperature for 12 h to give a colorless solution, which was then filtered. Slow evaporation of the filtrate gave white block-shaped single crystals in two weeks. Yield: 39.98 mg (68%). Anal. Calcd. for C10H26N4O12S4Zn(%): C, 20.43; H, 4.46; N, 9.53. Found(%): C, 20.45; H, 4.47; N, 9.51. IR (KBr, cm-1): 3 487(s), 3 342(s), 1 589(vs), 1 401(m), 1 388(vs), 1 248(m), 1 206(w), 1 093(w), 904(w), 781(w), 689(m), 611(m), 405(w).
1.2.2 Synthesis of [Zn(bpe)(mtyaa)2(H2O)2]n (2)
A solution of ZnCl2 (27 mg, 0.2 mmol) and bpe (36 mg, 0.2 mmol) was added to a solution of Hmtyaa (38 mg, 0.2 mmol) in H2O (7 mL) which was adjusted to pH≈7.0 with dilute sodium hydroxide (1 mol·L-1). The final mixture was sealed in a 15 mL PTFE-lined stainless-steel acid digestion bomb and heated at 75 ℃ for 15 h. Little red crystals were obtained and filtered. The filtrate was allowed to slowly evaporate at ambient temperature over 3 days, and quantities of white triangular prism crystals were collected in 95% yield. Anal. Calcd. for C22H26N6O6S4Zn(%): C, 39.79; H, 3.95; N, 12.65. Found(%): C, 39.81; H, 3.96; N, 12.62. IR (KBr, cm-1): 3 427(s), 1 615(vs), 1 590(vs), 1 428(m), 1 384(vs), 1 219(w), 1 198(w), 1 074(m), 826(w), 685(w), 621(w), 545(w).
1.3 X-ray crystallography
X-ray crystallographic data of both compounds were collected at room temperature using epoxy-coated crystals mounted on glass fiber. All measurements were made on a Smart CCD diffractometer with graphite-monochromated Mo Kα radiation (λ=0.071 073 nm) by using φ-ω scan mode at room temperature. The struc-tures were solved by direct methods and refined with the full-matrix least squares technique using the SHELXS-97 and SHELXL-97 programs[21-22]. Anisotropic thermal parameters were assigned to all non-hydrogen atoms. The hydrogen atoms bonding to C and N atoms were fixed geometrically at calculated distances and allowed to ride on the parent atoms, and the hydrogen atoms of water molecules were either found from the difference Fourier map or fixed stereochemically. Crystallographic data and structural refine ment parameters are summarized in Table 1, and the selected bond lengths and angles are given in Table 2 and 3.
Compound 1 2 Formula C10H26N4O12S4Zu C22H26N6O6S4Zu Formula weight 587.96 664.10 Temperature/K 293(2) 293(2) Crystal system Triclinic Monoclinic Space group P1 C2/c a / nm 0.675 5(2) 1.93301(5) b / nm 0.859 2(3) 1.203 4(3) c / nm 1.063 3(3) 1.489 1(4) α/(°) 96.452(5) 90 β/(°) 100.038(5) 125.713(4) γ/(°) 103.392(5) 90 V / nm3 0.583 6(3) 2.812 7(12) Z 1 4 Crystal size/mm 0.26×0.24×0.23 0.32×0.28×0.26 Dc / (g·cm-3) 1.673 1.568 μ/mm-1 1.471 1.219 F(000) 304 1 368 θ range for data collection / (°) 1.97~25.00 2.13~25.00 Reflection collected, unique 2 915, 2 020 (Rint=0.078 3) 6 848, 2 467 (Rint=0.067 8) Observed reflections [I>2σ(I)] 1 794 2 051 Parameter refined 142 179 Goodness-of-fit on F2 1.002 1.002 Final R indices [I>2σ(I)] R1=0.045 2; wR2=0.125 42 R1=0.037 8; wR2=0.095 3 (Δρ)max, (Δρ)min/(e·nm-3) 671, -657 425, -369 Compound 1 O2-Zn1 0.203 8(2) O3-Zn1 0.217 0(2) O4-Zn1 0.206 5(2) O2a-Zn1-O2 180 O2-Zn1-O4a 90.63(10) O2-Zn1-O4 89.37(10) O4a-Zn1-O4 180 O2-Zn1-O3 90.49(10) O4-Zn1-O3 88.86(10) O2-Zn1-O3a 89.51(10) O4-Zn1-O3a 91.14(10) O3-Zn1-O3a 180 Compound 2 N3-Zn1 0.216 2(2) O2-Zn1 0.2152 2(19) O3-Zn1 0.211 54(18) O3-Zn1-O3a 180 O3-Zn1-O2 90.87(7) O3a-Zn1-O2 89.13(7) O2-Zn1-O2a 180 O3-Zn1-N3 92.06(8) O3a-Zn1-N3 87.94(8) O2-Zn1-N3 89.58(8) O2a-Zn1-N3 90.42(8) O3a-Zn1-N3a 92.06(8) N3-Zn1-N3a 180 Symmetry codes: a: -x+1, -y+2, -z+2 for 1; a: -x+1/2, -y+3/2, -z+1 for 2. D-H…A d(D-H) / nm d(H…A)/nm d(D…A) / nm ∠DHA / (°) Compound 1 O6-H6A...N2 0.085 0.225 0.286 6(4) 129.2 O6-H6B...O3a 0.085 0.241 0.283 1(4) 111.3 O5-H5B...O1a 0.085 0.217 0.290 0(5) 143.3 O6-H6B...O4b 0.085 0.254 0.338 8(4) 173.5 O4-H4B...N1b 0.085 0.203 0.286 1(4) 168.5 O4-H4A...O1c 0.085 0.195 0.271 8(4) 150.9 O3-H3A...O5d 0.085 0.194 0.275 0(5) 158.3 O4-H4A...Sle 0.085 0.300 0.349 4(3) 119.2 O3-H3В...O1e 0.085 0.200 0.282 6(4) 164.1 Compound 2 O3-H3B...O1 0.095 0.170 0.260 3(3) 158.4 O3-H3A...N1a 0.093 0.189 0.282 1(3) 175.7 Symmetry codes: a: x, y, z-1; b: -x+1, -y+2, -z+1; c:-x+1, -y+2, -z+2; d: -x+1, -y+1, -z+1; e: x-1, y, z for 1; a: x, -y+1, z-1/2 for 2. CCDC: 653462, 1; 653467, 2.
2 Results and discussion
2.1 Structure description of compounds 1 and 2
2.1.1 Crystal structure of [Zn(mtyaa)2(H2O)4]·4H2O (1)
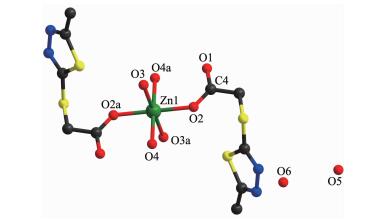
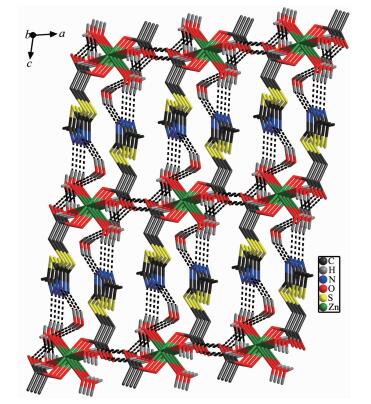
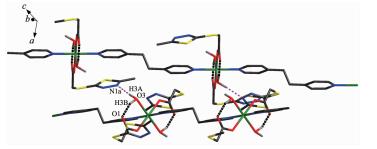
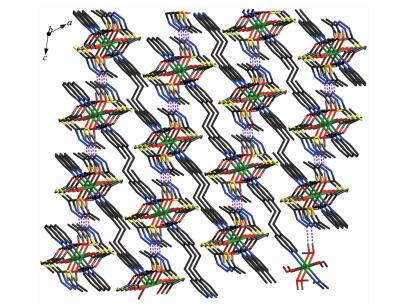
Each Zn(Ⅱ) ion is six coordinated and exhibits a distorted octahedral geometry. Two oxygens O2 and O2a from two ligands and two coordinated water donor O4 and O4a form the equatorial plane with an average length of 0.205 17 nm and an average O2-Zn1-O4 angle of 90.00(1)°, and the other two water molecules O3, O3a, occupy axial positions with an average length of 0.217 0(2) nm and the O3-Zn1-O3a angle of 180° (Fig. 1). As depicted in Fig. 1, mtyaa ligand acts as a monodentate ligand. Numerous hydrogen-bonding interactions occurred such as O(water)…O(carboxyl), O(water)…O(water), O(water)…N and O(water)…S, connect compound 1 into a 2D structure network (Fig. 2). The center-to-center separation for the parallel-arranged ring are 0.354 and 0.344 nm, respectively, indicating the presence of strong face-to face π-π stacking interactions in compound 1, which further stabilize the 2D crystal structure. The two dimensional planar is further connected into three dimensional network by hydrogen bonds (Fig. 3).
2.1.2 Crystal Structure of [Zn(bpe)(mtyaa)2(H2O)2]n (2)
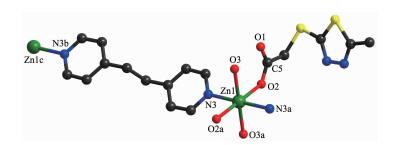
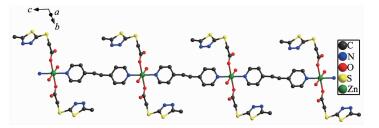
As illustrated in Fig. 4, in compound 2, each Zn(Ⅱ) is six coordinated and exhibits a distorted octahedral geometry. Two carboxylate oxygens O2 and O2a from two mtyaa ligands and two coordinated water molecules O3 and O3a form the equatorial plane with an average length of 0.213 38(18) nm and the average angle of O(2)-Zn1-O(3) is 90.00°. Two nitrogen molecules N3 and N3a from two bpe ligands occupy axial positions with the length of 0.216 2(2) nm and the N3-Zn1-N3a angle of 180°. The adjacent Zn(Ⅱ) ions are bridged into one dimensional chain structure by nitrogen atoms coming from mtyaa ligands (Fig. 5). Numerous hydrogen-bonding interactions such as O(water)…O(carboxyl) and O(water)…N, connect the adjacent chains into three dimensional network (Fig. 6 and 7).
2.2 Thermal and XRD analysis
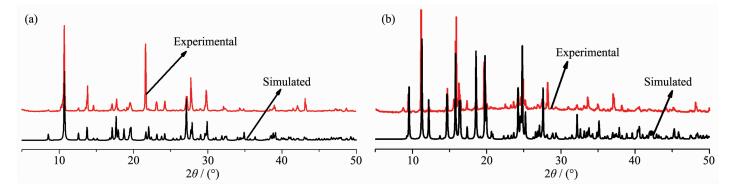
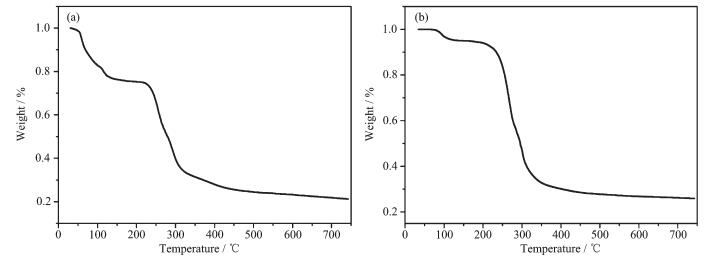
To confirm whether the crystal structures are truly representative of the bulk materials, XRD experiments were carried out for 1 and 2. The XRD experimental and computer-simulated patterns of the corresponding compounds are shown in Fig. 8, which show that the bulk synthesized materials and the measured single crystals are the same. In order to investigate the thermal stability of the complex, their thermal behaviours were studied by TGA (Fig. 9). For compound 1, a rapid weight loss is observed from 30 to 159 ℃, which is attributed to loss of the coordinated water molecules and free water, with a weight loss of 24.28% (Calcd. 24.49%). The TGA curve of 2 shows that 2 undergoes dehydration between 78 and 135 ℃, which is attributed to loss of the lattice water molecules, with a weight loss of 5.28% (Calcd. 5.42%). The decomposition of the anhydrous residue of 1 and 2 occurs at 221 and 198 ℃, respectively.
3 Conclusions
In summary, by using N-donor ligand (bpe) and Hmtyaa ligand, we have synthesized two new coor-dination compounds [Zn(mtyaa)2(H2O)4]·4H2O (1) and [Zn(bpe)(mtyaa)2(H2O)2]n (2). Both compounds are connected into three dimensional network by hydrogen bonds between coordinated and free water molecules with carboxylate oxygen atoms.
-
-
[1]
Qian J J, Jiang F L, Yuan D Q, et al. Chem. Commun., 2012, 48:9696-9698 doi: 10.1039/c2cc35068h
-
[2]
Van de Voorde B, Bueken B, Denayer J, et al. Chem. Soc. Rev., 2014, 43:5766-5788 doi: 10.1039/C4CS00006D
-
[3]
Lei C, Chao S T, Wu H K, et al. Z. Kristallogr.-New Cryst. Struct., 2016, 231:405-406
-
[4]
Yu J C, Cui Y J, Wu C D, et al. J. Am. Chem. Soc., 2015, 137:4026-4029 doi: 10.1021/ja512552g
-
[5]
Xu Z, Han L, Zhuang L, et al. Inorg. Chem., 2015, 54:4737-4743 doi: 10.1021/acs.inorgchem.5b00110
-
[6]
Banerjee D, Hu Z C, Li J. Dalton Trans., 2014, 43:10668-10685 doi: 10.1039/C4DT01196A
-
[7]
Wang M S, Guo S P, Li Y, et al. J. Am. Chem. Soc., 2009, 131:13572-13573 doi: 10.1021/ja903947b
-
[8]
Jariwala D, Sangwan V K, Lauhon L J, et al. Chem. Soc. Rev., 2013, 42:2824-2860 doi: 10.1039/C2CS35335K
-
[9]
Getman R B, Bae Y S, Wilmer C E, et al. Chem. Rev., 2012, 112:703-723 doi: 10.1021/cr200217c
-
[10]
Chu Q, Su Z, Fan J, et al. Cryst. Growth Des., 2011, 11:3885-3894 doi: 10.1021/cg200416y
-
[11]
Liu Y H, Lu Y L, Wu H C, et al. Inorg. Chem., 2002, 41:2592-2597 doi: 10.1021/ic011192h
-
[12]
Wei Y L, Hou H W, Li L K, et al. Cryst. Growth Des., 2005, 5:1405-1413 doi: 10.1021/cg049596i
-
[13]
Desiraju G R. Crystal Engineering: the Design of Organic Solids. Amsterdam: Elsevier, 1989.
-
[14]
Desiraju G R, Steiner T. Weak Hydrogen Bond in Structural Chemistry and Biology. Oxford:Oxford University Press, 1999.
-
[15]
Desiraju G R. Angew. Chem. Int. Ed., 1995, 34:2311-2327 doi: 10.1002/(ISSN)1521-3773
-
[16]
Ahmad M, Sharma M K, Das R, et al. Cryst. Growth Des., 2012, 12:15711578
-
[17]
Ma M H, Pan Z R, Xu J, et al. Chin. J. Struct. Chem., 2010, 29(6):843-852
-
[18]
潘兆瑞, 周宏, 鲜华.无机化学学报, 2010, 26(11):1955-1960 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?flag=1&file_no=20101108&journal_id=wjhxxbcnPAN Zhao-Rui, ZHOU Hong, XIE Hua. Chinese J. Inorg. Chem., 2010, 26(11):1955-1960 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?flag=1&file_no=20101108&journal_id=wjhxxbcn
-
[19]
潘兆瑞.无机化学学报, 2010, 27(10):2027-2032 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?flag=1&file_no=20111022&journal_id=wjhxxbcnPAN Zhao-Rui. Chinese J. Inorg. Chem., 2010, 27(10):2027-2032 http://www.wjhxxb.cn/wjhxxbcn/ch/reader/view_abstract.aspx?flag=1&file_no=20111022&journal_id=wjhxxbcn
-
[20]
Srogl J, Liu W S, Marshall D, et al. J. Am. Chem. Soc., 1999, 121:9449-9450 doi: 10.1021/ja991654e
-
[21]
Sheldrick G M. Acta Crystallogr. Sect. A, 1990, A46:457
-
[22]
Sheldrick G M. SHELXL-97, Program for Crystal Structure Refinement, University of Göttingen, Germany, 1997.
-
[1]
-
Table 1. Crystallographic data and structure refinement details for compounds 1 and 2
Compound 1 2 Formula C10H26N4O12S4Zu C22H26N6O6S4Zu Formula weight 587.96 664.10 Temperature/K 293(2) 293(2) Crystal system Triclinic Monoclinic Space group P1 C2/c a / nm 0.675 5(2) 1.93301(5) b / nm 0.859 2(3) 1.203 4(3) c / nm 1.063 3(3) 1.489 1(4) α/(°) 96.452(5) 90 β/(°) 100.038(5) 125.713(4) γ/(°) 103.392(5) 90 V / nm3 0.583 6(3) 2.812 7(12) Z 1 4 Crystal size/mm 0.26×0.24×0.23 0.32×0.28×0.26 Dc / (g·cm-3) 1.673 1.568 μ/mm-1 1.471 1.219 F(000) 304 1 368 θ range for data collection / (°) 1.97~25.00 2.13~25.00 Reflection collected, unique 2 915, 2 020 (Rint=0.078 3) 6 848, 2 467 (Rint=0.067 8) Observed reflections [I>2σ(I)] 1 794 2 051 Parameter refined 142 179 Goodness-of-fit on F2 1.002 1.002 Final R indices [I>2σ(I)] R1=0.045 2; wR2=0.125 42 R1=0.037 8; wR2=0.095 3 (Δρ)max, (Δρ)min/(e·nm-3) 671, -657 425, -369 Table 2. Selected bond lengths (nm) and angles (°) in compounds 1 and 2
Compound 1 O2-Zn1 0.203 8(2) O3-Zn1 0.217 0(2) O4-Zn1 0.206 5(2) O2a-Zn1-O2 180 O2-Zn1-O4a 90.63(10) O2-Zn1-O4 89.37(10) O4a-Zn1-O4 180 O2-Zn1-O3 90.49(10) O4-Zn1-O3 88.86(10) O2-Zn1-O3a 89.51(10) O4-Zn1-O3a 91.14(10) O3-Zn1-O3a 180 Compound 2 N3-Zn1 0.216 2(2) O2-Zn1 0.2152 2(19) O3-Zn1 0.211 54(18) O3-Zn1-O3a 180 O3-Zn1-O2 90.87(7) O3a-Zn1-O2 89.13(7) O2-Zn1-O2a 180 O3-Zn1-N3 92.06(8) O3a-Zn1-N3 87.94(8) O2-Zn1-N3 89.58(8) O2a-Zn1-N3 90.42(8) O3a-Zn1-N3a 92.06(8) N3-Zn1-N3a 180 Symmetry codes: a: -x+1, -y+2, -z+2 for 1; a: -x+1/2, -y+3/2, -z+1 for 2. Table 3. Hydrogen-bonding parameters in compounds 1 and 2
D-H…A d(D-H) / nm d(H…A)/nm d(D…A) / nm ∠DHA / (°) Compound 1 O6-H6A...N2 0.085 0.225 0.286 6(4) 129.2 O6-H6B...O3a 0.085 0.241 0.283 1(4) 111.3 O5-H5B...O1a 0.085 0.217 0.290 0(5) 143.3 O6-H6B...O4b 0.085 0.254 0.338 8(4) 173.5 O4-H4B...N1b 0.085 0.203 0.286 1(4) 168.5 O4-H4A...O1c 0.085 0.195 0.271 8(4) 150.9 O3-H3A...O5d 0.085 0.194 0.275 0(5) 158.3 O4-H4A...Sle 0.085 0.300 0.349 4(3) 119.2 O3-H3В...O1e 0.085 0.200 0.282 6(4) 164.1 Compound 2 O3-H3B...O1 0.095 0.170 0.260 3(3) 158.4 O3-H3A...N1a 0.093 0.189 0.282 1(3) 175.7 Symmetry codes: a: x, y, z-1; b: -x+1, -y+2, -z+1; c:-x+1, -y+2, -z+2; d: -x+1, -y+1, -z+1; e: x-1, y, z for 1; a: x, -y+1, z-1/2 for 2. -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 8
- 文章访问数: 990
- HTML全文浏览量: 88



 下载:
下载:








 下载:
下载:

