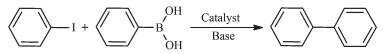
 图Scheme 1
Representative Suzuki-Miyaura cross-coupling reaction
Scheme1.
Representative Suzuki-Miyaura cross-coupling reaction
图Scheme 1
Representative Suzuki-Miyaura cross-coupling reaction
Scheme1.
Representative Suzuki-Miyaura cross-coupling reaction

具有金属-金属作用单核自组装体的合成及其对Suzuki偶联反应的高效催化
-
关键词:
- 金属-金属作用
- / 自组装
- / 催化
- / Suzuki偶联反应
English
Mononuclear Assemblies with Metal-Metal Interaction: Syntheses and Catalytical Performance in Suzuki-Coupling Reaction
-
Key words:
- metal-metal interaction
- / self-assembly
- / catalysis
- / Suzuki-coupling reaction
-
Spontaneous and precise assembly of compounds into giant, well-defined, functional superstructures are attractive for their novel structures[1-4] and promising applications in molecular recognition, catalysis, guest inclusion, luminescence, anion complexation and so on[5-9]. Over the last decade, numerous novel metal-organic molecules have been constructed by metal-directed self-assembly[10-14]. The Fujita group has estab-lished a series of complexes that can be self-assembled by simply mixing ligands and bare square-planar Pd(Ⅱ) ions[15-16]. And in our previous research, we have reported an array of well-defined metallic supramolecular structures formed by quantitatively assembling[17-18]. More recently, transition metals with specific coordination geometries have been employed for the rational design and construction of highly ordered supramolecular structures[19].
Owing to the fact that the aryl-aryl structure motif is an important building block in organic chemistry, the Suzuki reaction is widely applied in academic research as well as in industrial synthesis of fine chemicals and highly complex pharmaceuticals[20]. A representative Suzuki-Miyaura cross-coupling reac-tion is shown in Scheme 1. In the Suzuki reaction, Pd-based catalysts coordinated with organophosphorus ligands are frequently used in Suzuki-crossing reactions. Since the organophosphorus ligands are poisonous, from the environmental point of view, making the Suzuki reaction green is a continuous process pursued by organic chemists. In the past few years, considerable attention has been paid to functional metal-organic assemblies that show promise in catalysis with environment-friendly[21]. Especially, palladium and platinum were employed in the Suzuki coupling reactions for their high stability and remarkable efficiency[22].
 图Scheme 1
Representative Suzuki-Miyaura cross-coupling reaction
Scheme1.
Representative Suzuki-Miyaura cross-coupling reaction
图Scheme 1
Representative Suzuki-Miyaura cross-coupling reaction
Scheme1.
Representative Suzuki-Miyaura cross-coupling reaction
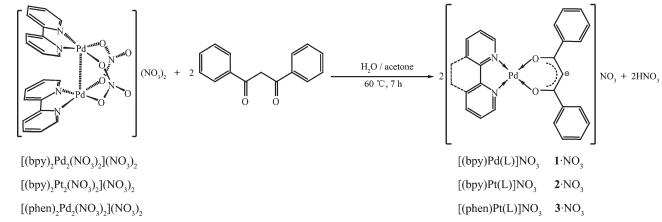
In this work, we designed and synthesized three mononuclear complexes using the self-assembly appr-oach, namely [(bpy)Pd(L)]NO3 (1·NO3·H2O), [(bpy)Pt(L)]NO3 (2·NO3·H2O), and [(phen)Pd(L)]NO3 (3·NO3·H2O), respectively. All of these three complexes have been intensively studied by NMR and ESI-MS, and X-ray single-crystal diffraction analysis have been empl-oyed for complex 1·PF6·CH3CN. In addition, consid-ering the structural characteristics and the palladium and platinum (Ⅱ, Ⅱ) properties, these three well-defined complexes have been developed and applied into Suzuki-coupling reactions, as expected, all of these three complexes show excellent catalysis properties.
1 Experimental
1.1 Materials and instruments
All chemicals for synthesis and analysis were obtained commercially with analytical grade and used without further purification. All solvents were of reagent pure grade and were purified according to conventional methods.
The ESI-MS were performed on a JEOL Accu-TOF mass spectrometer. 1H and 13C NMR spectra were performed on a Bruker AV 400 MHz spectrometer.
1.2 Syntheses and characterization of mononuclear complexes[23]
The self-assembly of mononuclear Pd complex 1·NO3·H2O was shown in Scheme 2. Ligand L (11.2 mg, 0.05 mmol) was treated with [(bpy)2Pd2(NO3)2](NO3)2 (19.3 mg, 0.025 mmol) in a mixture of water and acetone with 2:1 molar ratio. The mixture was stirred at 60 ℃ for 7 h to give 1·NO3·H2O. 1H NMR of 1·NO3·H2O (400 MHz, DMSO-d6, 298 K, TMS): δ 8.27 (m, J=7.9 Hz, 4H), 8.11 (t, J=7.7 Hz, 2H), 7.83 (d, J=7.5 Hz, 4H), 7.61 (m, J=7.4 Hz, 4H), 7.39 (t, J=7.8 Hz, 4H), 6.76 (s, 1H). 13C NMR for 1·NO3·H2O (400 MHz, DMSO-d6, 298 K, TMS): δ 181.32, 155.96, 147.28, 142.56, 135.15, 133.30, 129.30, 128.48, 124.62, 96.84, 49.07. ESI-MS (CH3CN, m/z): Calcd. for [(bpy)Pd(L)]+ 485.05, Found 485.01. Elemental analysis calculated for 1·NO3·H2O (C25H21N3O6Pd, %): C: 53.06, H: 3.74, N: 7.43. Found(%): C: 53.03, H: 3.76, N: 7.42. A ten-fold excess of KPF6 was added to the solution, the yellow precipitation were collected by centrifugation, washed with minimum amount of water and dried in vacuum to give 1·PF6·H2O as pale yellow solid (32.1 mg, 0.049 mmol, 97% yield). Single crystals of 1·PF6·CH3CN were obtained by the slow vapor diffusion of diethyl ether into their acetonitrile solutions over two weeks. The needle-shaped pale yellow crystals were collected by filtration, washed with water several times and dried in vacuum. 1H NMR of 1·PF6·H2O (400 MHz, DMSO-d6, 298 K, TMS): δ 8.38 (d, J=7.8 Hz, 2H), 8.35 (d, J=7.8 Hz, 2H), 8.19 (t, J=7.8 Hz, 2H), 7.93 (d, J=7.4 Hz, 4H), 7.70 (t, J=6.6 Hz, 2H), 7.62 (t, J=7.3 Hz, 2H), 7.43 (t, J=7.8 Hz, 4H), 6.86 (s, 1H). 13C NMR for 1·PF6 (400 MHz, DMSO-d6, 298 K, TMS): δ 181.14, 155.83, 147.13, 142.48, 134.98, 133.28, 129.24, 128.41, 124.57, 96.70, 31.15. ESI-MS (CH3CN, m/z): Calcd. for [(bpy)Pd(L)]+ 485.05, Found 485.03. Elemental analysis calculated for 1·PF6·H2O (C25H21F6N2O3PPd, %): C: 46.28, H: 3.26, N: 4.32. Found(%): C: 46.30, H: 3.30, N: 4.29. Elemental analysis calculated for 1·PF6·CH3CN (C27H22F6N3O2PPd, %): C: 48.27, H: 3.30, N: 6.25. Found(%): C: 48.25, H: 3.32, N: 6.23.
Ligand L (11.2 mg, 0.05 mmol) was treated with [(bpy)2Pt2(NO3)2](NO3)2 (23.7 mg, 0.025 mmol) in a mixture of water and acetone with 2:1 molar ratio at 60 ℃ for 7 h to give 2·NO3·H2O. 1H NMR of 2·NO3·H2O (400 MHz, DMSO-d6, 298 K, TMS): δ 8.64 (d, J=5.5 Hz, 2H), 8.42 (d, J=8.0, 2H), 8.24 (t, J=7.8 Hz, 2H), 8.03 (d, J=7.8 Hz, 4H), 7.73 (m, J=7.4 Hz, 4H), 7.49 (t, J=7.8 Hz, 4H), 6.94 (s, 1H). 13C NMR (400 MHz, DMSO-d6, 298 K, TMS): δ 178.37, 156.39, 146.50, 141.87, 134.79, 133.25, 129.39, 128.11, 124.78, 97.32, 49.07. ESI-MS (CH3CN, m/z): Calcd. for [[(bpy)Pt(L)]]+ 574.11, Found 574.07. Elemental analysis calculated for 2·NO3·H2O (C25H21N3O6Pt, %): C: 45.87, H: 3.23, N: 6.42. Found(%): C: 45.85, H: 3.26, N: 6.40. A ten-fold excess of KPF6 was added to the above solution, the yellow precipitation were collected by centrifuga-tion, washed with minimum amount of water and dried in vacuum to give 2·PF6·H·H2O as yellow solid (35.6 mg, 0.048 mmol, 95% yield). 1H NMR confirmed the quantitative formation of 2·PF6·H·H2O. 1H NMR (400 MHz, DMSO-d6, 298 K, TMS): δ 8.63 (d, J=5 Hz, 2H), 8.41 (d, J=7.8 Hz, 2H), 8.23 (t, J=7.8 Hz, 2H), 8.02 (d, J=7.4 Hz, 4H), 7.72 (m, 4H), 7.48 (t, J=7.9 Hz, 4H), 6.93 (s, 1H). 13C NMR (400 MHz, DMSO-d6, 298 K, TMS): δ 178.80, 156.66, 146.77, 142.06, 135.10, 133.29, 129.47, 128.32, 128.21, 124.87, 97.67. ESI-MS (CH3CN, m/z): Calcd. for [(bpy)Pt(L)]+ 574.11, Found 574.09. Elemental analysis calculated for 2·PF6·H·H2O (C25H21F6N2O3PPt, %): C: 40.71, H: 2.87, N: 3.80. Found: C: 40.69, H: 2.88, N: 3.78.
Ligand L (11.2 mg, 0.05 mmol) was treated with [(phen)2Pd2(NO3)2](NO3)2 (20.5 mg, 0.025 mmol) in a mixture of water and acetone with 2:1 molar ratio at 60 ℃ for 7 h to give 3·NO3·H2O. 1H NMR of 3·NO3·H2O: (400 MHz, DMSO-d6, 298 K, TMS): δ 8.93 (d, J=8.2 Hz, 2H), 8.87 (d, J=5.2 Hz, 2H), 8.23 (s, 2H), 8.15 (m, J=4.7 Hz, 6H), 7.69 (t, J=7.3 Hz, 2H), 7.55 (t, J=7.6 Hz, 4H), 7.02 (s, 4H). 13C NMR (400 MHz, DMSO-d6, 298 K, TMS): δ 180.27, 147.95, 145.69, 140.94, 134.31, 133.23, 130.46, 129.09, 128.33, 126.65, 49.07. ESI-MS (CH3CN, m/z): Calcd. for [(phen)Pd(L)]+ 509.05, Found 509.01. Elemental analysis calculated for 3·NO3·H2O (C27H21N3O6Pt, %): C: 54.97, H: 3.59, N: 7.12. Found(%): C: 55.00, H: 3.56, N: 7.11. A ten-fold excess of KPF6 was added to the solution, the yellow precipitation were collected by centrifugation, washed with minimum amount of water and dried in vacuum to give pale yellow solid of 3·PF6·H2O. (33.2 mg, 0.049 mmol, 97% yield). 1H NMR confirmed the quantitative formation of 3·PF6·H2O. 1H NMR of 3·PF6·H2O: (400 MHz, DMSO-d6, 298 K, TMS): δ 8.56 (d, J=8.1 Hz, 2H), 8.48 (d, J=4.4 Hz, 2H), 7.98 (s, 2H), 7.86 (m, 2H), 7.80 (d, J=7.4 Hz, 4H), 7.59 (t, J=7.3 Hz, 2H), 7.37 (t, J=7.8 Hz, 4H), 6.61 (s, 1H). 13C NMR (400 MHz, DMSO-d6, 298 K, TMS): δ 180.46, 148.01, 145.86, 141.00, 134.46, 133.26, 130.52, 129.13, 128.36, 126.66, 96.02. ESI-MS (CH3CN, m/z): Calcd. for [(phen)Pd(L)]+ 509.05, Found 509.04. Elemental anal-ysis calculated for 3·PF6·H2O (C27H21F6N2O3PPd, %): C: 48.20, H: 3.15, N: 4.16. Found: C: 48.18, H: 3.15, N: 4.17.
1.3 X-ray crystallography of complex 1·PF6· CH3CN
X-ray diffraction data of the crystals of complex 1·PF6·CH3CN was collected at 150(2) K by using Bruker Smart Apex CCD area detector equipped with a graphite monochromated Mo Kα radiation (λ=0.071 073 nm). The structure of 1·PF6·CH3CN was solved by direct method and refined by employing full matrix least-square on F2 by using SHELXTL (Bruker, 2000) program and expanded using Fourier techniques[24-25]. All non-H atoms of the complex 1·PF6·CH3CN were refined with anisotropic thermal parameters. The hydrogen atoms were included in idealized positions with isotropic displacement parameters constrained to 1.5 times the Uequiv of their attached carbon atoms for methylene hydrogens, and 1.2 times the Uequiv of their attached carbon atoms for all others. SQUEEZE option was employed to treat the disordered counter anions. The crystallographic data of complex 1·PF6·CH3CN were listed in Table 1 and the selected hydrogen bond lengths and bond angles of complex 1·PF6·CH3CN were listed in Table S1 and S2.
Formula C27H22F6N3O2PPd F(000) 1 344 Formula weight 671.84 Index ranges -8 ≤ h ≤ 8, -21 ≤ k ≤ 21≤k, -23 ≤l ≤ 23 Crystal system triclinic Monoclinic θ range / (°) 2.360~25.246 Space group P21/n Reflection collected 12 779 a / nm 0.711 28(6) Observed reflection with [I > 2σ(I)] 3 521 b/ nm 1.807 66(19) Number of parameter 356 c / nm 1.970 19(15) Independent reflection 4 436 (Rint=0.060 1) β/(°) 94.296(5) Goodness-of-fit on F2 1.096 Volume / nm3 2.526 1(4) Final R indices [I > 2σ(I)] R1=0.041 1, wR2=0.100 6 Z 4 R indices (all data) R1=0.052, wR2=0.104 3 Dc / (g·cm-3) 1.767 Largest diff. peak and hole / (e·nm-3) 1 415 and -1 274 μ/ mm-1 0.875 CCDC: 1566337, 1·PF6·CH3CN.
2 Results and discussion
2.1 Characterization of 1·PF6·H2O, 2·PF6·H·H2O and 3·PF6·H2O
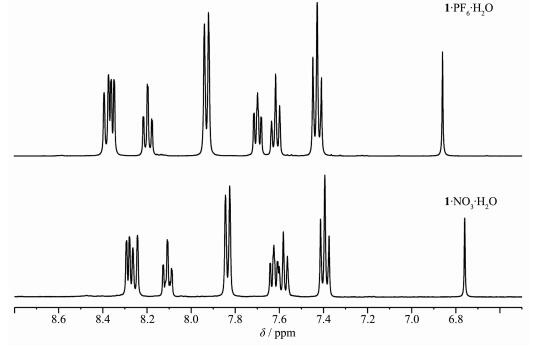
NMR were fully carried out to characterize the complexes of 1·PF6·H2O, 2·PF6·H·H2O and 3·PF6·H2O. Analysis by 1H NMR spectroscopy of 1·NO3·H2O in DMSO-d6 solutions clearly showed an array of well-defined resonance and suggested the self-assembly of [(bpy)2Pd2(NO3)2](NO3)2 and L to form a single product (Fig.S1~S2). Upon replaced by PF6-, a series of peaks shifted downfield as shown in Fig. 1. The results of 1H NMR spectroscopy indicated that the formation of a 1:1 complex of 1·PF6·H2O. Detailed analysis of 1H NMR spectra belonged to the complex 1·PF6·H2O was disc-ussed as below: for complex 1·PF6·H2O, the featured single peak at 6.86 corresponded to methylene-H, the triplet at 7.43 were assigned to aromatic-H2, aromatic-H2′, aromatic-H4 and aromatic-H4′, the triplet at 7.62 with integral of 2 H assigned to pyridine-H7 and pyridine-H7′, and the triplet at 7.70 with integral of 2 H assigned to pyridine-H8 and pyridine-H8′, the doublet at 7.93 with integral of 4 H assigned to aromatic -H1, aromatic-H1′, aromatic-H5 and aromatic-H5′, the triplet at 8.19 with integral of 2 H assigned to aromatic-H3 and aromatic-H3′, the downfield 4 H assigned to pyridine-H6, pyridine-H6′, pyridine-H9 and pyridine-H9′, respectively. And the results of 13C NMR spectroscopy as shown in Fig.S8 agreed well with the analysis results of 1H NMR spectroscopy. These results were consistent with those of the complexes 2·NO3·H2O, 2·PF6·H·H2O, 3·NO3·H2O and 3·PF6·H2O (Fig.S3~S6 and Fig.S9~S12).
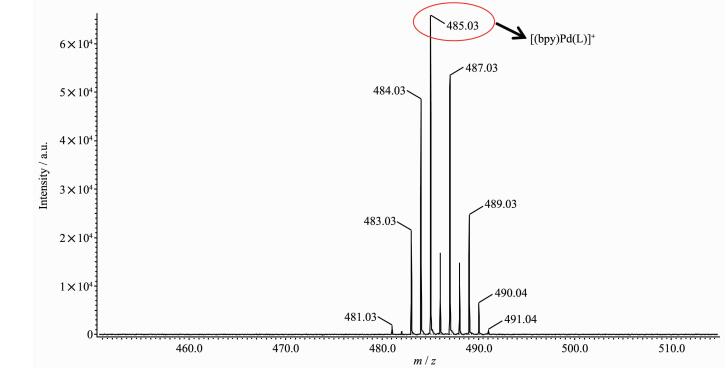
ESI-MS studies also confirmed the structure of 1·NO3·H2O, 1·PF6·H2O, 2·NO3·H2O, 2·PF6·H·H2O, 3·NO3·H2O and 3·PF6·H2O in solution (Fig. 2, S13~S17). Isotope patterns matched those simulated and peak separations consistent with the charges. When an acetonitrile solution of 1·PF6·H2O was subjected to the ESI-MS, prominent peaks for [(bpy)Pd(L)]+ were clearly observed at 485.03, indicating the complete formation of metal-organic complexes. Additionally, the striking peak at 485.03 also confirmed the spontaneous deprotonation of 1, 3-diphenylpropane-1, 3-dione (L) in solution driven by coordination effect. Similarly, the ESI-MS study of 2·PF6·H·H2O and 3·PF6·H2O afforded a series of peaks at m/z 574.09 and 509.04 were similar to that of complex 1·PF6·H2O.
2.2 Crystal structure of 1·PF6·CH3CN
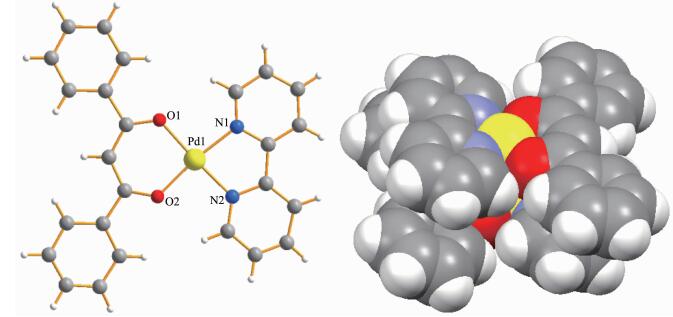
The molecular structure of complex 1·PF6·CH3CN was unambiguously determined by reliable methods of X-ray diffraction analysis. As shown in Fig. 3, complex 1·PF6·CH3CN crystallizes in monoclinic space group P21/n. The crystal structure of 1·PF6·CH3CN displays a mononuclear palladium(Ⅱ) complex with planar conformation, and a parallel pattern is formed between planes. A dimeric crystal structure is formed by the efficient π-π stacking interactions and the metal-metal interactions, which make the complex of 1·PF6·CH3CN be an efficient catalyst. The central palladium(Ⅱ) is coordinated by two N atoms (the two N atoms of bpy) and two O atoms (the two O atoms of L) in a square coordination mode. The distances of Pd(1)-O(1) and Pd(1)-O(2) are 0.200 7 and 0.197 8 nm, respectively. And the distances between Pd(1) and the two N atoms are 0.201 6 and 0.198 5 nm, respectively. While the intermolecular Pd(Ⅱ)…Pd(Ⅱ) distance is about 0.322 4 nm, which indicates that the interactions exist between them, and the interaction may be suitable for the catalysis applications of the complex 1·PF6·CH3CN. The angles of O(1)-Pd(1)-O(2) and N(1)-Pd(1)-N(2) were 93.06° and 82.10°, respectively. The dihedral angle, defined by planes O(1)-Pd(1)-O(2) and N(1)-Pd(1)-N(2), is 5.48°. Extending a, b and c axes with PF6- anions and acetonitrile molecules frozen inside as shown in Fig. 3 and S18. The structure determined by X-ray crystallographic analysis agreed well with the NMR and ESI-MS analysis. We had tried many times to obtain the crystals of 2·PF6·CH3CN and 3·PF6·CH3CN, but failed.
2.3 Catalytic activity
For the importance of the Suzuki cross-coupling reaction and the structural characteristics of palladium and platinum (Ⅱ, Ⅱ) complexes, 1·PF6·H2O, 2·PF6·H·H2O and 3·PF6·H2O were devoted to explore the catalyst activity. In our previous work, we have discussed the catalyst activity of pyrazolate-based dipalladium(Ⅱ) complexes[26]. In this work, different solvents, temperature, reaction time and reagents were examined to optimize the process conditions.
Firstly, effects of different solvents were investigated, and the optimum conditions were shown in Table 2. According to previous experiments records[27], 1, 4-dioxane and ethanol were prepared for the catalyst activity, and it was observed that different solvents are suited for different reactions. Meanwhile, the temperature and the reaction time were adjusted to achieve the optimal strategy.
Entry Catalyst Co-catalyst Solvent T / ℃ Time / h Yield / % 1 1·PFc·HeO K3PO4 1, 4-dioxane 100 12 94 2 2·PF6·H2O K3PO4 1, 4-dioxane 100 36 87 3 3·PF6·H2O K3PO4 1, 4-dioxane 100 24 89 4 1·PB6·H2O K3PO4 ethanol 80 12 91 5 2·PF6·H2O K3PO4 ethanol 80 24 88 6 3·PF6·H2O K3PO4 ethanol 80 12 92 Next, the influence of reagents was explored in the controlled experiments. Different yields but satisfactory results were obtained for the aryl-aryl reactions (Table 3). But for the heterocyclic-based reactions, since the previous set of experiments have consistently use unprotected starting pyrazol-based, the palladium or platinum ions in 1·PF6·H2O, 2·PF6·H·H2O and 3·PF6·H2O complexes could coordinate with the unprotected starting pyrazole, leading to side-products and yield decreasing. Then a series of protected starting pyrazol-based were employed for the cross-coupling reactions, as expected, the yields of adducts were higher. With the optimized reaction conditions in hand, a broad substrate listed in Table 3 is observed. The results of control experiment and blank experiment show that good catalytic effect would be found only when main catalysts and sub-catalysts coexist simultaneously. It seems that the electronic effect of metal-metal interaction and the steric effect of the catalysts make the reaction efficiency.
Entry Catalyst Co-catalyst Substrate A Substrate B Solvent T/℃ Yield/% 1 1·PF6·H2O None 

ethanol 100 11.4 2 None K3PO4 

ethanol 100 7.3 3 None None 

ethanol 100 7.8 4 1·PF6·H2O K3PO4 

1, 4-dioxane 100 92 5 2·PF6·H2O K3PO4 

ethanol 80 88 6 3·PF6·H2O K3PO4 

ethanol 80 89 7 1·PF6·H2O K3PO4 

1, 4-dioxane 100 86 8 2·PF6·H2O K3PO4 

ethanol 80 79 9 3·PF6·H2O K3PO4 

1, 4-dioxane 100 81 10 1·PF6·H2O K3PO4 

1, 4-dioxane 100 83 11 2·PF6·H2O K3PO4 

ethanol 80 73 12 3·PF6·H2O K3PO4 

ethanol 80 75 13 1·PF6·H2O K3PO4 

1, 4-dioxane 100 88 14 2·PF6·H2O K3PO4 

ethanol 80 75 15 3·PF6·H2O K3PO4 

ethanol 80 82 16 1·PF6·H2O K3PO4 

ethanol 80 35 17 2·PF6·H2O K3PO4 

ethanol 80 26 18 3·PF6·H2O K3PO4 

ethanol 80 29 19 1·PF6·H2O K3PO4 

ethanol 80 25 20 2·PF6·H2O K3PO4 

ethanol 80 27 21 3·PF6·H2O K3PO4 

ethanol 80 22 22 1·PF6·H2O K3PO4 

ethanol 80 72 23 2·PF6·H2O K3PO4 

ethanol 80 68 24 3·PF6·H2O K3PO4 

ethanol 80 69 25 1·PF6·H2O K3PO4 

ethanol 80 67 26 2·PF6·H2O K3PO4 

ethanol 80 64 27 3·PF6·H2O K3PO4 

ethanol 80 73 3 Conclusions
In summary, we have synthesized three mono-metallic complexes in quantitative yields by a directed self-assembly of diketone-based ligands with [(bpy)Pd(NO3)]NO3, [(bpy)Pt(NO3)]NO3 and [(phen)Pd(NO3)]NO3 in a 2:1 molar ratio. The assemblies have been chara-cterized by NMR and ESI-MS, and the complex of 1·PF6·CH3CN was fully defined by single-crystal X-ray diffraction method. These characterizations show the structural similarity of these assemblies. The single-crystal structures show that weak intramolecular Pd…Pd interactions exist in 1·PF6·CH3CN. More signifi-cantly, these metal-organic species with metal-metal interaction have potential application in the field of Suzuki cross-coupling reaction.
Supporting information is available at http://www.wjhxxb.cn
-
-
[1]
Frank M, Ahrens J, Bejenke I, et al. J. Am. Chem. Soc., 2016, 138:8279-8287 doi: 10.1021/jacs.6b04609
-
[2]
Klein C, Gütz C, Bogner M, et al. Angew. Chem., Int. Ed., 2014, 53:3739-3742 doi: 10.1002/anie.v53.14
-
[3]
Luo D, Zhou X P, Li D. Angew. Chem., Int. Ed., 2015, 54:6190-6195 doi: 10.1002/anie.201501081
-
[4]
Yoshizawa M, Yoshizawa Y, Kusukawa T, et al. Angew. Chem. Int. Ed., 2002, 41:1347-1349 doi: 10.1002/1521-3773(20020415)41:8<>1.0.CO;2-M
-
[5]
Cui Y, Chen Z M, Jiang X F, et al. Dalton Trans., 2017, 46:5801-5805 doi: 10.1039/C7DT00179G
-
[6]
Jiang X F, Huang H, Chai Y F, et al. Nat. Chem., 2017, 9:188-193 doi: 10.1038/nchem.2637
-
[7]
Ronson T K, Pilgrim B S, Nitschke J R. J. Am. Chem. Soc., 2016, 138:10417-10420 doi: 10.1021/jacs.6b06710
-
[8]
Liu Y, Kravtsov V, Walsh R D, et al. Chem. Commun., 2004:2806-2807
-
[9]
Sun Q F, Liu L X, Huang H P, et al. Inorg. Chem., 2008, 47:2142-2154 doi: 10.1021/ic701344p
-
[10]
Ward M D, Raithby P R. Chem. Soc. Rev., 2013, 42:1619-1636 doi: 10.1039/C2CS35123D
-
[11]
Fleming J S, Mann K L V, Carraz C A, et al. Angew. Chem. Int. Ed., 1998, 37:1279-1281 doi: 10.1002/(ISSN)1521-3773
-
[12]
Fiedler D, Leung D H, Bergman R G, et al. Acc. Chem. Res., 2005, 38:349-358 doi: 10.1021/ar040152p
-
[13]
Fujita M, Tominaga M, Hori A, et al. Acc. Chem. Res., 2005, 38:369-378 doi: 10.1021/ar040153h
-
[14]
Seidel S R, Stang P J. Acc. Chem. Res., 2002, 35:972-983 doi: 10.1021/ar010142d
-
[15]
Tominaga M, Suzuki K, Kawano M, et al. Angew. Chem. Int. Ed., 2004, 43:5621-5625 doi: 10.1002/(ISSN)1521-3773
-
[16]
Suzuki K, Kawano M, Fujita M. Angew. Chem. Int. Ed., 2007, 46:2819-2822 doi: 10.1002/(ISSN)1521-3773
-
[17]
Wang Y Q, Jiang X F, Li H, et al. Chem. Asian J., 2015, 10:1146-1149 doi: 10.1002/asia.v10.5
-
[18]
Xie Z T, Guo C, Yu S Y, et al. Angew. Chem. Int. Ed., 2012, 51:1177-1181 doi: 10.1002/anie.201106504
-
[19]
Baxter P N W. Comprehensive Supramolecular Chemistry: Vol. 9. Sauvage J P, Hosseini M W. Ed. New York: Pergamon, 1996.
-
[20]
Basu B, Biswas K, Kundu S, et al. Green Chem., 2010, 12:1734-1738 doi: 10.1039/c0gc00122h
-
[21]
Wang F, Li C, Chen H, et al. J. Am. Chem. Soc., 2013, 135:5588-5601 doi: 10.1021/ja310501y
-
[22]
Muratsugu S, Maity N, Baba H, et al. Dalton Trans., 2017, 46:3125-3134 doi: 10.1039/C7DT00124J
-
[23]
Yu S Y, Fujita M, Yamaguchi K. J. Chem. Soc. Dalton Trans., 2001:3145-3146
-
[24]
Sheldrick G M. SHELXS-97, Program for the Solution of Crystal Structures, University of Göttingen, Germany, 1997.
-
[25]
Sheldrick G M. SHELXL-97, Program for the Refinement of Crystal Structures, University of Göttingen, Germany, 1997.
-
[26]
陈涵, 于智淳, 邓威, 等.无机化学学报, 2017, 33:939-946 doi: 10.11862/CJIC.2017.090CHEN Han, YU Zhi-Chun, DENG Wei, et al. Chinese J. Inorg. Chem., 2017, 33:939-946 doi: 10.11862/CJIC.2017.090
-
[1]
-
Table 1. Crystallographic data for complex 1·PF6·CH3CN
Formula C27H22F6N3O2PPd F(000) 1 344 Formula weight 671.84 Index ranges -8 ≤ h ≤ 8, -21 ≤ k ≤ 21≤k, -23 ≤l ≤ 23 Crystal system triclinic Monoclinic θ range / (°) 2.360~25.246 Space group P21/n Reflection collected 12 779 a / nm 0.711 28(6) Observed reflection with [I > 2σ(I)] 3 521 b/ nm 1.807 66(19) Number of parameter 356 c / nm 1.970 19(15) Independent reflection 4 436 (Rint=0.060 1) β/(°) 94.296(5) Goodness-of-fit on F2 1.096 Volume / nm3 2.526 1(4) Final R indices [I > 2σ(I)] R1=0.041 1, wR2=0.100 6 Z 4 R indices (all data) R1=0.052, wR2=0.104 3 Dc / (g·cm-3) 1.767 Largest diff. peak and hole / (e·nm-3) 1 415 and -1 274 μ/ mm-1 0.875 Table 2. Catalytic activity of complexes 1·PF6·H2O, 2·PF6·H·H2O and 3·PF6·H2O
Entry Catalyst Co-catalyst Solvent T / ℃ Time / h Yield / % 1 1·PFc·HeO K3PO4 1, 4-dioxane 100 12 94 2 2·PF6·H2O K3PO4 1, 4-dioxane 100 36 87 3 3·PF6·H2O K3PO4 1, 4-dioxane 100 24 89 4 1·PB6·H2O K3PO4 ethanol 80 12 91 5 2·PF6·H2O K3PO4 ethanol 80 24 88 6 3·PF6·H2O K3PO4 ethanol 80 12 92 Table 3. Catalytic activity of complexes 1·PF6·H2O, 2·PF6·H·H2O and 3·PF6·H2O
Entry Catalyst Co-catalyst Substrate A Substrate B Solvent T/℃ Yield/% 1 1·PF6·H2O None 

ethanol 100 11.4 2 None K3PO4 

ethanol 100 7.3 3 None None 

ethanol 100 7.8 4 1·PF6·H2O K3PO4 

1, 4-dioxane 100 92 5 2·PF6·H2O K3PO4 

ethanol 80 88 6 3·PF6·H2O K3PO4 

ethanol 80 89 7 1·PF6·H2O K3PO4 

1, 4-dioxane 100 86 8 2·PF6·H2O K3PO4 

ethanol 80 79 9 3·PF6·H2O K3PO4 

1, 4-dioxane 100 81 10 1·PF6·H2O K3PO4 

1, 4-dioxane 100 83 11 2·PF6·H2O K3PO4 

ethanol 80 73 12 3·PF6·H2O K3PO4 

ethanol 80 75 13 1·PF6·H2O K3PO4 

1, 4-dioxane 100 88 14 2·PF6·H2O K3PO4 

ethanol 80 75 15 3·PF6·H2O K3PO4 

ethanol 80 82 16 1·PF6·H2O K3PO4 

ethanol 80 35 17 2·PF6·H2O K3PO4 

ethanol 80 26 18 3·PF6·H2O K3PO4 

ethanol 80 29 19 1·PF6·H2O K3PO4 

ethanol 80 25 20 2·PF6·H2O K3PO4 

ethanol 80 27 21 3·PF6·H2O K3PO4 

ethanol 80 22 22 1·PF6·H2O K3PO4 

ethanol 80 72 23 2·PF6·H2O K3PO4 

ethanol 80 68 24 3·PF6·H2O K3PO4 

ethanol 80 69 25 1·PF6·H2O K3PO4 

ethanol 80 67 26 2·PF6·H2O K3PO4 

ethanol 80 64 27 3·PF6·H2O K3PO4 

ethanol 80 73 -

 扫一扫看文章
扫一扫看文章
计量
- PDF下载量: 3
- 文章访问数: 1560
- HTML全文浏览量: 94


 下载:
下载:




 下载:
下载:

