Citation:
Liu Gang, Wang Tie. Research Progress in Thermoelectric Materials for Sensor Application[J]. Acta Chimica Sinica,
;2017, 75(11): 1029-1035.
doi:
10.6023/A17060259

-
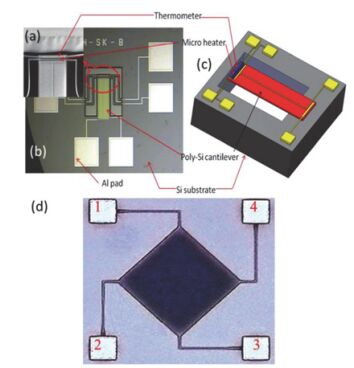
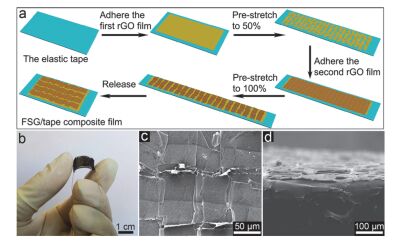
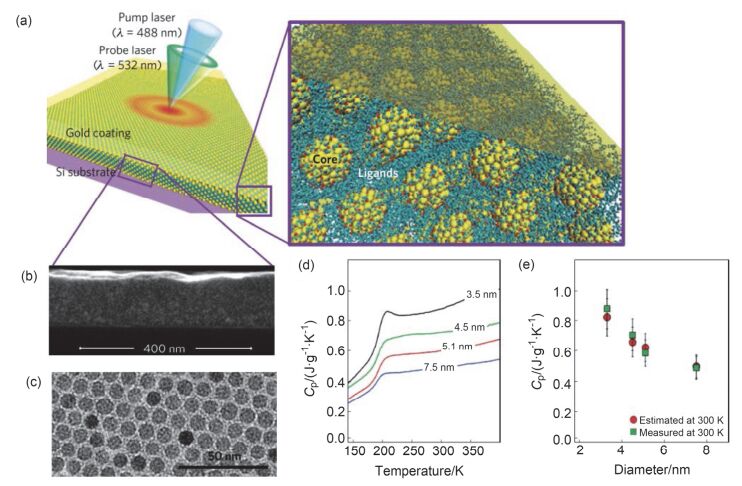
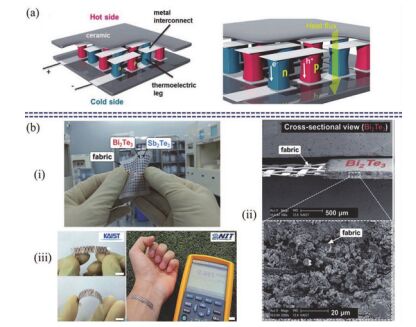
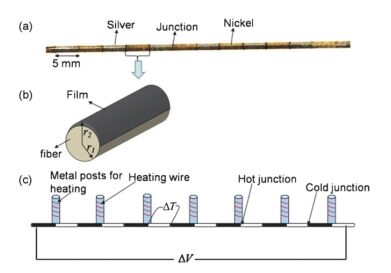
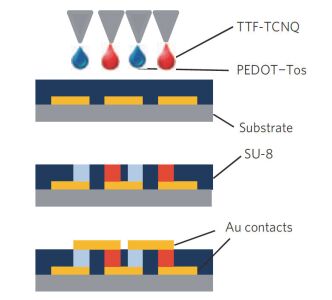
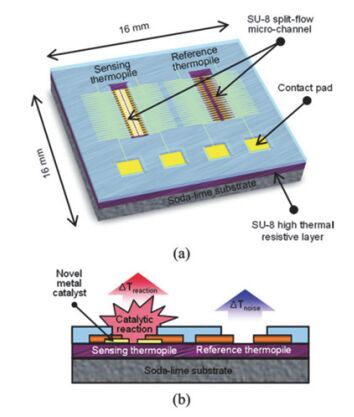
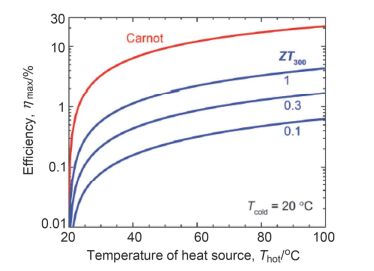
Sensors are core components for modern intelligent industry. Thermoelectric materials, which have significant influence on the design and functions for a variety types of sensors, attracted more and more attentions recently. In this paper, different categories of thermoelectric materials, such as silicon, carbon, lead, tellurium, precious metal, organic and catalysis based thermoelectric materials, are discussed in detail on their high sensitivity, fast response, and stability as potential candidates for specific sensors. The silicon-based thermoelectric materials are of particular efficiency in sensor data process and transmission due to their high purity. Carbon-based thermoelectric materials, including graphene and carbon nanotubes, advantage in their excellent conductivity, flexible structure, and manufactural controllability. Lead-based thermoelectric materials are mainly used as infrared sensors because of their natural sensitivity to infrared specially. Telluride-based thermoelectric materials, especially Bismuth Telluride and Antimony Telluride, can form PN junction and be applied as soft sensors. Products based on these materials have already been developed for detecting pulses. The precious metals-based thermoelectric materials, e.g. gold or silver, are commonly used as dopant in the organic thermoelectric materials to adjust their sensitivity. Organic thermoelectric materials benefit from their good stability and variability, while copper-bismuth alloy based thermoelectric materials are widely investigated to make gas sensors. In general, the inorganic thermoelectric materials normally feature high electrical conductivity, which enhances the sensitivity of sensors, whereas the organic thermoelectric materials have high stability to maintain the stability of sensors. At present, the miniaturization of sensors is the mainstream for both material study and device fabrication. Low dimensional thermoelectric materials, especially nano-scaled materials such as quantum dots, nanowires, etc., will for sure promote the progressing of sensor development. For example, carbon nanotube can be knit into specific sheets as we designed with tunable conductivity, which makes them of remarkable industrial potentials as soft sensors. Designing and fabricating multi-functional and space-saving thermoelectric materials with well aligned and effectively assembled nanomaterials would be a feasible and practicable approach for future sensors.
-
Keywords:
- sensor,
- thermoelectric materials,
- silicon nanowire,
- graphene
-

-
-
[1]
Chowdhury, I.; Prasher, R.; Lofgreen, K.; Chrysler, G.; Nara-simhan, S.; Mahajan, R.; Koester, D.; Alley, R.; Venkatasubramanian, R. Nature Nanotech. 2009, 4, 235. doi: 10.1038/nnano.2008.417
-
[2]
Li, J. F.; Liu, W.; Zhao, L. D.; Zhou, M. NPG Asia Mater. 2010, 2, 152. doi: 10.1038/asiamat.2010.138
-
[3]
Rama, V.; Siivola, E.; Thomas, C.; O'Quinn, B. Nature 2001, 413, 597. doi: 10.1038/35098012
-
[4]
Delaire, O.; Ma, J.; Marty, K.; May, A. F.; McGuire, M. A.; Du, M. H.; Singh, D. J.; Podlesnyak, A.; Ehlers, G.; Lumsden, M. D.; Sales, B. C. Nature Mater. 2011, 10, 614. doi: 10.1038/nmat3035
-
[5]
Coucheron, D. A.; Fokine, M.; Patil, N.; Breiby, D. W.; Buset, O. T.; Healy, N.; Peacock, A. C.; Hawkins, T.; Jones, M.; Ballato, J.; Gibson, U. J. Nat. Commun. 2016, 7, 13265. doi: 10.1038/ncomms13265
-
[6]
(a) Xie, P.; Xiong, Q.; Fang, Y.; Qing, Q.; Lieber, C. M. Nature Nanotech. 2011, 7, 119; (b) Boukai, A. I.; Bunimovich, Y.; Tahir-Kheli, J.; Yu, J. K.; Goddard, W. A., 3rd; Heath, J. R. Nature 2008, 451, 168; (c) Hochbaum, A. I.; Chen, R.; Delgado, R. D.; Liang, W.; Garnett, E. C.; Najarian, M.; Majumdar, A.; Yang, P. Nature 2008, 451, 163.
-
[7]
Mao, J.; Liu, Z.; Ren, Z. npj Quantum Materials 2016, 1, 16028. doi: 10.1038/npjquantmats.2016.28
-
[8]
McGrail, B. T.; Sehirlioglu, A.; Pentzer, E. Angew. Chem. 2015, 54, 1710. doi: 10.1002/anie.201408431
-
[9]
Kroon, R.; Mengistie, D. A.; Kiefer, D.; Hynynen, J.; Ryan, J. D.; Yu, L.; Muller, C. Chem. Soc. Rev. 2016, 45, 6147. doi: 10.1039/C6CS00149A
-
[10]
Wang, Z.; Leonov, V.; Fiorini, P.; Van Hoof, C. Sens. Actuators, A:Physical 2009, 156, 95. doi: 10.1016/j.sna.2009.02.028
-
[11]
Liu, X.; Wang, Y.; Huang, Y.; Feng, X.; Fan, Q.; Huang, W. Acta Chim. Sinica 2016, 74, 664.
-
[12]
He, W.; Zhang, G.; Zhang, X.; Ji, J.; Li, G.; Zhao, X. Appl. Energy 2015, 143, 1. doi: 10.1016/j.apenergy.2014.12.075
-
[13]
Marichy, C.; Bechelany, M.; Pinna, N. Adv. Mater. 2012, 24, 1017. doi: 10.1002/adma.201104129
-
[14]
Pu, X.; Liu, M.; Chen, X.; Sun, J.; Du, C.; Zhang, Y.; Zhai, J.; Hu, W.; Wang, Z. L. Science Advances 2017, 3, e1700015. doi: 10.1126/sciadv.1700015
-
[15]
Zhang, C.; Meng, Y.; Kuang, J.; Xu, L. Acta Chim. Sinica 2015, 73, 409.
-
[16]
Qian, X.; Su, M.; Li, F.; Song, Y. Acta Chim. Sinica 2016, 74, 565. doi: 10.3866/PKU.WHXB201511301
-
[17]
Zhu, W.; Deng, Y.; Cao, L. Nano Energy 2017, 34, 463. doi: 10.1016/j.nanoen.2017.03.020
-
[18]
Zhang, F.; Zang, Y.; Huang, D.; Di, C. A.; Zhu, D. Nat. Commun. 2015, 6, 8356. doi: 10.1038/ncomms9356
-
[19]
Wang, H.; He, Y. Sensors 2017, 17, 268. doi: 10.3390/s17020268
-
[20]
Rao, S.; Pangallo, G.; Della Corte, F. G. Sensors 2016, 16, 67. doi: 10.3390/s16010067
-
[21]
Li, W.; Feng, Z.; Dai, E.; Xu, J.; Bai, G. Sensors 2016, 16, 1880.
-
[22]
Zhan, B.; Li, C.; Yang, J.; Jenkins, G.; Huang, W.; Dong, X. Small 2014, 10, 4042.
-
[23]
Singh, S.; Lee, S.; Kang, H.; Lee, J.; Baik, S. Energy Storage Materials 2016, 3, 55. doi: 10.1016/j.ensm.2016.01.004
-
[24]
Quan, Z.; Luo, Z.; Wang, Y.; Xu, H.; Wang, C.; Wang, Z.; Fang, J. Nano Lett. 2013, 13, 3729. doi: 10.1021/nl4016705
-
[25]
Hong, M.; Chen, Z. G.; Yang, L.; Zou, J. Nanoscale 2016, 8, 8681. doi: 10.1039/C6NR00719H
-
[26]
Snyder, G. J.; Lim, J. R.; Huang, C. K.; Fleurial, J. P. Nature Mater. 2003, 2, 528. doi: 10.1038/nmat943
-
[27]
Galli, G.; Donadio, D. Nature Nanotech. 2010, 5, 701. doi: 10.1038/nnano.2010.199
-
[28]
Zhou, H.; Kropelnicki, P.; Lee, C. Nanoscale 2015, 7, 532. doi: 10.1039/C4NR04184D
-
[29]
Jung, S. W.; Shin, J. Y.; Pi, K.; Goo, Y. S.; Cho, D. D. Sensors 2016, 16, 2035. doi: 10.3390/s16122035
-
[30]
Weiss, N. O.; Zhou, H.; Liao, L.; Liu, Y.; Jiang, S.; Huang, Y.; Duan, X. Adv. Mater. 2012, 24, 5782. doi: 10.1002/adma.201201482
-
[31]
Liu, Q.; Chen, J.; Li, Y.; Shi, G. ACS Nano 2016, 10, 7901. doi: 10.1021/acsnano.6b03813
-
[32]
Wu, G.; Zhang, Z. G.; Li, Y.; Gao, C.; Wang, X.; Chen, G. ACS Nano 2017, 11, 5746. doi: 10.1021/acsnano.7b01279
-
[33]
Chen, J.; Wang, L.; Gui, X.; Lin, Z.; Ke, X.; Hao, F.; Li, Y.; Jiang, Y.; Wu, Y.; Shi, X.; Chen, L. Carbon 2017, 114, 1. doi: 10.1016/j.carbon.2016.11.074
-
[34]
Ong, W.-L.; Rupich, S. M.; Talapin, D. V.; McGaughey, A. J. H.; Malen, J. A. Nature Mater. 2013, 12, 410. doi: 10.1038/nmat3596
-
[35]
Lu, Z.; Zhang, H.; Mao, C.; Li, C. M. Appl. Energy 2016, 164, 57. doi: 10.1016/j.apenergy.2015.11.038
-
[36]
Wu, H.; Huang, Y.; Xu, F.; Duan, Y.; Yin, Z. Adv. Mater. 2016, 28, 9881. doi: 10.1002/adma.201602251
-
[37]
Cao, Z.; Koukharenko, E.; Tudor, M. J.; Torah, R. N.; Beeby, S. P. Sens. Actuators A:Physical 2016, 238, 196. doi: 10.1016/j.sna.2015.12.016
-
[38]
Yadav, A.; Pipe, K. P.; Shtein, M. J. Power Sources 2008, 175, 909. doi: 10.1016/j.jpowsour.2007.09.096
-
[39]
Russ, B.; Glaudell, A.; Urban, J. J.; Chabinyc, M. L.; Segalman, R. A. Nature Rev. Mater. 2016, 1, 16050. doi: 10.1038/natrevmats.2016.50
-
[40]
Bubnova, O.; Khan, Z. U.; Malti, A.; Braun, S.; Fahlman, M.; Berggren, M.; Crispin, X. Nature Mater. 2011, 10, 429. doi: 10.1038/nmat3012
-
[41]
Zhang, Q.; Sun, Y.; Xu, W.; Zhu, D. Adv. Mater. 2014, 26, 6829. doi: 10.1002/adma.v26.40
-
[42]
Ju, H.; Kim, J. Chem. Eng. J. 2016, 297, 66. doi: 10.1016/j.cej.2016.03.137
-
[43]
Song, H.; Cai, K. Energy 2017, 125, 519. doi: 10.1016/j.energy.2017.01.037
-
[44]
Kim, G. H.; Shao, L.; Zhang, K.; Pipe, K. P. Nature Mater. 2013, 12, 719-23. doi: 10.1038/nmat3635
-
[45]
Park, S. C.; Yoon, S. I.; Lee, C. I.; Kim, Y. J.; Song, S. Analyst 2009, 134, 236. doi: 10.1039/B807882C
-
[1]
-

-
-
[1]
Chaolin Mi , Yuying Qin , Xinli Huang , Yijie Luo , Zhiwei Zhang , Chengxiang Wang , Yuanchang Shi , Longwei Yin , Rutao Wang . Galvanic Replacement Synthesis of Graphene Coupled Amorphous Antimony Nanoparticles for High-Performance Sodium-Ion Capacitor. Acta Physico-Chimica Sinica, 2024, 40(5): 2306011-0. doi: 10.3866/PKU.WHXB202306011
-
[2]
Pengcheng Yan , Peng Wang , Jing Huang , Zhao Mo , Li Xu , Yun Chen , Yu Zhang , Zhichong Qi , Hui Xu , Henan Li . Engineering Multiple Optimization Strategy on Bismuth Oxyhalide Photoactive Materials for Efficient Photoelectrochemical Applications. Acta Physico-Chimica Sinica, 2025, 41(2): 100014-0. doi: 10.3866/PKU.WHXB202309047
-
[3]
Songmei Ma , Ying Zhang , Gang Liu , Wenlong Xu . Comprehensive Experiment Teaching Exploration and Practice in Polymeric Materials Integrating Research-Driven Learning, Creativity-Enhanced Competency, and Science-Education Synergy: A Case Study of Machine Learning-Assisted Intelligent Handwriting Recognition System. University Chemistry, 2026, 41(1): 289-297. doi: 10.12461/PKU.DXHX202509083
-
[4]
Anbang Du , Yuanfan Wang , Zhihong Wei , Dongxu Zhang , Li Li , Weiqing Yang , Qianlu Sun , Lili Zhao , Weigao Xu , Yuxi Tian . Photothermal Microscopy of Graphene Flakes with Different Thicknesses. Acta Physico-Chimica Sinica, 2024, 40(5): 2304027-0. doi: 10.3866/PKU.WHXB202304027
-
[5]
Zhihuan XU , Qing KANG , Yuzhen LONG , Qian YUAN , Cidong LIU , Xin LI , Genghuai TANG , Yuqing LIAO . Effect of graphene oxide concentration on the electrochemical properties of reduced graphene oxide/ZnS. Chinese Journal of Inorganic Chemistry, 2024, 40(7): 1329-1336. doi: 10.11862/CJIC.20230447
-
[6]
Hailang JIA , Yujie LU , Pengcheng JI . Preparation and properties of nitrogen and phosphorus co-doped graphene carbon aerogel supported ruthenium electrocatalyst for hydrogen evolution reaction. Chinese Journal of Inorganic Chemistry, 2025, 41(11): 2327-2336. doi: 10.11862/CJIC.20250021
-
[7]
Yuanchun Pan , Xinyun Lin , Leyi Yang , Wenya Hu , Dekui Song , Nan Liu . Artificial Intelligence Science Practice: Preparation of Electronic Skin by Chemical Vapor Deposition of Graphene. University Chemistry, 2025, 40(11): 272-280. doi: 10.12461/PKU.DXHX202412052
-
[8]
Cun WANG , Shaohan XU , Yuqian ZHANG , Yaoyao ZHANG , Tao GONG , Rong WEN , Yuhang LIAO , Yanrong REN . Terbium complex electrochemiluminescent emitters: Synthesis and application in the detection of epinephrine. Chinese Journal of Inorganic Chemistry, 2025, 41(7): 1351-1360. doi: 10.11862/CJIC.20240427
-
[9]
Tao Xu , Wei Sun , Tianci Kong , Jie Zhou , Yitai Qian . Stable Graphite Interface for Potassium Ion Battery Achieving Ultralong Cycling Performance. Acta Physico-Chimica Sinica, 2024, 40(2): 2303021-0. doi: 10.3866/PKU.WHXB202303021
-
[10]
Yang Meiqing , Lu Wang , Haozi Lu , Yaocheng Yang , Song Liu . Recent Advances of Functional Nanomaterials for Screen-Printed Photoelectrochemical Biosensors. Acta Physico-Chimica Sinica, 2025, 41(2): 100018-0. doi: 10.3866/PKU.WHXB202310046
-
[11]
Yue Zhang , Bao Li , Lixin Wu . GO-Assisted Supramolecular Framework Membrane for High-Performance Separation of Nanosized Oil-in-Water Emulsions. Acta Physico-Chimica Sinica, 2024, 40(5): 2305038-0. doi: 10.3866/PKU.WHXB202305038
-
[12]
Ke Zhao , Zhen Liu , Luyao Liu , Changyuan Yu , Jingshun Pan , Xuguang Huang . Functionalized Reflective Structure Fiber-Optic Interferometric Sensor for Trace Detection of Lead Ions. Acta Physico-Chimica Sinica, 2024, 40(4): 2304029-0. doi: 10.3866/PKU.WHXB202304029
-
[13]
Qingtao CHEN , Xiangdong SHI , Xianghai RAO , Liying JIANG , Chunxiao JIA , Fenghua CHEN . Catalytic and in situ surface-enhanced Raman scattering detection properties of graphene oxide/gold nanorod assembly. Chinese Journal of Inorganic Chemistry, 2026, 42(1): 120-128. doi: 10.11862/CJIC.20250091
-
[14]
Qiaoqiao BAI , Anqi ZHOU , Xiaowei LI , Tang LIU , Song LIU . Construction of pressure-temperature dual-functional flexible sensors and applications in biomedicine. Chinese Journal of Inorganic Chemistry, 2024, 40(12): 2259-2274. doi: 10.11862/CJIC.20240128
-
[15]
Lin′an CAO , Dengyue MA , Gang XU . Research advances in electrically conductive metal-organic frameworks-based electrochemical sensors. Chinese Journal of Inorganic Chemistry, 2025, 41(10): 1953-1972. doi: 10.11862/CJIC.20250160
-
[16]
Xingchao Zhao , Xiaoming Li , Ming Liu , Zijin Zhao , Kaixuan Yang , Pengtian Liu , Haolan Zhang , Jintai Li , Xiaoling Ma , Qi Yao , Yanming Sun , Fujun Zhang . Photomultiplication-Type All-Polymer Photodetectors and Their Applications in Photoplethysmography Sensor. Acta Physico-Chimica Sinica, 2025, 41(1): 100007-0. doi: 10.3866/PKU.WHXB202311021
-
[17]
Jie XIE , Hongnan XU , Jianfeng LIAO , Ruoyu CHEN , Lin SUN , Zhong JIN . Nitrogen-doped 3D graphene-carbon nanotube network for efficient lithium storage. Chinese Journal of Inorganic Chemistry, 2024, 40(10): 1840-1849. doi: 10.11862/CJIC.20240216
-
[18]
Tian TIAN , Meng ZHOU , Jiale WEI , Yize LIU , Yifan MO , Yuhan YE , Wenzhi JIA , Bin HE . Ru-doped Co3O4/reduced graphene oxide: Preparation and electrocatalytic oxygen evolution property. Chinese Journal of Inorganic Chemistry, 2025, 41(2): 385-394. doi: 10.11862/CJIC.20240298
-
[19]
Jiarong Feng , Yejie Duan , Chu Chu , Dezhen Xie , Qiu'e Cao , Peng Liu . Preparation and Application of a Streptomycin Molecularly Imprinted Electrochemical Sensor: A Suggested Comprehensive Analytical Chemical Experiment. University Chemistry, 2024, 39(8): 295-305. doi: 10.3866/PKU.DXHX202401016
-
[20]
Yunting Shang , Yue Dai , Jianxin Zhang , Nan Zhu , Yan Su . Something about RGO (Reduced Graphene Oxide). University Chemistry, 2024, 39(9): 273-278. doi: 10.3866/PKU.DXHX202306050
-
[1]
Metrics
- PDF Downloads(39)
- Abstract views(3782)
- HTML views(529)

 Login In
Login In




 DownLoad:
DownLoad: