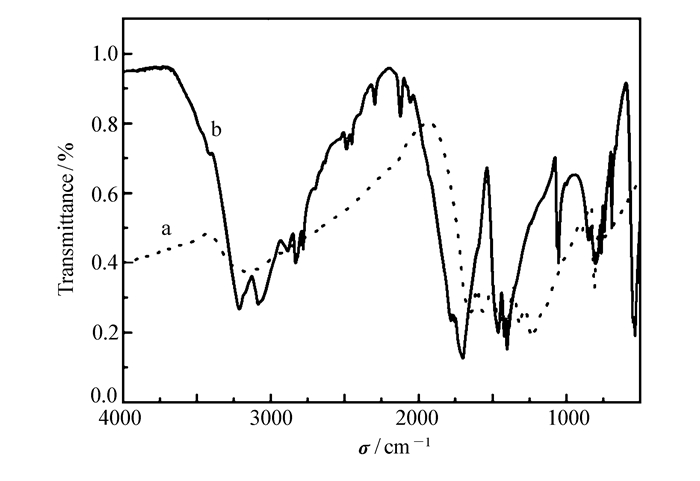
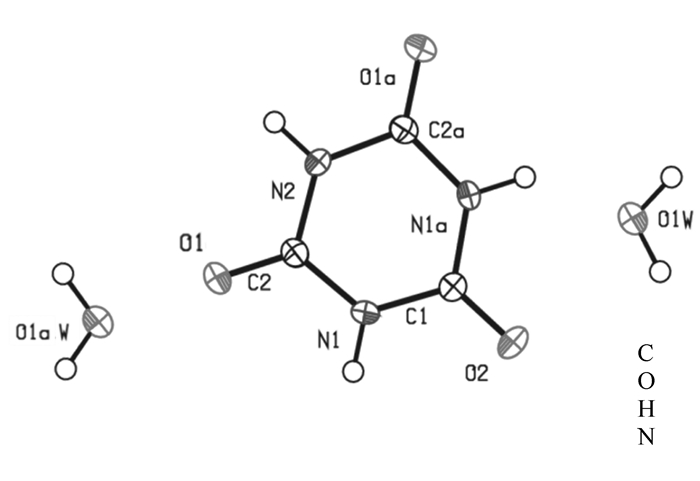
-
[1]
Patil S P, Padmanabhan D. A Facile Preparation of (2, 4, 6-14C)-Cyanuric Acid under Solvent-Free Conditions[J]. Label Compd Radiophar,
2002,45:539-542.
doi: 10.1002/jlcr.579
-
[2]
Liu L, Li X, Wang J. The Application of Trichlorisocyanuric Acid in Industrial Circulating Water[J]. Chem Eng Des Commun,
2017,43(12)3.
-
[3]
Onoda H, Takenaka A. Influence of Addition of Urea and Its Related Compounds on Formation of Various Neodymium and Cerium Phosphates[J]. Mater Chem Phys,
2003,82:194-198.
doi: 10.1016/S0254-0584(03)00205-0
-
[4]
Qiu Y, Gao L. Blue-Emitting Cyanuric Acid-Melamine Complexes from Urea Thermolysis[J]. Mater Res Bull,
2005,40(5):794-799.
doi: 10.1016/j.materresbull.2005.02.003
-
[5]
Thottempudi V, Shreeve J M. Synthesis and Promising Properties of a New Family of High-Density Energetic Salts of 5-Nitro-3-trinitromethyl-1H-1, 2, 4-Triazole and 5, 5'-Bis(trinitromethyl)-3, 3'-azo-1H-1, 2, 4-Triazole[J]. J Am Chem Soc,
2011,133(49):19982-19992.
doi: 10.1021/ja208990z
-
[6]
Gbel M, Kaeaghiosff K, Klap T M. Nitrotetrazolate-2N-oxides and the Strategy of N-Oxide Introduction[J]. J Am Chem Soc,
2010,132(48):17216-17226.
doi: 10.1021/ja106892a
-
[7]
Jooy H, Shreeve M. High-Density Energetic Mono- or Bis(oxy)-5-Nitroimino Tetrazoles[J]. Angew Chem Int Ed,
2010,49(40):7320-7323.
doi: 10.1002/anie.201003866
-
[8]
Hammerla A, Klaptket M, Rocha R. Azide-tetrazole Ring-Chain Isomerism in Polyazido-1, 3, 5-triazines, Triazido-s-heptazine, and Diazidotetrazines[J]. Eur J Inorg Chem,
2006,16(11):2210-2228.
-
[9]
Fischer A, Antonietti M, Thomas A. Growth Confined by the Nitrogen Source:Synthesis of Pure Methal Nitride Nanoparticles in Mesoporous Graphitic Carbon Nitride[J]. Adv Mater,
2007,9(2):264-267.
-
[10]
Banertk K, Jooy H, Rüffer T. The Exciting Chemistry of Tetraazidomethane[J]. Angew Chem Int Ed,
2007,46(7):1168-1171.
doi: 10.1002/anie.200603960
-
[11]
Zhang Q, Shreeve J M. Energetic Ionic Liquids as Explosives and Propellant Fuels:A New Journey of Ionic Liquid Chemistry[J]. Chem Rev,
2014,114(20):10527-10574.
doi: 10.1021/cr500364t
-
[12]
Yin P, Parrish D A, Shreeve J M. Bis(nitroamino-1, 2, 4-triazolates):N-Bridging Strategy Toward Insensitive Energetic Materials[J]. Angew Chem Int Ed,
2014,53(47):12889-12892.
doi: 10.1002/anie.201408127
-
[13]
Zhang Y, Parrish D A, Shreeve J M. Derivatives of 5-Nitro-1, 2, 3-2H-triazole-High Performance Energetic Materials[J]. J Mater Chem A,
2013,1(3):585-593.
doi: 10.1039/C2TA00136E
-
[14]
Yin P, Zhang Q, Zhang J. N-Trinitroethylamino Functionalization of Nitroimidazoles:A New Strategy for High Performance Energetic Materials[J]. J Mater Chem A,
2013,1(25):7500-7510.
doi: 10.1039/c3ta11356f
-
[15]
Wu Q, Zhu W, Xiao H. A New Design Strategy for High-Energy Low-Sensitivity Explosives:Combining Oxygen Balance Equal to Zero, a Combination of Nitro and Amino Groups, and N-Oxide in One Molecule of 1-Amino-5-nitrotetrazole-3N-oxide[J]. J Mater Chem A,
2014,2(32):13006-13015.
doi: 10.1039/C4TA01879F
-
[16]
Dippold A A, Klap T M. A Study of Dinitro-bis-1, 2, 4-triazole-1, 1'-diol and Derivatives:Design of High-Performance Insensitive Energetic Materials by the Introduction of N-Oxides[J]. J Am Chem Soc,
2013,135(26):9931-9938.
doi: 10.1021/ja404164j
-
[17]
Fisher N, Fisher D, Klap T M. Pushing the Limits of Energetic Materials-the Synthesis and Characterization of Dihydroxylammonium 5, 5'-Bistetrazole-1, 1'-Diolate[J]. J Mater Chem,
2012,22(38):20418-20422.
doi: 10.1039/c2jm33646d
-
[18]
Joo Y H, Shreeve J M. 1-Substituted 5-Aminotetrazoles:Syntheses from CNN3 with Primary Amines[J]. Org Lett,
2008,10(20):4665-4667.
doi: 10.1021/ol8019742
-
[19]
Joo Y H, Shreeve J M. Energetic Mono-, Di-, and Trisubstituted Nitroiminotetrazoles[J]. Angew Chem Int Ed,
2009,48(3):564-567.
doi: 10.1002/anie.200804755
-
[20]
Gerd, Janna, Klapotke. Synthesis, Properties and Dimerization Study of Isocyanic Acid[J]. Z Naturforsch B Chem Sci,
2002,57:19-24.
doi: 10.1515/znb-2002-0103
-
[21]
Chun H, Min S S. Methods and Devices for Preparing Biuret and Cyanuric Acid: CN Pat.10117296[P]. 2008.
-
[22]
She D, Yu H, Huang Q. Liquid-Phase Synthesis of Cyanuric Acid from Urea[J]. Molecules,
2010,15:1898-1902.
doi: 10.3390/molecules15031898
-
[23]
Kroke E. gt-C3N4-The First Stable Binary Carbon(Ⅳ) Nitride[J]. Angew Chem Int Ed,
2014,53:11134-11136.
doi: 10.1002/anie.201406427
-
[24]
Yan S C, Li Z S, Zou Z G. Photodegradation Performance of g-C3N4 Fabricated by Directly Heating Melamine[J]. Langmuir,
2009,25(17):10397-10401.
doi: 10.1021/la900923z
-
[25]
WANG Baopin. The Methods of Cyanuric Acid Degradation and Its Industrial Application[D]. Beijing: Beijing University of Chemical Technology, 2013(in Chinese).
-
[26]
MA Luye. Study on Synthesis and Application of Cyanuric Acid[D]. Jinan: Shandong University, 2017(in Chinese).
-
[27]
HU Yangyong. Research on Thermostabiliity of High Temperature and High Concentration Ammonium Nitrate Solution[D]. Huainan: AnHui University of Science and Technology, 2014(in Chinese).

 Login In
Login In

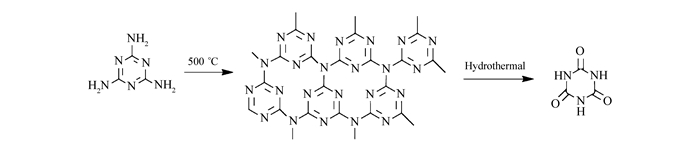





 DownLoad:
DownLoad: