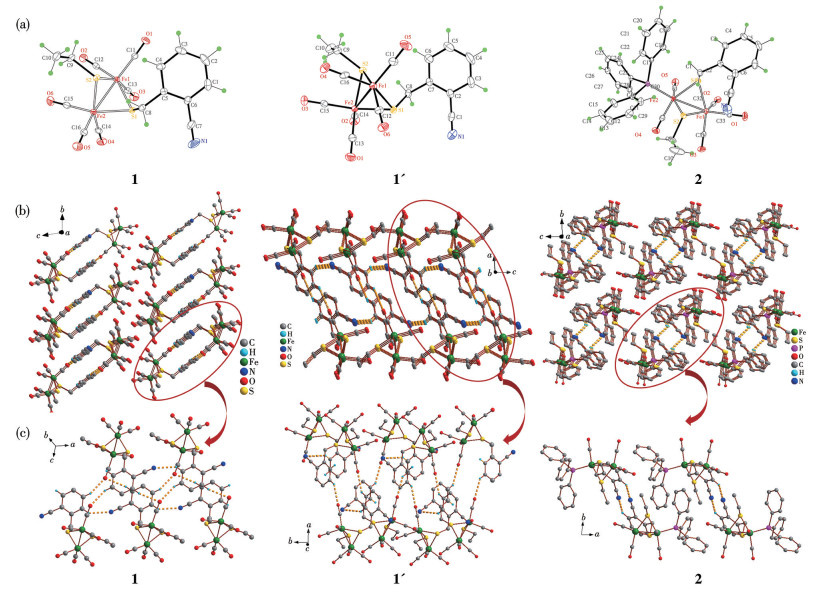
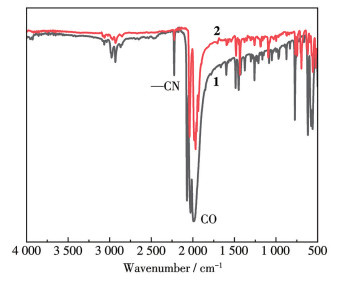
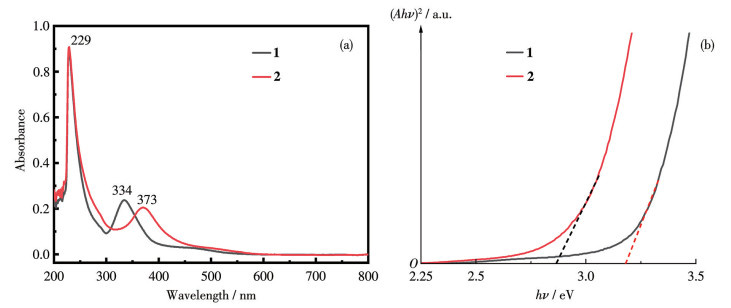
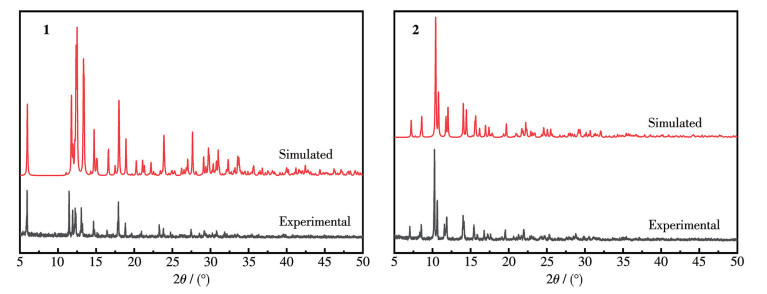
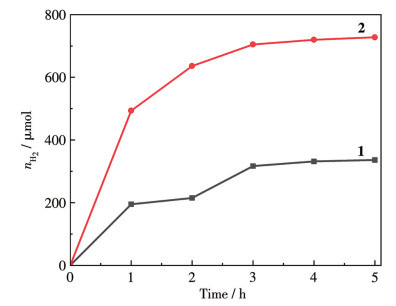
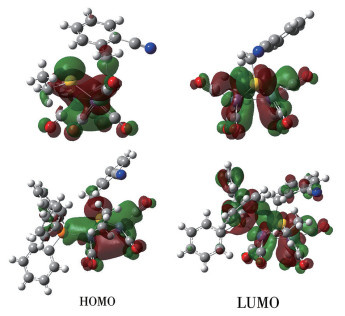
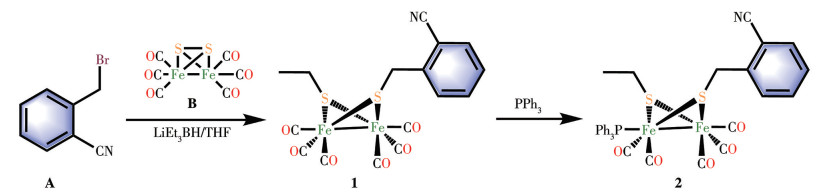
-
[1]
ALI S, ASHRAF J, GHUFRAN M, PENG X B. The role of market forces and regulatory factors on green innovation in China: A panel data analysis[J]. Environ. Dev. Sustain., 2025, DOI:10.1007/s10668-025-06475-y
doi: 10.1007/s10668-025-06475-y
-
[2]
MIDILLI A, AY M, DINCER I, ROSEN M A. On hydrogen and hydrogen energy strategies Ⅰ: Current status and needs[J]. Renewable Sustainable Energy Rev., 2005, 9(3): 255-271
doi: 10.1016/j.rser.2004.05.003
-
[3]
NOCERA D G. Solar fuels and solar chemicals industry[J]. Accounts Chem. Res., 2017, 50(3): 616-619
doi: 10.1021/acs.accounts.6b00615
-
[4]
GRAY H B. Powering the planet with solar fuel[J]. Abstracts of papers of the American Chemical Society, 2013, 246: 9-motion
-
[5]
MAEDA K, TERAMURA K, LU D L, TAKATA T, SAITO N, INOUE Y, DOMEN K. Photocatalyst releasing hydrogen from water‒Enhancing catalytic performance holds promise for hydrogen production by water splitting in sunlight[J]. Nature, 2006, 440(7082): 295
doi: 10.1038/440295a
-
[6]
ZOU Z G, YE J H, SAYAMA K, ARAKAWA H. Direct splitting of water under visible light irradiation with an oxide semiconductor photocatalyst[J]. Nature, 2001, 414(6864): 625-627
doi: 10.1038/414625a
-
[7]
SIDABRAS J W, STRIPP S T. A personal account on 25 years of scientific literature on [FeFe]-hydrogenase[J]. J. Biol. Inorg. Chem., 2023, 28(4): 355-378
doi: 10.1007/s00775-023-01992-5
-
[8]
CASTNER A T, JOHNSON B A, COHEN S M, OTT S. Mimicking the electron transport chain and active site of [FeFe] hydrogenases in one metal-organic framework: Factors that influence charge transport[J]. J. Am. Chem. Soc., 2021, 143(21): 7991-7999
doi: 10.1021/jacs.1c01361
-
[9]
HESSLER T, HARRISON S T L, BANFIELD J F, HUDDY R J. Harnessing fermentation may enhance the performance of biological sulfate-reducing bioreactors[J]. Environ. Sci. Technol., 2024, 58(6): 2830-2846
doi: 10.1021/acs.est.3c04187
-
[10]
YADAV S, HAAS R, BOYDAS E B, REOMELT M, HAPPE T, APFEL U P, STRIPP S T. Oxygen sensitivity of [FeFe]-hydrogenase: A comparative study of active site mimics inside vs. outside the enzyme[J]. Phys. Chem. Chem. Phys., 2024, 26(28): 19105-19116
doi: 10.1039/D3CP06048A
-
[11]
REALINI F, ELLEOUET C, PÉTILLOM F Y, SCHOLLHAMMER P. Tri- and tetra-substituted derivatives of [Fe2(CO)6(μ-dithiolate)] as novel dinuclear platforms related to the H-cluster of [FeFe]H2ases[J]. Eur. J. Inorg. Chem., 2022(19): e202200133
-
[12]
AGUADO S, GÓMEZ-GALLEGO M, GARCÍA-ÁLVAREZ P, CABEZA J A, SIERRAM A. Transition metal-NHC complexes with embedded [FeFe]-hydrogenase mimics[J]. Organometallics, 2025, 44(14): 1576-1585
doi: 10.1021/acs.organomet.5c00197
-
[13]
SANO Y, ONODA A, HAYASHI T. A hydrogenase model system based on the sequence of cytochrome c: Photochemical hydrogen evolution in aqueous media[J]. Chem. Commun., 2011, 47(29): 8229-8231
doi: 10.1039/c1cc11157d
-
[14]
PULLEN S, FEI H H, ORTHABER A, COHEN S M, OTT S. Enhanced photochemical hydrogen production by a molecular diiron catalyst incorporated into a metal-organic framework[J]. J. Am. Chem. Soc., 2013, 135(45): 16997-17003
doi: 10.1021/ja407176p
-
[15]
VÖLLER J S. Air-stable [FeFe] hydrogenases[J]. Nat. Catal., 2018, 1(8): 564
doi: 10.1038/s41929-018-0137-y
-
[16]
KARAYILAN M, BREZINSKI W P, CLARY K E, LICHTENBERGER D L, GLASS R S, PYUN J. Catalytic metallopolymers from [2Fe-2S] clusters: Artificial metalloenzymes for hydrogen production[J]. Angew. Chem. ‒Int. Edit., 2019, 58(23): 7537-7550
doi: 10.1002/anie.201813776
-
[17]
GAO S, LIU Y, SHAO Y D, JIANG D Y, DUAN Q. Iron carbonyl compounds with aromatic dithiolate bridges as organometallic mimics of [FeFe] hydrogenises[J]. Coord. Chem. Rev., 2020, 402: 213081
doi: 10.1016/j.ccr.2019.213081
-
[18]
JIN G X, WANG F B, ZHAO H R, WANG X H, LI Y L, SUN Y, CHENG J Y, SHENG X H, WANG H Y, MA J P, LIU Q K. [Fe2S2]-hydrogenase-mimic-containing supramolecule and coordination polymers: Syntheses, H2 evolution properties, and their structure-function relationship study[J]. Cryst. Growth Des., 2024, 24(7): 2667-2671
doi: 10.1021/acs.cgd.4c00144
-
[19]
MERINERO A D, COLLADO A, CASARRUBIOS L, GÓMEZ- GALLEGO M, RAMÍREZ DE ARELLANO C, ARELLANO C, CABALLERO A, ZAPATA F, SIERRA M A. Triazole-containing [FeFe] hydrogenase mimics: Synthesis and electrocatalytic behavior[J]. Inorg. Chem., 2019, 58(23): 16267-16278
doi: 10.1021/acs.inorgchem.9b02813
-
[20]
LU L X, LIU S X, XU J, JIN Z Q, CHENG J J, ZHAO J Y, WANG F B, WANG H Y. [FeFe]-hydrogenase-containing compound and its photocatalytic H2-production performance[J]. Chinese J. Inorg. Chem., 2025, 41(12): 2584-2590
-
[21]
JIN G X, HAN C C, ZHAO H R, WU X W, LI Y L, WANG H Y, MA J P. Small-molecules-induced metal-organic-framework-based photosensitizer for greatly enhancing H2 production efficiency[J]. ACS Mater. Lett., 2024, 6(2): 375-383
doi: 10.1021/acsmaterialslett.3c01286
-
[22]
CHEN X H, YANG F, HAN C C, HAN L C, WANG F B, JIN G X, WANG H Y, MA J P. [Fe2S2-Agx]-hydrogenase active-site-containing coordination polymers and their photocatalytic H2 evolution reaction properties[J]. Inorg. Chem., 2022, 61(34): 13261-13265
doi: 10.1021/acs.inorgchem.2c01818
-
[23]
COSTENTIN C, DROUET S, ROBERT M, SAVÉANT J M. Turnover numbers, turnover frequencies, and overpotential in molecular catalysis of electrochemical reactions. Cyclic voltammetry and preparative-scale electrolysis[J]. J. Am. Chem. Soc., 2012, 134(27): 11235-11242
doi: 10.1021/ja303560c

 Login In
Login In






 DownLoad:
DownLoad: