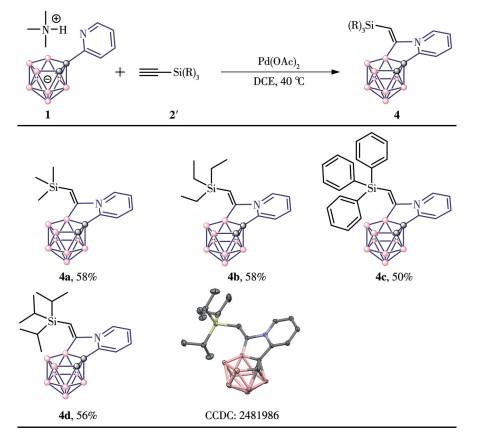
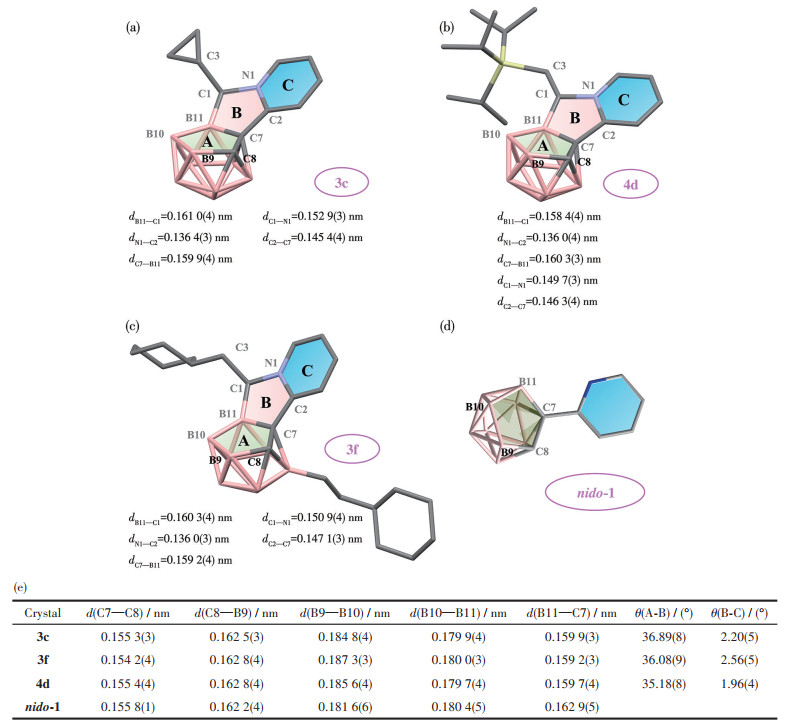
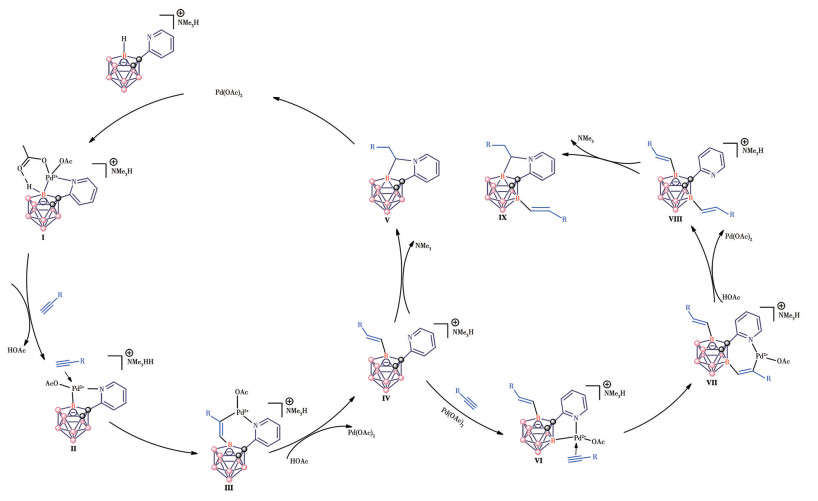
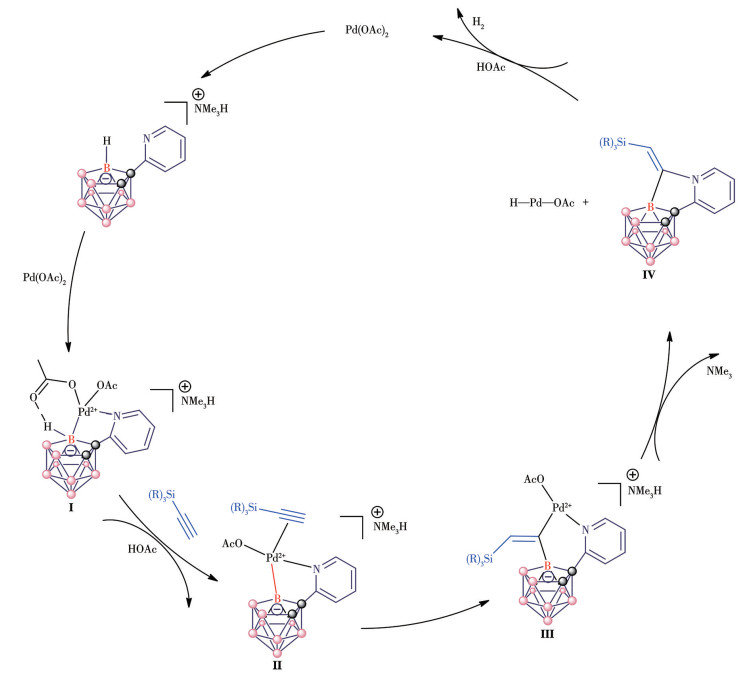
-
[1]
ITO H, OZAKI K, ITAMI K. Annulative π-extension (APEX): Rapid access to fused arenes, heteroarenes, and nanographenes[J]. Angew. Chem. ‒Int. Edit.,
2017,56(37):11144-11164.
doi: 10.1002/anie.201701058
-
[2]
VON GROTTHUSS E, JOHN A, KAESE T, WAGNER M. Doping polycyclic aromatics with boron for superior performance in materials science and catalysis[J]. Asian J. Org. Chem.,
2017,7(1):37-53.
-
[3]
WANG Z S, JIANG L F, JI J W, ZHOU F L, LAN J B, YOU J S. Construction of cationic azahelicenes: Regioselective three-component annulation using in situ activation strategy[J]. Angew. Chem. ‒Int. Edit.,
2020,59(52):23532-23536.
doi: 10.1002/anie.202010051
-
[4]
BORISSOV A, MAURYA Y K, MOSHNIAHA L, WONG WS, ŻYŁA-KARWOWSKA M, STĘPIEŃ M. Recent advances in heterocyclic nanographenes and other polycyclic heteroaromatic compounds[J]. Chem. Rev.,
2021,122(1):565-788.
-
[5]
ALZAHRANI A Z. First-principles study on the structural and electronic properties of graphene upon benzene and naphthalene adsorption[J]. Appl. Surf. Sci.,
2010,257(3):807-810.
doi: 10.1016/j.apsusc.2010.07.069
-
[6]
CHEN F, TAO N J. Electron transport in single molecules: From benzene to graphene[J]. Accounts Chem. Res.,
2009,42(3):429-438.
doi: 10.1021/ar800199a
-
[7]
DOMENICO J, SCHNEIDER A M, SOHLBERG K. From benzene to graphene: Exploring the electronic structure of single-layer and bilayer graphene using polycyclic aromatic hydrocarbons[J]. J. Chem. Educ.,
2019,96(10):2225-2237.
doi: 10.1021/acs.jchemed.9b00331
-
[8]
CHI X, ZHANG Z X, LI M Q, JIAO Y, LI X M, MENG F C, XUE B, WU D Q, ZHANG F. Vinylene-linking of polycyclic aromatic hydrocarbons to π-extended two-dimensional covalent organic framework photocatalyst for H2O2 synthesis[J]. Angew. Chem. ‒Int. Edit.,
2024,64(7)e202418895.
-
[9]
GRIMES R N. Carboranes[M]. [S. l. ]: Academic Press, 2016: 7-17
-
[10]
SOLA M. Aromaticity rules[J]. Nat. Chem.,
2022,14(6):585-590.
doi: 10.1038/s41557-022-00961-w
-
[11]
POATER J, VIÑAS C, OLID D, SOLÀ M, TEIXIDOR F. Aromaticity and extrusion of benzenoids linked to [o-COSAN]-: Clar has the answer[J]. Angew. Chem. ‒Int. Edit.,
2022,61(22)e202200672.
doi: 10.1002/anie.202200672
-
[12]
GAO Y H, SZATHMÁRI B, BUZSÁKI D, KELEMEN Z. Reassessing the possibility of π-σ-π full electron delocalization through 3D aromatic carboranes[J]. Chem. Eur. J.,
2025,31(41)e202501806.
doi: 10.1002/chem.202501806
-
[13]
MUKHERJEE S, THILAGAR P. Boron clusters in luminescent materials[J]. Chem. Commun.,
2016,52(6):1070-1093.
doi: 10.1039/C5CC08213G
-
[14]
OCHI J, TANAKA K, CHUJO Y. Recent progress in the development of solid-state luminescent o-carboranes with stimuli responsivity[J]. Angew. Chem. ‒Int. Edit.,
2020,59(25):9841-9855.
doi: 10.1002/anie.201916666
-
[15]
FISHER S P, TOMICH A W, LOVERA S O, KLEINSASSER J F, GUO J, ASAY M J, NELSON H M, LAVALLO V. Nonclassical applications of closo-carborane anions: From main group chemistry and catalysis to energy storage[J]. Chem. Rev.,
2019,119(14):8262-8290.
doi: 10.1021/acs.chemrev.8b00551
-
[16]
WANG Q Y, WANG J, WANG S, WANG Z Y, CAO M, HE C L, YANG J Q, ZANG S Q, MAK T C W. o-Carborane-based and atomically precise metal clusters as hypergolic materials[J]. J. Am. Chem. Soc.,
2020,142(28):12010-12014.
doi: 10.1021/jacs.0c04638
-
[17]
WANG Z L, GOU X Y, SHI Q Y, LIU K, CHANG X M, WANG G, XU W J, LIN S M, LIU T H, FANG Y. Through-space charge transfer: A new way to develop a high-performance fluorescence sensing film towards opto-electronically inert alkanes[J]. Angew. Chem. ‒Int. Edit.,
2022,61(35)e202207619.
doi: 10.1002/anie.202207619
-
[18]
CHEN M, SUN Z F, WANG L Y, ZONG J B, WEI G F, LU C S, TANG S, TU D S, YAN H. Direct B—H bond activation polymerization of boron clusters[J]. J. Am. Chem. Soc.,
2025,147(47):43946-43956.
doi: 10.1021/jacs.5c16451
-
[19]
ZHOU Y T, CHENG K, LIU B, CAO Y C, FAN J X, LIU Z G, ZHAO Y D. Recent progress of nano-drugs in neutron capture therapy[J]. Theranostics,
2024,14(8):3193-3212.
doi: 10.7150/thno.95034
-
[20]
MA W L, WANG Y Y, XUE Y L, WANG M M, LU C S, GUO W H, LIU Y H, SHU D Y, SHAO G Q, XU Q F, TU D S, YAN H. Molecular engineering of AIE-active boron clustoluminogens for enhanced boron neutron capture therapy[J]. Chem. Sci.,
2024,15(11):4019-4030.
doi: 10.1039/D3SC06222H
-
[21]
MA W L, ZHANG J Y, ZONG J B, REN H Y, TU D S, XU Q F, ZHONG TANG B, YAN H. Luminescence modulation in boron-cluster-based luminogens via boron isotope effects[J]. Angew. Chem. ‒Int. Edit.,
2024,63(52)e202410430.
doi: 10.1002/anie.202410430
-
[22]
KREBS J, HÄFNER A, FUCHS S, GUO X, RAUCH F, EICHHORN A, KRUMMENACHER I, FRIEDRICH A, JI L, FINZE M, LIN Z, BRAUNSCHWEIG H, MARDER T B. Backbone-controlled LUMO energy induces intramolecular C—H activation in ortho-bis-9-borafluorene-substituted phenyl and o-carboranyl compounds leading to novel 9, 10-diboraanthracene derivatives[J]. Chem. Sci.,
2022,13(47):14165-14178.
doi: 10.1039/D2SC06057D
-
[23]
JI L, RIESE S, SCHMIEDEL A, HOLZAPFEL M, FEST M, NITSCH J, CURCHOD B F E, FRIEDRICH A, WU L, AL MAMARI H H, HAMMER S, PFLAUM J, FOX M A, TOZER D J, FINZE M, LAMBERT C, MARDER T B. Thermodynamic equilibrium between locally excited and charge-transfer states through thermally activated charge transfer in 1-(pyren-2′-yl)-o-carborane[J]. Chem. Sci.,
2022,13(18):5205-5219.
doi: 10.1039/D1SC06867A
-
[24]
ZHANG C H, LIU X C, WANG J Y, YE Q. A three-dimensional inorganic analogue of 9, 10-diazido-9, 10-diboraanthracene: A Lewis superacidic azido borane with reactivity and stability[J]. Angew. Chem. ‒Int. Edit.,
2022,61(36)e202205506.
doi: 10.1002/anie.202205506
-
[25]
YRUEGAS S, AXTELL J C, KIRLIKOVALI K O, SPOKOYNY A M, MARTIN C D. Synthesis of 9-borafluorene analogues featuring a three‑dimensional 1, 1′‑bis(o‑carborane) backbone[J]. Chem. Commun.,
2019,55(20):2892-2895.
doi: 10.1039/C8CC10087J
-
[26]
AXTELL J C, KIRLIKOVALI K O, DJUROVICH P I, JUNG D, NGUYEN V T, MUNEKIYO B, ROYAPPA A T, RHEINGOLD A L, SPOKOYNY A M. Blue phosphorescent zwitterionic iridium complexes featuring weakly coordinating nido-carborane-based ligands[J]. J. Am. Chem. Soc.,
2016,138(48):15758-15765.
doi: 10.1021/jacs.6b10232
-
[27]
PARK K, BAE G, MOON J, CHOE J, SONG K H, LEE S. Synthesis of symmetrical and unsymmetrical diarylalkynes from propiolic acid using palladium-catalyzed decarboxylative coupling[J]. Org. Chem.,
2010,75(18):6244-6251.
doi: 10.1021/jo101398a
-
[28]
DOUCET H, HIERSO J C. Palladium-based catalytic systems for the synthesis of conjugated enynes by Sonogashira reactions and related alkynylations[J]. Angew. Chem. ‒Int. Edit.,
2007,46(6):834-871.
doi: 10.1002/anie.200602761
-
[29]
DOLOMANOV O V, BOURHIS L J, GILDEA R J, HOWARD J A K, PUSCHMANN H. OLEX2: A complete structure solution, refinement and analysis program[J]. J. Appl. Crystallogr.,
2009,42(2):339-341.
doi: 10.1107/S0021889808042726
-
[30]
DOLOMANOV O V, BOURHIS L J, GILDEA R J, HOWARD J A K, PUSCHMANN H. Tables of bond lengths determined by X-ray and neutron diffraction. Part 1. Bond lengths in organic compounds[J]. J. Chem. Soc. Perkin Trans. 2,
1987,12:S1-S19.
-
[31]
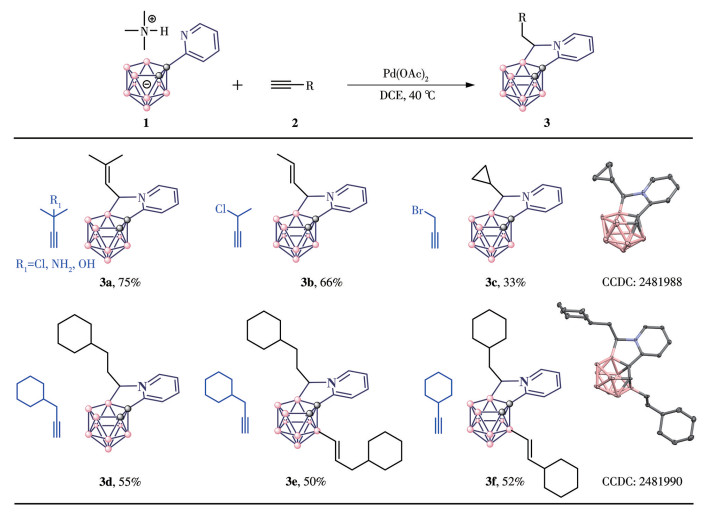
SUN F X, TAN S M, CAO H J, XU J K, BREGADZE V I, TU D S, LU C S, YAN H. Palladium-catalyzed hydroboration of alkynes with carboranes: Facile construction of a library of boron cluster-based AIE-active luminogens[J]. Angew. Chem. ‒Int. Edit.,
2022,61(33)e202207125.
doi: 10.1002/anie.202207125
-
[32]
SUN F X, TAN S M, CAO H J, LU C S, TU D S, POATER J, SOLÀ M, YAN H. Facile construction of new hybrid conjugation via boron cage extension[J]. J. Am. Chem. Soc.,
2023,145(6):3577-3587.
doi: 10.1021/jacs.2c12526
-
[33]
AL MASUM M, MEGURO M, YAMAMOTO Y. The two component palladium catalyst system for intermolecular hydroamination of allenes[J]. Tetrahedron Lett.,
1997,38(34):6071-6074.
doi: 10.1016/S0040-4039(97)01370-1
-
[34]
WARRATZ S, KORNHAASS C, CAJARAVILLE A, NIEPÖTTER B, STALKE D, ACKERMANN L. Ruthenium(Ⅱ)-catalyzed C—H activation/alkyne annulation by weak coordination with O2 as the sole oxidant[J]. Angew. Chem. ‒Int. Edit.,
2015,54(18):5513-5517.
doi: 10.1002/anie.201500600
-
[35]
MATHER B D, VISWANATHAN K, MILLER K M, LONG T E. Michael addition reactions in macromolecular design for emerging technologies[J]. Prog. Polym. Sci.,
2006,31(5):487-531.
doi: 10.1016/j.progpolymsci.2006.03.001
-
[36]
TEJEDOR D, MÉNDEZ-ABT G, COTOS L, GARCÍA-TELLADO F. Propargyl Claisen rearrangement: Allene synthesis and beyond[J]. Chem. Soc. Rev.,
2013,42(2):458-471.
doi: 10.1039/C2CS35311C
-
[37]
MURAI M, NISHIMURA K, TAKAI K. Palladium-catalyzed double-bond migration of unsaturated hydrocarbons accelerated by tantalum chloride[J]. Chem. Commun.,
2019,55(19):2769-2772.
doi: 10.1039/C9CC00223E

 Login In
Login In






 DownLoad:
DownLoad: