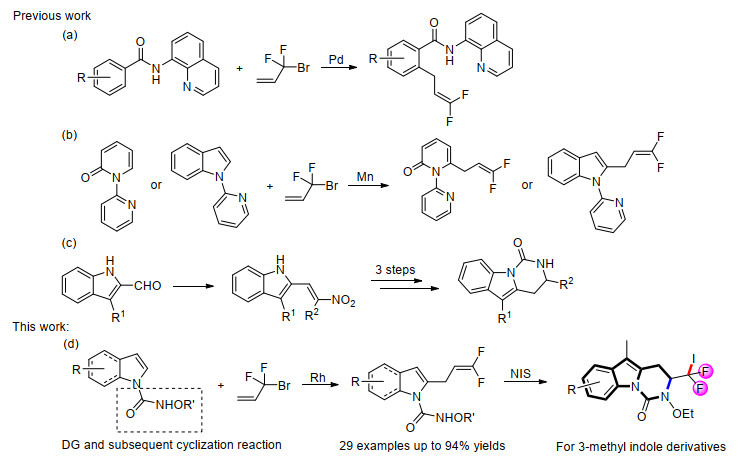
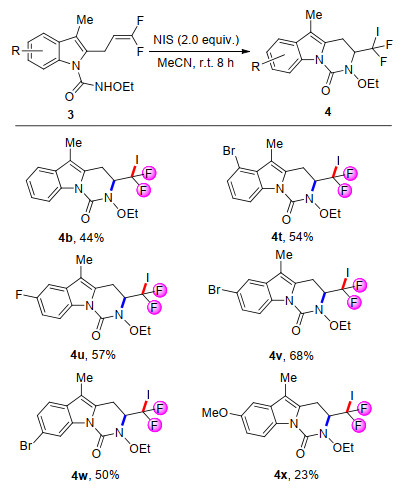
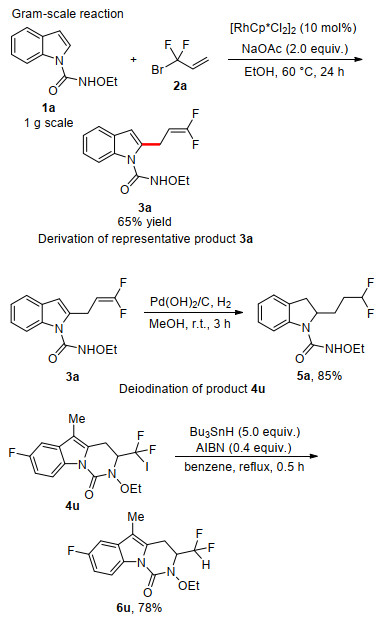
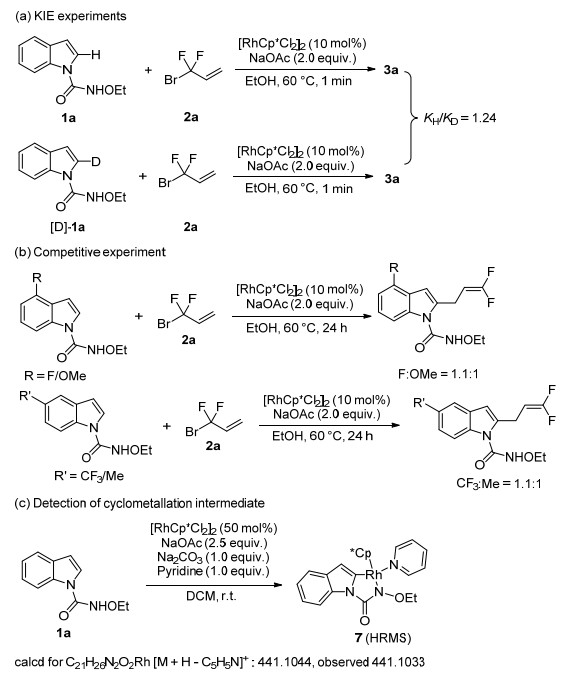
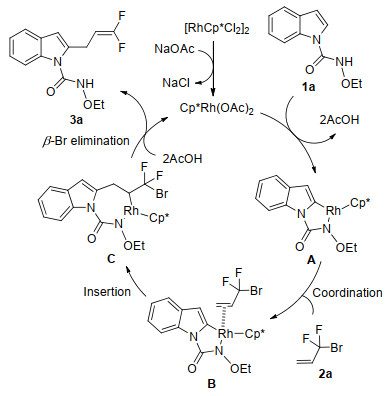
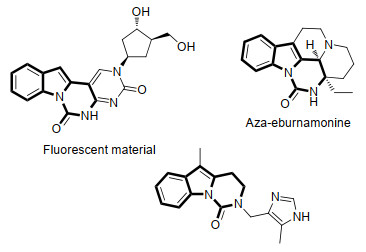
-
[1]
(a) Mizuta, M.; Seio, K.; Miyata, K.; Sekine, M. J. Org. Chem. 2007, 72, 5046.
(b) Bandurco, V. T.; Wong, E. M.; Levine, S. D.; Hajos, Z. G. J. Med. Chem. 1981, 24, 1455.
(c) Hammer, H.; Winterfeldt, E. Tetrahedron 1981, 37, 3609.
(d) Kato, M.; Nishino, S.; Iro, K.; Yamakuni, H.; Takasugi, H. Chem. Pharm. Bull. 1994, 42, 2556.
(e) Wright, W. B.; Brabander, H. J. J. Med. Chem. 1968, 11, 1164.
-
[2]
(a) Zhang, Y.; Zheng, J.; Cui, S. J. Org. Chem. 2014, 79, 6490.
(b) Zheng, J.; Zhang, Y.; Cui, S. Org. Lett. 2014, 16, 3560.
(c) Chen, X.; Hu, X.; Bai, S.; Deng, Y.; Jiang, H.; Zeng, W. Org. Lett. 2016, 18, 192.
-
[3]
(a) Muller, K.; Faeh, C.; Diederich, F. Science 2007, 317, 1881.
(b) Hagmann, W. K. J. Med. Chem. 2008, 51, 4359.
(c) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320.
(d) Wang, J.; Sánchez-Roselló, M.; Aceña, J. L.; del Pozo, C.; Sorochinsky, A. E.; Fustero, S.; Soloshonok, V. A.; Liu, H. Chem. Rev. 2014, 114, 2432.
(e) Zhou, Y.; Wang, J.; Gu, Z.; Wang, S.; Zhu, W.; Aceña, J. L.; Soloshonok, V. A.; Izawa, K.; Liu, H. Chem. Rev. 2016, 116, 422.
-
[4]
(a) Price, D. A.; Armour, D.; de Groot, M.; Leishman, D.; Napier, C.; Perros, M.; Stammen, B. L.; Wood, A. Bioorg. Med. Chem. Lett. 2006, 16, 4633.
(b) Xue, F.; Li, H.; Delker, S. L.; Fang, J.; Martásek, P.; Roman, L. J.; Poulos, T. L.; Silverman, R. B. J. Am. Chem. Soc. 2010, 132, 14229.
(c) Zhou, Q.; Ruffoni, A.; Gianatassio, R.; Fujiwara, Y.; Sella, E.; Shabat, D.; Baran, P. S. Angew. Chem., Int. Ed. 2013, 52, 3949.
(d) Massa, M. A.; Spangler, D. P.; Durley, R. C.; Hickory, B. S.; Connolly, D. T.; Witherbee, B. J.; Smith, M. E.; Sikorski, J. A. Bioorg. Med. Chem. Lett. 2001, 11, 1625.
-
[5]
Middleton, W. J.; Bingham, E. M. J. Org. Chem. 1980, 45, 2883.
doi: 10.1021/jo01302a025
-
[6]
(a) Fujita, T.; Sugiyama, K.; Sanada, S.; Ichitsuka, T.; Ichikawa, J. Org. Lett. 2016, 18, 248.
(b) S. Akiyama, S.; Kubota, K.; Mikus, M. S.; Paioti, P. H. S.; Romiti, F.; Liu, Q.; Zhou, Y.; Hoveyda, A. H.; Ito, H. Angew. Chem., Int. Ed. 2019, 58, 11998.
(c) Cogswell, T. J.; Dahlen A.; Knerr, L. Chem.-Eur. J. 2019, 25, 1184.
-
[7]
Min, Q. Q.; Yin, Z.; Feng, Z.; Guo, W. H.; Zhang, X. J. Am. Chem. Soc. 2014, 136, 1230.
doi: 10.1021/ja4114825
-
[8]
Li, C.; Zhang, D.; Zhu, W.; Wan, P.; Liu, H. Org. Chem. Front. 2016, 3, 1080.
doi: 10.1039/C6QO00178E
-
[9]
Ni, J.; Zhao, H.; Zhang, A. Org. Lett. 2017, 19, 3159.
doi: 10.1021/acs.orglett.7b01282
-
[10]
(a) Wu, X.; Wang, B.; Zhou, S.; Zhou, Y.; Liu, H. ACS Catal. 2017, 7, 2494.
(b) Wu, X.; Wang, B.; Zhou, Y.; Liu, H. Org. Lett. 2017, 19, 1294.
(c) Xie, Y.; Wu, X.; Li, C.; Wang, J.; Li, J.; Liu, H. J. Org. Chem. 2017, 82, 5263.
(d) Fei, X.; Li, C.; Yu X.; Liu, H. J. Org. Chem. 2019, 84, 6840.
(e) Yang, L.; Li, C.; Wang, D.; Liu, H. J. Org. Chem. 2019, 84, 7320.
-
[11]
(a) Dai, H.; Yu, C.; Wang, Z.; Yan, H.; Lu, C. Org. Lett. 2016, 18, 3410.
(b) Dutta, U.; Deb, A.; Lupton, D. W.; Maiti, D. Chem. Commun. 2015, 51, 17744.
-
[12]
(a) Zhang, L.; Zhu, M.; Ni, S.; Wen, L.; Li, M. ACS Catal. 2019, 9, 1680.
(b) Kong, W.; Chen, X.; Wang, M.; Dai, H.; Yu, J. Org. Lett. 2018, 20, 284.
-
[13]
Nihei, T.; Iwai, N.; Matsuda, T.; Kitazume, T. J. Org. Chem. 2005, 70, 5912.
doi: 10.1021/jo050634u

 Login In
Login In






 DownLoad:
DownLoad: