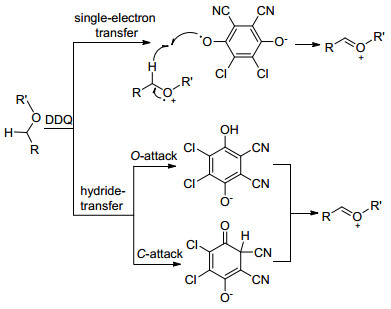
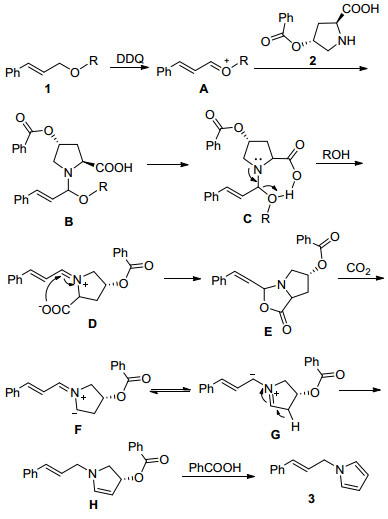
-
[1]
Curran, D.; Grimshaw, J.; Perera, S. D. Chem. Soc. Rev. 1991, 20, 391.
doi: 10.1039/cs9912000391
-
[2]
(a) Azizian, J.; Karimi, A. R.; Kazemizadeh, Z.; Mohammadi, A. A.; Mohammadizadeh, M. R. J. Org. Chem. 2005, 70, 1471.
(b) Meshram, H. M.; Prasad, B. R. V.; Aravind Kumar, D. Tetrahedron Lett. 2010, 51, 3477.
(c) Deb, I.; Coiro, D. J.; Seidel, D. Chem. Commun. 2011, 47, 6473.
(d) Mao, H.; Wang, S.; Yu, P.; Lv, H.; Xu, R.; Pan, Y. J. Org. Chem. 2011, 76, 1167.
(e) Zou, Z.; Deng, Z.; Yu, X.; Zhang, M.; Zhao, S.; Luo, T.; Yin, X.; Xu, H.; Wang, W. Sci. China Chem. 2012, 55, 43.
(f) Satish, G.; Reddy, K. H. V.; Ramesh, K.; Kumar, B. S. P. A.; Nageswar, Y. V. D. Tetrahedron Lett. 2014, 55, 2596.
-
[3]
(a) Miller, A. J. M.; Kaminsky, W.; Goldberg, K. I. Organometallics 2014, 33, 1245.
(b) Song, Z.-Z.; Gong, J.-L.; Zhang, M.; Wu, X.-F. Asian J. Org. Chem. 2012, 1, 214.
(c) Shen, Z.; Dai, J.; Xiong, J.; He, X.; Mo, W.; Hu, B.; Sun, N.; Hu, X. Adv. Synth. Catal. 2011, 353, 3031.
-
[4]
(a) Zhang, Y.; Li, C.-J. Angew. Chem., Int. Ed. 2006, 45, 1949.
(b) Zhang, Y.; Li, C.-J. J. Am. Chem. Soc. 2006, 128, 4242.
(c) Tu, W.; Liu, L.; Floreancig, P. E. Angew. Chem., Int. Ed. 2008, 47, 4184.
(d) Tu, W.; Floreancig, P. E. Angew. Chem., Int. Ed. 2009, 48, 4567.
(e) Liu, L.; Floreancig, P. E. Angew. Chem., Int. Ed. 2010, 49, 5894.
(f) Richter, H.; Rohlmann, R.; García Mancheño, O. Chem.-Eur. J. 2011, 17, 11622.
(g) Muramatsu, W.; Nakano, K.; Li, C.-J. Org. Lett. 2013, 15, 3650.
(h) Meng, Z.; Sun, S.; Yuan, H.; Lou, H.; Liu, L. Angew. Chem. Int. Ed. 2014, 53, 543.
(i) Chen, W.; Xie, Z.; Zheng, H.; Lou, H.; Liu, L. Org. Lett. 2014, 16, 5988.
(j) Yu, B.; Jiang, T.; Li, J.; Su, Y.; Pan, X.; She, X. Org. Lett. 2009, 11, 3442.
(k) Liu, L.; Floreancig, P. E. Org. Lett. 2010, 12, 4686.
(l) Liu, X.; Sun, B.; Xie, Z.; Qin, X.; Liu, L.; Lou, H. J. Org. Chem. 2013, 78, 3104.
(m) Sun, K.; Wang, X.; Li, G.; Zhu, Z.; Jiang, Y.; Xiao, B. Chem. Commun. 2014, 50, 12880.
(n) Pandit, R. P.; Lee, Y. R. Adv. Synth. Catal. 2014, 356, 3171.
-
[5]
(a) Reisman, S. E.; Doyle, A. G.; Jacobsen, E. N. J. Am. Chem. Soc. 2008, 130, 7198.
(b) McQuaid, K. M.; Sames, D. J. Am. Chem. Soc. 2009, 131, 402.
(c) Maity, P.; Srinivas, H. D.; Watson, M. P. J. Am. Chem. Soc. 2011, 133, 17142.
(d) Chen, Y.; Chen, M.; Liu, Y. Angew. Chem. 2012, 124, 6599.
(e) Rueping, M.; Volla, C. M. R.; Atodiresei, I. Org. Lett. 2012, 14, 4642.
(f) Lu, C.; Su, X.; Floreancig, P. E. J. Org. Chem. 2013, 78, 9366.
(g) Srinivas, H. D.; Maity, P.; Yap, G. P. A.; Watson, M. P. J. Org. Chem. 2015, 80, 4003.
(h) Kong, A.; Andreansky, E. S.; Blakey, S. B. J. Org. Chem. 2017, 82, 4477.
(i) Fan, W.-T.; Li, N.-K.; Xu, L.; Qiao, C.; Wang, X.-W. Org. Lett. 2017, 19, 6626.
(j) Lee, S.; Kaib, P. S. J.; List, B. J. Am. Chem. Soc. 2017, 139, 2156.
-
[6]
(a) Zhang, S.-Y.; Zhang, F.-M.; Tu, Y.-Q. Chem. Soc. Rev. 2011, 40, 1937.
(b) Morales-Rivera, C. A.; Floreancig, P. E.; Liu, P. J. Am. Chem. Soc. 2017, 139, 17935.

 Login In
Login In

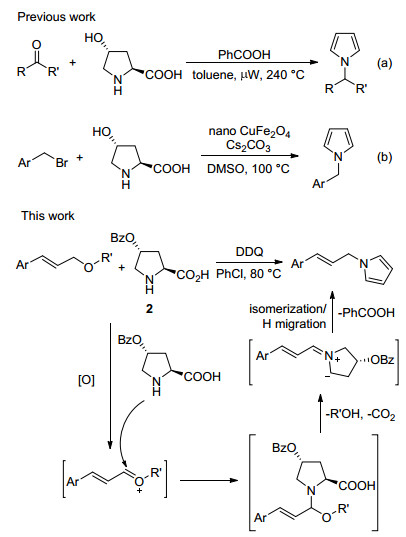





 DownLoad:
DownLoad: