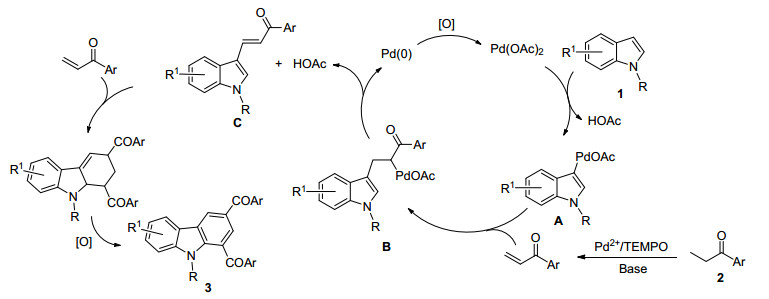
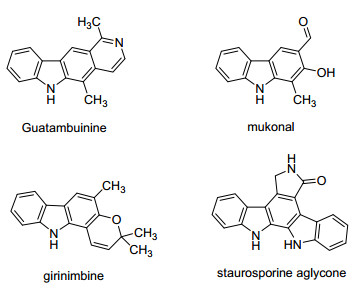
-
[1]
(a) Zhang, F. -F. ; Gan, L. -L. ; Zhou, C. -H. Bioorg. Med. Chem. Lett. 2010, 20, 1881.
(b) Blunt, J. W. ; Copp, B. R. ; Munro, M. H. G. ; Northcote, P. T. ; Prinsep, M. R. Nat. Prod. Rep. 2003, 20, 1.
(c) Deslandes, S. ; Chassaing, S. ; Delfourne, E. Mar. Drugs 2009, 7, 754.
(d) Maneerat, W. ; Ritthiwigrom, T. ; Cheenpracha, S. ; Promgool, T. ; Yossathera, K. ; Deachathai, S. ; Phakhodee, W. ; Laphookhieo, S. J. Nat. Prod. 2012, 75, 741.
-
[2]
(a) Roy, J. ; Jana, A. K. ; Mal, D. Tetrahedron 2012, 68, 6099.
(b) Knölker, H. -J. ; Reddy, K. R. Chem. Rev. 2002, 102, 4303.
(c) Schmidt, A. W. ; Reddy, K. R. ; Knölker, H. -J. Chem. Rev. 2012, 112, 3193.
-
[3]
(a) Ackermann, L. ; Vicente, R. ; Kapdi, A. R. Angew. Chem. , Int. Ed. 2009, 48, 9792.
(b) Lyons, T. W. ; Sanford, M. S. Chem. Rev. 2010, 110, 1147.
(c) Liu, C. ; Zhang, H. ; Shi, W. ; Lei, A. Chem. Rev. 2011, 111, 1780.
(d) Yeung, C. S. ; Dong, V. M. Chem. Rev. 2011, 111, 1215.
-
[4]
Zhao, J.; Larock, R. C. J. Org. Chem. 2006, 71, 5340.
doi: 10.1021/jo060727r
-
[5]
(a) Tsang, W. C. P. ; Zheng, N. ; Buchwald, S. L. J. Am. Chem. Soc. 2005,127, 14560.
(b) Jordan-Hore, J. A. ; Johansson, C. C. ; Beck, E. M. ; Gaunt, M. J. J. Am. Chem. Soc. 2008, 130, 16184.
(c) Cho, S. H. ; Yoon, J. ; Chang, S. J. Am. Chem. Soc. 2011, 133, 5996.
(d) Takamatsu, K. ; Hirano, K. ; Satoh, T. ; Miura, M. Org. Lett. 2014, 16, 2892.
-
[6]
(a) Yamashita, M. ; Horiguchi, H. ; Hirano, K. ; Satoh, T. ; Miura, M. J. Org. Chem. 2009, 74, 7481.
(b) Jia, J. ; Shi, J. ; Zhou, J. ; Liu, X. ; Song, Y. ; Xu, H. E. ; Yi, W. Chem. Commun. 2015, 51, 2925.
-
[7]
(a) Ozaki, K. ; Zhang, H. ; Ito, H. ; Lei, A. ; Itami, K. Chem. Sci. 2013, 4, 3416.
(b) Verma, A. K. ; Danodia, A. K. ; Saunthwal, R. K. ; Patel, M. ; Choudhary, D. Org. Lett. 2015, 17, 3658.
(c) Laha, J. K. ; Dayal, N. Org. Lett 2015, 17, 4742.
(d) Chen, S. ; Li, Y. ; Ni, P. ; Huang, H. ; Deng, G. J. Org. Lett 2016.
-
[8]
Grimster, N. P.; Gauntlett, C.; Godfrey, C. R.; Gaunt, M. J. Angew. Chem., Int. Ed. 2005, 44, 3125.
doi: 10.1002/(ISSN)1521-3773
-
[9]
Guo, T.; Jiang, Q.; Huang, F.; Chen, J.; Yu, Z. Org. Chem. Front. 2014, 1, 707.
doi: 10.1039/C4QO00122B
-
[10]
(a) Muzart, J. Eur. J. Org. Chem. 2010, 3779.
(b) Newhouse, T. ; Turlik, A. ; Chen, Y. Synlett 2016, 27, 331.
-
[11]
(a) Nicolaou, K. C. ; Gray, D. L. F. ; Montagnon, T. ; Harrison, S. T. Angew. Chem. , Int. Ed. 2002, 41, 996.
(b) Nicolaou, K. C. ; Montagnon, T. ; Baran, P. S. Angew. Chem. , Int. Ed. 2002, 41, 993.
(c) Nicolaou, K. C. ; Montagnon, T. ; Baran, P. S. ; Zhong, Y. L. J. Am. Chem. Soc. 2002, 124, 2245.
(d) Uyanik, M. ; Akakura, M. ; Ishihara, K. J. Am. Chem. Soc. 2009, 131, 251. (e) Nicolaou, K. C. ; Zhong, Y. L. ; Baran, P. S. J. Am. Chem. Soc. 2000, 122, 7596.
-
[12]
(a) Bhattacharya, A. ; DiMichele, L. M. ; Dolling, U. H. ; Douglas, A. W. ; Grabowski, E. J. J. J. Am. Chem. Soc. 1988, 110, 3318.
(b) Walker, D. ; Hiebert, J. D. Chem. Rev. 1967, 67, 153.
-
[13]
(a) Diao, T. ; Stahl, S. S. J. Am. Chem. Soc. 2011, 133, 14566.
(b) Gao, W. M. ; He, Z. Q. ; Qian, Y. ; Zhao, J. ; Huang, Y. Chem. Sci. 2012, 3, 883.
(c) Diao, T. ; Wadzinski, T. J. ; Stahl, S. S. Chem. Sci. 2012, 3, 887.
(d) Diao, T. ; Pun, D. ; Stahl, S. S. J. Am. Chem. Soc. 2013, 135, 8205.
(e) Bigi, M. A. ; White, M. C. J. Am. Chem. Soc. 2013, 135, 7831.
-
[14]
(a) Zhang, M. ; Zhang, Y. ; Jie, X. ; Zhao, H. ; Li, G. ; Su, W. Org. Chem. Front. 2014, 1, 843.
(b) Wei, Y. ; Hu, P. ; Zhang, M. ; Su, W. Chem. Rev. 2017.
-
[15]
(a) Zhang, M. ; Zhou, J. ; Kan, J. ; Wang, M. ; Su, W. ; Hong, M. Chem. Commun. 2010, 46, 5455.
(b) Zhou, J. ; Wu, G. ; Zhang, M. ; Jie, X. ; Su, W. Chem. -Eur. J. 2012, 18, 8032.
(c) Zhang, M. ; Hu, P. ; Zhou, J. ; Wu, G. ; Huang, S. ; Su, W. Org. Lett. 2013, 15, 1718.
(d) Shang, Y. ; Jie, X. ; Zhou, J. ; Hu, P. ; Huang, S. ; Su, W. Angew. Chem. Int. Ed. 2013, 52, 1299.
-
[16]
Jie, X.; Shang, Y.; Zhang, X.; Su, W. J. Am. Chem. Soc. 2016, 138, 5623.
doi: 10.1021/jacs.6b01337
-
[17]
Xiao, B.; Li, Y. M.; Liu, Z. J.; Yang, H. Y.; Fu, Y. Chem. Commun. 2012, 48, 4854.
doi: 10.1039/c2cc31737k
-
[18]
Klare, H. F.; Oestreich, M.; Ito, J.; Nishiyama, H.; Ohki, Y.; Tatsumi, K. J. Am. Chem. Soc. 2011, 133, 3312.
doi: 10.1021/ja111483r
-
[19]
Taylor, J. E.; Jones, M. D.; Williams, J. M. J.; Bull, S. D. Org. Lett. 2010, 12, 5740.
doi: 10.1021/ol1025348
-
[20]
Qi, T.; Qiu, W.; Liu, Y.; Zhang, H.; Gao, X.; Liu, Y.; Lu, K.; Du, C.; Yu, G.; Zhu, D. J. Org. Chem. 2008, 73, 4638.
doi: 10.1021/jo800622y

 Login In
Login In






 DownLoad:
DownLoad: