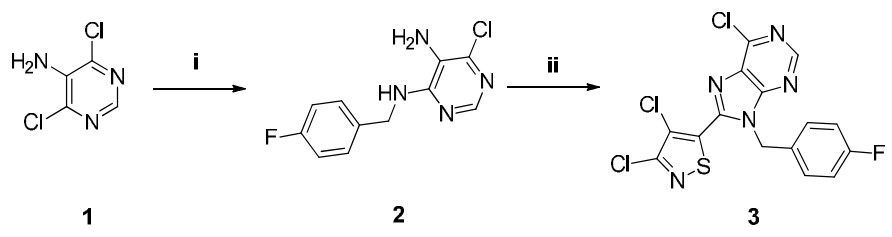
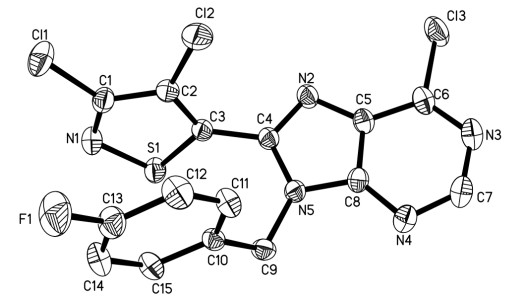
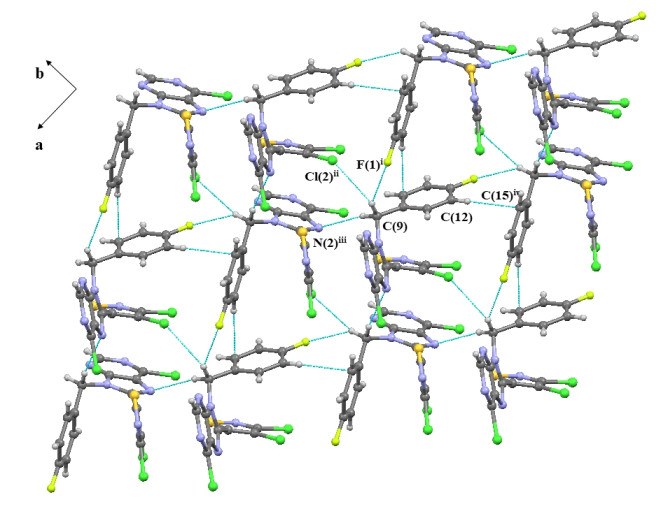
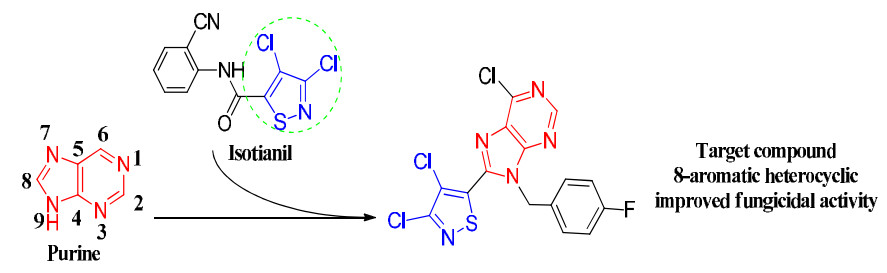
-
[1]
Rosemeyer, H. The chemodiversity of purine as a constituent of natural products. Chem. Biodiversity 2004, 1, 361–401.
doi: 10.1002/cbdv.200490033
-
[2]
Legraverend, M.; Grierson, D. S. The purines: potent and versatile small molecule inhibitors and modulators of key biological targets. Bioorg. Med. Chem. 2006, 14, 3987–4006.
doi: 10.1016/j.bmc.2005.12.060
-
[3]
Welsch, M. E.; Snyder, S. A.; Stockwell, B. R. Privileged scaffolds for library design and drug discovery. Curr. Opin. Chem. Biol. 2010, 14, 347–361.
doi: 10.1016/j.cbpa.2010.02.018
-
[4]
Hart, S.; Novotny-Diermayr, V.; Goh, K. C.; Williams, M.; Tan, Y. C.; Ong, L. C.; Cheong, A.; Ng, B. K.; Amalini, C.; Madan, B.; Nagaraj, H.; Jayaraman, R.; Pasha, K. M.; Ethirajulu, K.; Chng, W. J.; Mustafa, N.; Goh, B. C.; Benes, C.; McDermott, U.; Garnett, M.; Dymock, B.; Wood, J. M. VS-5584, a novel and highly selective PI3K/MTOR kinase inhibitor for the treatment of cancer. Mol. Cancer Ther. 2013, 12, 151–161.
-
[5]
Lawhorn, B. G.; Philp, J.; Zhao, Y.; Louer, C.; Hammond, M.; Cheung, M.; Fries, H.; Graves, A. P.; Shewchuk, L.; Wang, L.; Cottom, J. E.; Qi, H.; Zhao, H.; Totoritis, R.; Zhang, G.; Schwartz, B.; Li, H.; Sweitzer, S.; Holt, D. A.; Gatto, G. J.; Kallander, L. S. Identification of purines and 7-deazapurines as potent and selective type I inhibitors of troponin I-interacting kinase (TNNI3K). J. Med. Chem. 2015, 58, 7431–7448.
doi: 10.1021/acs.jmedchem.5b00931
-
[6]
Yoon, J. S.; Jarhad, D. B.; Kim, G.; Nayak, A.; Zhao, L. X.; Yu, J.; Kim, H. R.; Lee, J. Y.; Mulamoottil, V. A.; Chandra, G.; Byun, W. S.; Lee, S. K.; Kim, Y. C.; Jeong, L. S. Design, synthesis and anticancer activity of fluorocyclopentenyl-purines and-pyrimidines. Eur. J. Med. Chem. 2018, 155, 406–417.
doi: 10.1016/j.ejmech.2018.06.003
-
[7]
Malínková, V.; Řezníčková, E.; Jorda, R.; Gucky, T.; Kryštof, V. Trisubstituted purine inhibitors of PDGFRalpha and their antileukemic activity in the human eosinophilic cell line EOL-1. Bioorg. Med. Chem. 2017, 25, 6523–6535.
doi: 10.1016/j.bmc.2017.10.032
-
[8]
Calderon-Arancibia, J.; Espinosa-Bustos, C.; Canete-Molina, A.; Tapia, R. A.; Faundez, M.; Torres, M. J.; Aguirre, A.; Paulino, M.; Salas, C. O. Synthesis and pharmacophore modelling of 2, 6, 9-trisubstituted purine derivatives and their potential role as apoptosis-inducing agents in cancer cell lines. Molecules 2015, 20, 6808–6826.
doi: 10.3390/molecules20046808
-
[9]
Gucký, T.; Řezníčková, E.; Muchová, T. R.; Jorda, R.; Klejová, Z.; Malínková, V.; Berka, K.; Bazgier, V.; Ajani, H.; Lepšík, M.; Divoký, V.; Kryštof, V. Discovery of N2-(4-amino-cyclohexyl)-9-cyclopentyl-N6-(4-morpholin-4-ylmethyl-phenyl)-9H-purine-2, 6-diamine as a potent FLT3 kinase inhibitor for acute myeloid leukemia with FLT3 mutations. J. Med. Chem. 2018, 61, 3855–3869.
doi: 10.1021/acs.jmedchem.7b01529
-
[10]
Liu, B.; Zhu, F. C.; Huang, Y.; Wang, Y. H.; Yu, F.; Fan, B. T.; Yao, J. Screening rules for leads of fungicides, herbicides, andinsecticides. J. Agric. Food Chem. 2010, 58, 2673–2684.
doi: 10.1021/jf902639x
-
[11]
Sun, S. S.; Li, Q.; Gao, W.; Li, X. T.; Chen, L.; Zhang, J. L. Synthesis, crystal structure and anti-TMV activity of (Z)-4-[3-(4-methyl-1, 2, 3-thiadiazol-5-yl)-3-(4-trifluoromethylphenyl)acryloyl]morpholine. Chin. J. Struct. Chem. 2021, 40, 109–113.
-
[12]
Wu, Q. F.; Zhao, B.; Fan, Z. J.; Guo, X. F.; Yang, D. Y.; Zhang, N. L.; Yu, B.; Zhou, S.; Zhao, J. B.; Chen, F. Discovery of novel piperidinylthiazole derivatives as broad-spectrum fungicidal candidates. J. Agric. Food Chem. 2019, 67, 1360–1370.
doi: 10.1021/acs.jafc.8b06054
-
[13]
Yu, B.; Zhou, S.; Cao, L. X.; Hao, Z. S.; Yang, D. Y.; Guo, X. F.; Zhang, N. L.; Bakulev, V. A.; Fan, Z. J. Design, synthesis, and evaluation of the antifungal activity of novel pyrazole-thiazole carboxamides as succinate dehydrogenase inhibitors. J. Agric. Food Chem. 2020, 68, 7093–7102.
doi: 10.1021/acs.jafc.0c00062
-
[14]
Yu, B.; Zhao, B.; Hao, Z. S.; Chen, L.; Cao, L. X.; Guo, X. F.; Zhang, N. L.; Yang, D. Y.; Tang, L. F.; Fan, Z. J. Design, synthesis and biological evaluation of pyrazole-aromatic containing carboxamides as potent SDH inhibitors. Eur. J. Med. Chem. 2021, 214, 113230.
doi: 10.1016/j.ejmech.2021.113230
-
[15]
Chen, L.; Zhu, Y. J.; Fan, Z. J.; Guo, X. F.; Zhang, Z. M.; Xu, J. H.; Song, Y. Q.; Yurievich, M. Y.; Belskaya, N. P.; Bakulev, V. A. Synthesis of 1, 2, 3-thiadiazole and thiazole-based strobilurins as potent fungicide candidates. J. Agric. Food Chem. 2017, 65, 745–751.
doi: 10.1021/acs.jafc.6b05128
-
[16]
Fan, Z. J.; Yang, Z. K.; Zhang, H. K.; Mi, N.; Wang, H.; Cai, F.; Zuo, X.; Zheng, Q. X.; Song, H. B. Synthesis, crystal structure, and biological activity of 4-methyl-1, 2, 3-thiadiazole-containing 1, 2, 4-triazolo[3, 4-b][1, 3, 4]thiadiazoles. J. Agric. Food Chem. 2010, 58, 2630–2636.
doi: 10.1021/jf9029628
-
[17]
Zhao, B.; Fan, S.; Fan, Z.; Wang, H.; Zhang, N.; Guo, X.; Yang, D.; Wu, Q.; Yu, B.; Zhou, S. Discovery of pyruvate kinase as a novel target of new fungicide candidate 3-(4-methyl-1, 2, 3-thiadiazolyl)-6-trichloromethyl-[1, 2, 4]-triazolo-[3, 4-b][1, 3, 4]-thiadizole. J. Agric. Food Chem. 2018, 66, 12439–12452.
doi: 10.1021/acs.jafc.8b03797
-
[18]
Kelley, J. L.; Krochmal, M. P.; Linn, J. A.; McLean, E. W.; Soroko, F. E. 6-(Alkylamino)-9-benzyl-9H-purines. A new class of anticonvulsant agents. J. Med. Chem. 2002, 31, 606–612.
-
[19]
Ibrahim, N.; Legraverend, M. High-yielding two-step synthesis of 6, 8-disubstituted N-9-unprotected purines. J. Comb. Chem. 2009, 11, 658–666.
doi: 10.1021/cc900066v
-
[20]
Dolomanov, O. V.; Bourhis, L. J.; Gildea, R. J.; Howard, J. A. K.; Puschmann, H. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341.
doi: 10.1107/S0021889808042726
-
[21]
Sheldrick, G. M. SHELXT-integrated space-group and crystal-structure determination. Acta Cryst. 2015, A71, 3–8.
-
[22]
Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8.
-
[23]
Yang, D. Y.; Zhao, B.; Fan, Z. J.; Yu, B.; Zhang, N. L.; Li, Z. M.; Zhu, Y. L.; Zhou, J. H.; Kalinina, T. A.; Glukhareva, T. V. Synthesis and biological activity of novel succinate dehydrogenase inhibitor derivatives as potent fungicide candidates. J. Agric. Food Chem. 2019, 67, 13185–13194.
doi: 10.1021/acs.jafc.9b05751
-
[24]
Fan, Z.; Yang, Z.; Zhang, H.; Mi, N.; Wang, H.; Cai, F.; Zuo, X.; Zheng, Q.; Song, H. Synthesis, crystal structure, and biological activity of 4-methyl-1, 2, 3-thiadiazole-containing 1, 2, 4-triazolo[3, 4-b][1, 3, 4]thiadiazoles. J. Agric. Food Chem. 2010, 58, 2630–2636.
-
[25]
Wu, Q.; Zhao, B.; Fan, Z.; Guo, X.; Yang, D.; Zhang, N.; Yu, B.; Zhou, S.; Zhao, J.; Chen, F. Discovery of novel piperidinyl-thiazole derivatives as broad-spectrum fungicidal candidate. J. Agric. Food Chem. 2019, 67, 1360–1370.
-
[26]
Zong, G. N.; Li, F. Y.; Fan, Z. J.; Mao, W. T.; Song, H. B.; Chen, L.; Zhu, Y. J.; Xu, J. H.; Song, Y. Q.; Wang, J. R. Synthesis, crystal structure and biological activity of 2-(3, 4-dichloroisothiazol-5-yl)-4-(trifluoromethyl)-4, 5-dihydrothiazol-4-yl 3-methylbenzoate. Chin. J. Struct. Chem. 2015, 34, 871–878.
-
[27]
Standara, S.; Maliňáková, K.; Marek, R.; Marek, J.; Hocek, M.; Vaara, J.; Straka, M. Understanding the NMR chemical shifts for 6-halopurines: roleof structure, solvent and relativistic effects. Phys. Chem. Chem. Phys. 2010, 12, 5126–5139.

 Login In
Login In






 DownLoad:
DownLoad: